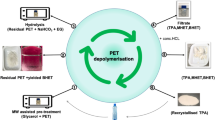
Abstract
In this study, the glycolysis of polyethylene terephthalate was studied in presence of deep eutectic solvents as catalyst. In the glycolysis of PET, five different deep eutectic solvents were synthesized using different hydrogen bond donors and acceptors. Among the synthesized DESs, the most efficient catalyst was found to be DES formed by potassium carbonate and ethylene glycol. Glycolysis reaction was performed between PET and ethylene glycol. The effects of process parameters such as temperature, ratio of EG/PET and ratio of DES/PET were examined. The maximum yield for monomer product, bis(2-hydroxyethyl) terephthalate was observed as 88% at the reaction temperature of 180 °C, ethylene glycol/deep eutectic solvent ratio of 15 and deep eutectic solvent/polyethylene terephthalate ratio of 6.





Similar content being viewed by others
References
Zhu M, Li Z, Wang Q, Zhou X, Lu X (2012) Characterization of solid acid catalysts and their reactivity in the glycolysis of poly(ethylene terephthalate). Ind Eng Chem Res 51(36):11659–11666
Imran M, Kim BK, Han M, Cho BG, Kim DH (2010) Sub-and supercritical glycolysis of polyethylene terephthalate (PET) into the monomer bis(2-hydroxyethyl) terephthalate (BHET). Polym Degrad Stab 95(9):1685–1693
Grigore M (2017) Methods of recycling, properties and applications of recycled thermoplastic polymers. Recycling 2(4):24
Ragaert K, Delva L, Van Geem K (2017) Mechanical and chemical recycling of solid plastic waste. Waste Manage 69:24–58
Raheem AB, Noor ZZ, Hassan A, Abd Hamid MK, Samsudin SA, Sabeen AH (2019) Current developments in chemical recycling of post-consumer polyethylene terephthalate wastes for new materials production: a review. J Clean Prod 225:1052–1064
Wang Q, Yao X, Geng Y, Zhou Q, Lu X, Zhang S (2015) Deep eutectic solvents as highly active catalysts for the fast and mild glycolysis of poly(ethylene terephthalate)(PET). Green Chem 17(4):2473–2479
Goje AS (2005) Recycling of waste poly(ethylene terephthalate) with naphthalene and neutral water. Polym Plast Technol Eng. 44(8–9):1643–1655
Genta M, Iwaya T, Sasaki M, Goto M, Hirose T (2005) Depolymerization mechanism of poly(ethylene terephthalate) in supercritical methanol. Ind Eng Chem Res 44(11):3894–3900
Shukla SR, Harad AM (2006) Aminolysis of polyethylene terephthalate waste. Polym Degrad Stab 91(8):1850–1854
Jain A, Soni RK (2007) Spectroscopic investigation of end products obtained by ammonolysis of poly (ethylene terephthalate) waste in the presence of zinc acetate as a catalyst. J Polym Res 14(6):475–481
López-Fonseca R, Duque-Ingunza I, de Rivas B, Flores-Giraldo L, Gutiérrez-Ortiz JI (2011) Kinetics of catalytic glycolysis of PET wastes with sodium carbonate. Chem Eng J 168(1):312–320
Troev K, Grancharov G, Tsevi R, Gitsov I (2003) A novel catalyst for the glycolysis of poly(ethylene terephthalate). J Appl Polym Sci 90(8):2301–2301
Viana ME, Riul A, Carvalho GM, Rubira AF, Muniz EC (2011) Chemical recycling of PET by catalyzed glycolysis: kinetics of the heterogeneous reaction. Chem. Eng. J. 173(1):210–219
Sangalang A, Bartolome L, Kim DH (2015) Generalized kinetic analysis of heterogeneous PET glycolysis: nucleation-controlled depolymerization. Polym Degrad Stab 115:45–53
Ghaemy M, Mossaddegh K (2005) Depolymerisation of poly(ethylene terephthalate) fibre wastes using ethylene glycol. Polym Degrad Stab 90(3):570–576
Yoshioka T, Handa T, Grause G, Lei Z, Inomata H, Mizoguchi T (2005) Effects of metal oxides on the pyrolysis of poly(ethylene terephthalate). J Anal Appl Pyrolysis 73(1):139–144
Yue QF, Wang CX, Zhang LN, Ni Y, Jin YX (2011) Glycolysis of poly(ethylene terephthalate) (PET) using basic ionic liquids as catalysts. Polym Degrad Stab 96(4):399–403
Yue QF, Xiao LF, Zhang ML, Bai XF (2013) The glycolysis of poly(ethylene terephthalate) waste: Lewis acidic ionic liquids as high efficient catalysts. Polymers (Basel) 5(4):1258–1271
Abbott AP, Boothby D, Capper G, Davies DL, Rasheed RK (2004) Deep Eutectic Solvents formed between choline chloride and carboxylic acids: Versatile alternatives to ionic liquids. J Am Chem Soc 126(29):9142–9147
Yusof R, Abdulmalek E, Sirat K, Rahman MBA (2014) Tetrabutylammonium bromide (TBABr)-based deep eutectic solvents (DESs) and their physical properties. Molecules 19(6):8011–8026
Musale RM, Shukla SR (2016) Deep eutectic solvent as effective catalyst for aminolysis of polyethylene terephthalate (PET) waste. Int J Plast Technol 20(1):106–120
Mjalli FS, Naser J, Jibril B, Al-Hatmi SS, Gano ZS (2014) Ionic liquids analogues based on potassium carbonate. Thermochim Acta 575:135–143
Hayyan M, Abo-Hamad A, AlSaadi MAH, Hashim MA (2015) Functionalization of graphene using deep eutectic solvents. Nanoscale Res Lett 10(1):324
Troter DZ, Todorović ZB, Đokić-Stojanović DR, Veselinović LM, Zdujić MV, Veljković VB (2018) Choline chloride-based deep eutectic solvents in CaO-catalyzed ethanolysis of expired sunflower oil. J Mol Liq 266:557–567
Chemat F, Anjum H, Shariff AM, Kumar P, Murugesan T (2016) Thermal and physical properties of (choline chloride + urea + l-arginine) deep eutectic solvents. J Mol Liq 218:301–308
Troter DZ et al (2017) The physicochemical and thermodynamic properties of the choline chloride-based deep eutectic solvents. J Serbian Chem Soc 82(9):1039–1052
Harifi-Mood AR, Buchner R (2017) Density, viscosity, and conductivity of choline chloride + ethylene glycol as a deep eutectic solvent and its binary mixtures with dimethyl sulfoxide. J Mol Liq 225:689–695
Yadav A, Trivedi S, Rai R, Pandey S (2014) Densities and dynamic viscosities of (choline chloride+glycerol) deep eutectic solvent and its aqueous mixtures in the temperature range (283.15-363.15)K. Fluid Phase Equilib 367:135–142
Khoonkari M, Haghighi AH, Sefidbakht Y, Shekoohi K, Ghaderian A (2015) Chemical recycling of PET wastes with different catalysts. Int J Polym Sci https://doi.org/10.1155/2015/124524
Al-Sabagh AM et al (2014) Glycolysis of poly(ethylene terephthalate) catalyzed by the Lewis base ionic liquid [Bmim][OAc]. Ind Eng Chem Res 53(48):18443–18451
Eshaq G, Elmetwally AE (2016) (Mg-Zn)-Al layered double hydroxide as a regenerable catalyst for the catalytic glycolysis of polyethylene terephthalate. J Mol Liq 214:1–6
Yunita I, Putisompon S, Chumkaeo P, Poonsawat T, Somsook E (2019) Effective catalysts derived from waste ostrich eggshells for glycolysis of post-consumer PET bottles. Chem Pap 73(6):1547–1560
Chen F, Zhou Q, Bu R, Yang F, Li W (2015) Kinetics of poly(ethylene terephthalate) fiber glycolysis in ethylene glycol. Fibers Polym 16(6):1213–1219
Acknowledgements
This study was supported by Ege University 18MÜH025 scientific research project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sert, E., Yılmaz, E. & Atalay, F.S. Chemical Recycling of Polyethlylene Terephthalate by Glycolysis Using Deep Eutectic Solvents. J Polym Environ 27, 2956–2962 (2019). https://doi.org/10.1007/s10924-019-01578-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-019-01578-w