Abstract
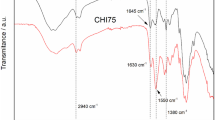
Chitosan (CTS) gel was successfully prepared by organic titanium (OT) ionic crosslinking agent. The viscoelastic properties of CTS gel was investigated through rheological measurement. The sol-gel transition point (t cr ) of gel prepared with CTS (22.5 g•L−1) and OT (1.5 %) was determined at 339 s, with the increases of either CTS or OT concentration, the gelation became faster and the sol-gel transition time became shorter. The optimal chitosan gel beads were prepared with 35 g•L−1 chitosan and 7 % organic titanium (OT) at room temperature for 1 h reaction. The SEM data indicated that the gel beads had special shape with rough and poriferous surface. The effective grain size range was 1.0–1.2 mm. It was confirmed by TG data that the gel beads performed better in thermal stability than original chitosan. Swelling experiments showed that the swelling ability of gel beads strongly depended on the pH value of buffer solution. On the whole, the modified CTS/OT gel beads showed high adsorption capacity, good mechanical properties, strong acidic and alkali resistances, it could serve as potential drug deliver and the absorbent for heavy metal ions.











Similar content being viewed by others
References
Boucard N, Viton C, Domard A (2005) New aspects of the formation of physical hydrogels of chitosan in a hydroalcoholic medium. Biomacromolecules 6(6):3227–3237
Kurita K (2006) Chitin and chitosan: functional biopolymers from marine crustaceans. Mar Biotechnol (N Y) 8(3):203–226
Xu Y, Zhan C, Fan L, Wang L, Zheng H (2007) Preparation of dual crosslinked alginate-chitosan blend gel beads and in vitro controlled release in oral site-specific drug delivery system. Int J Pharm 336(2):329–337
Liang S, Liu L, Huang Q, Yam KL (2009) Preparation of single or double-network chitosan/poly(vinyl alcohol) gel films through selectively cross-linking method. Carbohydr Polym 77(4):718–724
Barreiro-Iglesias R, Coronilla R, Concheiro A, Alvarez-Lorenzo C (2005) Preparation of chitosan beads by simultaneous cross-linking/insolubilisation in basic pH: rheological optimisation and drug loading/release behaviour. Eur J Pharm Sci 24(1):77–84
Liang ZP, Feng YQ, Meng SX, Liang ZY (2005) Preparation and properties of urease immobilized onto glutaraldehyde cross-linked chitosan beads. Chin Chem Lett 16(1):135–138
Gupta NV, Shivakumar HG (2010) Development of a gastroretentive drug delivery system based on superporous hydrogel. Trop J Pharm Res 9(3):257–264
Umadevi SK, Thiruganesh R, Suresh S, Reddy KB (2010) Formulation and evaluation of chitosan microspheres of aceclofenac for colon-targeted drug delivery. Biopharm Drug Dispos 31(7):407–427
Rinaudo M (2010) New way to crosslink chitosan in aqueous solution. Eur Polym J 46(7):1537–1544
Chenite A, Wang D, Chaput C, Kandani N (2001) Rheological characterisation of thermogelling chitosan/glycerol-phosphate solutions. Carbohydr Polym 46(1):39–47
Cho J, Heuzey MC, Bégin A, Carreau PJ (2005) Physical gelation of chitosan in the presence of beta-glycerophosphate: the effect of temperature. Biomacromolecules 6(6):3267–3275
Gupta KC, Jabrail FH (2006) Effects of degree of deacetylation and cross-linking on physical characteristics, swelling and release behavior of chitosan microspheres. Carbohydr Polym 66(1):43–45
Wu J, Su Z-G, Ma G-H (2006) A thermo- and pH-sensitive hydrogel composed of quaternized chitosan/glycerophosphate. Int J Pharm 315(1–2):1–11
Chen S, Liu M, Jin S, Wang B (2008) Preparation of ionic-crosslinked chitosan-based gel beads and effect of reaction conditions on drug release behaviors. Int J Pharm 349(1–2):180–187
Shu XZ, Zhu KJ (2002) Controlled drug release properties of ionically cross-linked chitosan beads: the influence of anion structure. Int J Pharm 233(1):217–225
Wu Z, Li S, Wan J, Wang Y (2012) Cr(VI) adsorption on an improved synthesized cross-linked chitosan resin. J Mol Liq 170:25–29
Schmuhl R, Krieg HM, Keizer K (2001) Adsorption of Cu(II) and Cr(VI) ions by chitosan: kinetics and equilibrium studies. Water SA 27(1):1–7
Webster A, Halling MD, Grant DM (2007) Metal complexation of chitosan and its glutaraldehyde cross-linked derivative. Carbohydr Res 342(9):1189–1201
Deleanu C, Simonescu CM, Constantinescu I (2008) Adsorption behaviour of Cu(II) ions from aqueous solution on chitosan. Revista Chimie 59(6):639–642
Chenite A, Gori S, Shive M, Desrosiers E, Buschmann MD (2006) Monolithic gelation of chitosan solutions via enzymatic hydrolysis of urea. Carbohydr Polym 64(3):419–424
Köpf H, Köpf-Maier P (1979) Titanocene dichloride -the first metallocene with cancerostatic activity. Journal Article 18(6):477–478
Shibata Y, Suzuki D, Omori S, Tanaka R, Murakami A, Kataoka Y, Baba K, Kamijo R, Miyazaki T (2010) The characteristics of in vitro biological activity of titanium surfaces anodically oxidized in chloride solutions. Biomaterials 31(33):8546–8555
Fang J, Zhao J, Sun Y, Ma H, Yu X, Ma Y, Ni Y, Zheng L, Zhou Y (2014) Biocompatibility and antibacterial properties of zinc-ion implantation on titanium. J Hard Tissue Biol 23(1):35–43
Felinto MCFC, Parra DF, da Silva CC, Angerami J, Oliveira MJA, Lugão AB (2007) The swelling behavior of chitosan hydrogels membranes obtained by UV- and γ-radiation. Nucl Instrum Methods Phys Res Sect B: Beam Interactions with Materials and Atoms 265(1):418–424
Doi M (1980) Molecular rheology of concentrated polymer systems. J Polym Sci B Polym Phys 18(5):1005–1020
Yu L, Wang D, Li H, Liu B, Wang X, Xu Y (2010) Preparation and characterization of magnetic resin made from chitosan and cerium. J Ocean Univ China (English Edition) 9(2):185–192
Chambon F, Henning Winter H (1985) Stopping of crosslinking reaction in a PDMS polymer at the gel point. Polym Bull 13(6):499–503
Winter HH, Mours M (1997) Rheology of polymers near liquid–solid transitions. Adv Polym Sci 134:165–234
Te Nijenhuis K, Winter HH (1989) Mechanical properties at the gel point of a crystallizing poly(vinyl chloride) solution. Macromolecules 22(1):411–414
Andrew B, Rodd DED, Ross-Murphy SB, Boger DV (2001) Dependence of linear viscoelastic critical strain and stress values on extent of gelation for a thermally activated gelling system. Rheol Acta 40(1):23–29
Meena A, Parikh T, Gupta SS, Serajuddin ATM (2014) Investigation of thermal and viscoelastic properties of polymers relevant to hot melt extrusion - II: cellulosic polymers. J Excipients Food Chem 5(1):46–55
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, Y., Shen, C. & Gao, S. Preparation and characterization of chitosan gel beads crosslinked by organic titanium. J Polym Res 22, 53 (2015). https://doi.org/10.1007/s10965-015-0693-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-015-0693-7