Abstract
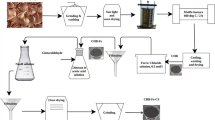
A new green composite microsphere (SA/ML-BT) was prepared from sodium alginate by interspersing modified lignin–bentonite clay (ML-BT) and its adsorption performance to Hg (II) from aqueous solution in a batch mode and column mode was systematically investigated. The morphology and structure of the SA/ML-BT were characterized by SEM, XRD and FTIR. BET analysis showed that the pore type of SA/ML-BT is mainly mesoporous. The enlarged surface area of 5.98 m2/g and the insertion of SA improved the attraction of Hg (II) to porous surface. In batch tests, different initial Hg (II) concentration, pH values and contact time were studied, which were fitted with pseudo-first-order, pseudo-second-order kinetic models and Langmuir/Freundlich isotherms. Zeta potential analysis showed that isoelectric point charge of SA/ML-BT was 3.24 and Hg (II) removal rate increased with pH increasing in the range of 1 to 3 then kept the maximum value of 93% in the range of 3 to 9. Likewise, in the fixed-bed column tests, the effects of initial concentration (10–30 mg/L), bed height (1–3 cm) and influent flow rate (0.5–1.5 mL/min) were evaluated, which were fitted with Yoon–Nelson and Bohart–Adam models. The SA/ML-BT remained 85% removal rate after three repeated cycles, which indicated a promising potential in sustainable applications. A removal capacity of 24.4 mg/g in batch and 42.1 mg/g in column tests were obtained respectively. These findings revealed that the proposed SA/ML-BT has great potential for metallic cation remediation.
Graphic Abstract














Similar content being viewed by others
References
J. Choi, A. Ide, Y.B. Truong, I.L. Kyratzis, R.A. Caruso, J. Mater Chem A. 1, 19 (2013)
N. Bolan, A. Kunhikrishnan, R. Thangarajan, J. Kumpiene, J. Park, T. Makino, M.B. Kirkham, K. Scheckel, J. Hazard Mater. 266, 141–166 (2014)
D.S. Tavares, C.B. Lopes, A.L. Daniel-da-Silva, A.C. Duarte, T. Trindade, E. Pereira, Chem Eng J. 254, 559–570 (2014)
Y. Ge, S. Wu, L. Qin, Z. Li, J. TaiWan Inst Chem E. 63, 500–505 (2016)
F. Karkeh-abadi, S. Saber-Samandari, S. Saber-Samandari, J. Hazard Mater. 312, 224–233 (2016)
Q. Wang, J. Ju, Y. Tan, L. Hao, Y. Ma, Y. Wu, H. Zhang, Y. Xia, K. Sui, J. Carbohydr. Polym. 205, 125–134 (2019)
M.M. Alghamdi, A.A. El-Zahhar, A.M. Idris, T.O. Said, T. Sahlabji, A. El Nemr, Sep. Purif. Technol. 224, 356–365 (2019)
R.R. Pawar, M. Kim, J.G. Kim, S.M. Hong, S.Y. Sawant, S.M. Lee, Appl. Clay Sci. 162, 339–350 (2018)
M.H. Sowlat, B. Kakavandi, S. Lotfi, M. Yunesian, M. Abdollahi, R.R. Kalantary, Environ. Sci. Pollut. Res. 24(13), 12092–12103 (2017)
M. Ahmadi, M.H. Niari, B. Kakavandi, J. Mol. Liq. 248, 184–196 (2017)
M. Ahmadi, M. Foladivanda, N. Jaafarzadeh, Z. Ramezani, B. Ramavandi, S. Jorfi, B. Kakavandi, J. Water Supply 66(2), 116–130 (2017)
Z.-J. Shao, X.-L. Huang, F. Yang, W.-F. Zhao, X.-Z. Zhou, C.-S. Zhao, J. Carbohydr. Polym. 187, 85–93 (2018)
M. Xie, L. Zeng, Q. Zhang, Y. Kang, H. Xiao, Y. Peng, X. Chen, J. Luo, J. Alloys Compds. 647, 892–905 (2015)
B. Yoosuk, P. Methakhup, P. Prasassarakich, Process Saf. Environ. 106, 173–179 (2017)
D. Zheng, B. Bai, X. Xu, Y. He, S. Li, N. Hu, H. Wang, Rsc Adv. 9(48), 27961–27972 (2019)
Y. Kong, L. Wang, Y. Ge, H. Su, Z. Li, J. Hazard Mater. 368, 33–41 (2019)
S. Mallakpour, V. Behranvand, F. Mallakpour, Carbohydr. Polym. 224, 115138 (2019)
W. Zhou, H. Zhang, Y. Liu, X. Zou, J. Shi, Y. Zhao, Y. Ye, Y. Yu, J. Guo, Carbohydr. Polym. 226, 115277 (2019)
C. Yadav, P.K. Maji, Carbohydr. Polym. 203, 396–408 (2019)
J. Zhang, R.J. Deng, B.Z. Ren, M. Yaseen, A. Hursthouse, Processes 8, 41 (2020)
G. Zhang, Y. Gao, Y. Zhang, Y. Guo, Environ. Sci. Technol. 44(16), 6384–6389 (2010)
Y. Fernandez-Nava, M. Ulmanu, I. Anger, E. Maranon, L. Castrillon, Water Air Soil Pollut. 215(1–4), 239–249 (2011)
M. Brigante, M. Avena, Micropor Mesopor Mat. 225, 534–542 (2016)
D.S.P. Franco, J.L.S. Fagundes, J. Georgin, N.P.G. Salau, G.L. Dotto, Chem. Eng. J. 397, 125423 (2020)
A. Goshadrou, A. Moheb, Desalination 269(1–3), 170–176 (2011)
A.A. Ahmad, B.H. Hameed, J. Hazard. Mater. 175(1–3), 298–303 (2010)
A.A. Ahmad, B.H. Hameed, J. Hazard Mater. 175(1–3), 298–303 (2010)
M. Auta, B.H. Hameed, Chem. Eng. J. 237, 352–361 (2014)
J.Z. Ma, M.A. Khan, M.Z. Xia, C.L. Fu, S.D. Zhu, Y.T. Chu, W. Lei, F.Y. Wang, Int. J. Biol. Macromol. 138, 188–197 (2019)
J. Ryu, M.Y. Lee, M.G. Song, S.H. Baeck, S.E. Shim, Y.J. Qian, Sep. Purif. Technol. 250, 117120 (2020)
J.U.K. Oubagaranadin, N. Sathyamurthy, Z.V.P. Murthy, J. Hazard Mater. 142(1–2), 165–174 (2007)
T. Phothitontimongkol, N. Siebers, N. Sukpirom, F. Unob, Appl. Clay Sci. 43(3–4), 343–349 (2009)
M. Monier, Int. J. Biol. Macromol. 50(3), 773–781 (2012)
N. Bicak, D.C. Sherrington, B.F. Senkal, React. Funct. Polym. 41(1–3), 69–76 (1999)
I. Ghodbane, O. Hamdaoui, J. Hazard Mater. 160(2–3), 301–309 (2008)
Acknowledgements
We acknowledge the supports from the National Natural Science Foundation of China (Nos. 21978057, 21707022), and Scientific Research Foundation of Guangxi Province (Nos. 2018GXNSFAA294101), and Guangxi Key Laboratory of Petrochemical Resource Processing and Process Intensification Technology (No.2018Z003), and 100-Talent Program of Guangxi, China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gong, L., Kong, Y., Wu, H. et al. Sodium Alginate Microspheres Interspersed with Modified Lignin and Bentonite (SA/ML-BT) as a Green and Highly Effective Adsorbent for Batch and Fixed-Bed Column Adsorption of Hg (II). J Inorg Organomet Polym 31, 659–673 (2021). https://doi.org/10.1007/s10904-020-01757-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01757-6