Abstract
Covariate models for population pharmacokinetics and pharmacodynamics are often built with a stepwise covariate modelling procedure (SCM). When analysing a small dataset this method may produce a covariate model that suffers from selection bias and poor predictive performance. The lasso is a method suggested to remedy these problems. It may also be faster than SCM and provide a validation of the covariate model. The aim of this study was to implement the lasso for covariate selection within NONMEM and to compare this method to SCM.
In the lasso all covariates must be standardised to have zero mean and standard deviation one. Subsequently, the model containing all potential covariate–parameter relations is fitted with a restriction: the sum of the absolute covariate coefficients must be smaller than a value, t. The restriction will force some coefficients towards zero while the others are estimated with shrinkage. This means in practice that when fitting the model the covariate relations are tested for inclusion at the same time as the included relations are estimated. For a given SCM analysis, the model size depends on the P-value required for selection. In the lasso the model size instead depends on the value of t which can be estimated using cross-validation.
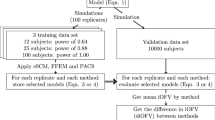
The lasso was implemented as an automated tool using PsN. The method was compared to SCM in 16 scenarios with different dataset sizes, number of investigated covariates and starting models for the covariate analysis. Hundred replicate datasets were created by resampling from a PK-dataset consisting of 721 stroke patients. The two methods were compared primarily on the ability to predict external data, estimate their own predictive performance (external validation), and on the computer run-time.
In all 16 scenarios the lasso predicted external data better than SCM with any of the studied P-values (5%, 1% and 0.1%), but the benefit was negligible for large datasets. The lasso cross-validation provided a precise and nearly unbiased estimate of the actual prediction error. On a single processor, the lasso was faster than SCM. Further, the lasso could run completely in parallel whereas SCM must run in steps.
In conclusion, the lasso is superior to SCM in obtaining a predictive covariate model on a small dataset or on small subgroups (e.g. rare genotype). Run in parallel the lasso could be much faster than SCM. Using cross-validation, the lasso provides a validation of the covariate model and does not require the user to specify a P-value for selection.
Similar content being viewed by others
References
Jonsson EN and Karlsson MO (1998). Automated covariate model building within NONMEM. Pharm Res 15(9): 1463–1468
Administration FaD (1999) Guidance for industry, population pharmacokinetics (http://www.fda.gov/cder/guidance/1852fnl.pdf)
Ribbing J and Jonsson EN (2004). Power, selection bias and predictive performance of the population pharmacokinetic covariate model. J Pharmacokinet Pharmacodyn 31(2): 109–134
O’Neill R and Wetherill GB (1971). The present state of multiple comparison methods. J R Stat Soc Ser B (Methodological) 33(2): 218–250
Steyerberg EW, Eijkemans MJ and Habbema JD (1999). Stepwise selection in small data sets: a simulation study of bias in logistic regression analysis. J Clin Epidemiol 52(10): 935–942
Picard RR and Berk KN (1990). Data Splitting. Am Statist 44(2): 140–147
Sauerbrei W (1999). The use of resampling methods to simplify regression models in medical statistics. J R Stat Soc Ser C-Appl Stat 48: 313–329
Tibshirani R (1996). Regression shrinkage and selection via the lasso. J R Stat Soc Ser B (Methodological) 58(1): 267–288
Kowalski KG and Hutmacher MM (2001). Efficient screening of covariates in population models using Wald’s approximation to the likelihood ratio test. J Pharmacokinet Pharmacodyn 28(3): 253–275
le Cessie S, Vanhouwelingen JC (1992). Ridge estimators in logistic-regression. Appl Stat-J R Stat Soc Ser C 41(1): 191–201
Van Houwelingen JC and Cessie S (1990). Predictive value of statistical-models. Stat Med 9(11): 1303–1325
Holford NH (1996). A size standard for pharmacokinetics. Clin Pharmacokinet 30(5): 329–332
Brendel K, Comets E, Laffont C, Laveille C and Mentre F (2006). Metrics for external model evaluation with an application to the population pharmacokinetics of gliclazide. Pharm Res 23(9): 2036–2049
Mentre F and Escolano S (2006). Prediction discrepancies for the evaluation of nonlinear mixed-effects models. J Pharmacokinet Pharmacodyn 33(3): 345–367
Yano Y, Beal SL and Sheiner LB (2001). Evaluating pharmacokinetic/pharmacodynamic models using the posterior predictive check. J Pharmacokinet Pharmacodyn 28(2): 171–192
Sheiner LB and Beal SL (1981). Some suggestions for measuring predictive performance. J Pharmacokinet Biopharm 9(4): 503–512
Bruno R, Vivler N, Vergniol JC, De Phillips SL, Montay G and Sheiner LB (1996). A population pharmacokinetic model for docetaxel (Taxotere): model building and validation. J Pharmacokinet Biopharm 24(2): 153–172
Steyerberg EW, Eijkemans MJC, Harrell FE and Habbema JDF (2000). Prognostic modelling with logistic regression analysis: a comparison of selection and estimation methods in small data sets. Stat Med 19(8): 1059–1079
Matthews I, Kirkpatrick C and Holford N (2004). Quantitative justification for target concentration intervention–parameter variability and predictive performance using population pharmacokinetic models for aminoglycosides. Br J Clin Pharmacol 58(1): 8–19
Harrell FE (2001) 5.2 Model Validation. In: Regression modeling strategies. Springer-Verlag, New York, pp 90–97
Bleeker SE, Moll HA, Steyerberg EW, Donders AR, Derksen-Lubsen G and Grobbee DE (2003). External validation is necessary in prediction research: a clinical example. J Clin Epidemiol 56(9): 826–832
Steyerberg EW, Bleeker SE, Moll HA, Grobbee DE and Moons KG (2003). Internal and external validation of predictive models: a simulation study of bias and precision in small samples. J Clin Epidemiol 56(5): 441–447
Breiman L and Spector P (1992). Submodel selection and evaluation in regression - the X-random case. Int Stat Rev 60(3): 291–319
Hastie T, Tibshirani R, Friedman J (2001) 7.10 Cross-validation. In: The elements of statistical learning. Springer-Verlag, New York, pp 214–217
Hjorth JSU (1994) Computer intensive statistical methods. Chapman & Hall
Hastie T, Tibshirani R, Friedman J (2001) The elements of statistical learning. Springer-Verlag
Bies RR, Muldoon MF, Pollock BG, Manuck S, Smith G and Sale ME (2006). A genetic algorithm-based, hybrid machine learning approach to model selection. J Pharmacokinet Pharmacodyn 33(2): 195–221
Ribbing J, Jonsson EN (2001) Cross model validation as a tool for population pharmacokinetic/pharmacodynamic covariate model building. PAGE 10. Abstr 215 [www.page-meeting.org/?abstract=215]
Karlsson MO and Sheiner LB (1993). The importance of modeling interoccasion variability in population pharmacokinetic analyses. J Pharmacokinet Biopharm 21(6): 735–750
Mandema JW, Verotta D and Sheiner LB (1992). Building population pharmacokinetic–pharmacodynamic models. I. Models for covariate effects. J Pharmacokinet Biopharm 20(5): 511–528
Zingmark PH, Ekblom M, Odergren T, Ashwood T, Lyden P and Karlsson MO (2003). Population pharmacokinetics of clomethiazole and its effect on the natural course of sedation in acute stroke patients. Br J Clin Pharmacol 56(2): 173–183
Lindbom L, Pihlgren P and Jonsson EN (2005). PsN-Toolkit – a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Programs Biomed 79(3): 241–257
Lindbom L, Ribbing J and Jonsson EN (2004). Perl-speaks-NONMEM (PsN) – a Perl module for NONMEM related programming. Comput Methods Programs Biomed 75(2): 85–94
Rodgers JL (1999). The bootstrap, the jackknife and the randomization test: a sampling taxonomy. Multivariate Behav Res 34(4): 441–456
Shao J (1996). Bootstrap model selection. J Am Stat Assoc 91(434): 655–665
Janmahasatian S, Duffull SB, Ash S, Ward LC, Byrne NM and Green B (2005). Quantification of lean bodyweight. Clin Pharmacokinet 44(10): 1051–1065
Green B and Duffull SB (2004). What is the best size descriptor to use for pharmacokinetic studies in the obese?. Br J Clin Pharmacol 58(2): 119–133
Wahlby U, Jonsson EN and Karlsson MO (2001). Assessment of actual significance levels for covariate effects in NONMEM. J Pharmacokinet Pharmacodyn 28(3): 231–252
Wahlby U, Jonsson EN and Karlsson MO (2002). Comparison of stepwise covariate model building strategies in population pharmacokinetic-pharmacodynamic analysis. AAPS PharmSci 4(4): 27
Wade JR, Beal SL and Sambol NC (1994). Interaction between structural, statistical, and covariate models in population pharmacokinetic analysis. J Pharmacokinet Biopharm 22(2): 165–177
Altman DG and Andersen PK (1989). Bootstrap investigation of the stability of a Cox regression model. Stat Med 8(7): 771–783
Efron B and Tibshirani R (1997). Improvements on cross-validation: The .632+ bootstrap method. J Am Stat Assoc 92(438): 548–560
Harrell FE (2001) 9.10 Penalized maximum likelihood estimation. In Regression modeling strategies. Springer-Verlag, New York, pp 207–210
Efron B, Hastie T, Johnstone I and Tibshirani R (2004). Least angle regression. Ann Stat 32(2): 407–451
Olofsen E (2006) Using the Lasso to simultaneously identify the covariate and variance-covariance structures of nonlinear mixed-effects models. PAGE 15. Abstr 999 [www.page- meeting.org/?abstract = 999]
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ribbing, J., Nyberg, J., Caster, O. et al. The lasso—a novel method for predictive covariate model building in nonlinear mixed effects models. J Pharmacokinet Pharmacodyn 34, 485–517 (2007). https://doi.org/10.1007/s10928-007-9057-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-007-9057-1