Abstract
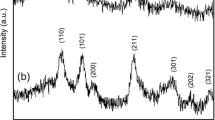
Tin oxide nanocubes are synthesized by hydrothermal approach by using the stannic chloride as precursor salt and sodium hydroxide as precipitating agent in aqueous media. Synthesized product is analyzed by various techniques: X-ray powder diffraction analysis (XRD) and scanning electron microscopy (SEM). Structural composition and parameters of the product are analyzed by (XRD). Morphology of the product is analyzed by (SEM). These synthesized nanocubes are used in two applications: (1) as a photocatalyst and (2) as a fuel additive. Synthesized nanoparticles are used as photocatalyst for the degradation of an organic dye (Congo red) in aqueous medium. Photocatalytic degradation kinetics of dye is monitored at different concentrations of hydrogen peroxide and catalyst in the presence of sunlight. The percentage conversion of dye is also calculated for different concentrations of hydrogen peroxide and catalyst. Efficiency of fuel is analyzed by studying various parameters: flash point, fire point, cloud point, pour point, kinematic viscosity, specific gravity and calorific value at different dosage of SnO2 (10, 20, 30 and 40 ppm). It is found that values of these parameters changes significantly by changing the dosage of catalyst.











Similar content being viewed by others
References
A. Babar, S. Shinde, A. Moholkar, K. Rajpure, J. Alloys Compd. 505, 743–749 (2010)
S. Jamil, M.R.S.A. Janjua, S.R. Khan, N. Jahan, Mater. Res. Exp. 4, 015902–015912 (2017)
S. Kokura, O. Handa, T. Takagi, T. Ishikawa, Y. Naito, T. Yoshikawa, Nanomedicine 6, 570–574 (2010)
M.U. Niemann, S.S. Srinivasan, A.R. Phani, A. Kumar, D.Y. Goswami, E.K. Stefanakos, J. Nanomater. (2008). doi:10.1155/2008/950967
A. Chen, S. Chatterjee, Chem. Soc. Rev, 42, 5425–5438 (2013)
M. Mahmoudi, S. Sant, B. Wang, S. Laurent, T. Sen, Adv. Drug Del. Rev. 63, 24–46 (2011)
P.A. Chen, Adv. Drug Del. Rev. 24, 27–31 (2010)
R. Brayner, S.A. Dahoumane, C. Yéprémian, C. Djediat, M. Meyer, A. Couté, F. Fiévet, Langmuir 26, 6522–6528 (2010)
M.M. Ba-Abbad, A.A.H. Kadhum, A.B. Mohamad, M.S. Takriff, K. Sopian, Int. J. Electrochem. Sci. 7, 4871–4888 (2012)
A.K. Gupta, M. Gupta, Biomaterials 26, 3995–4021 (2005)
M. Gondal, T.A. Saleh, Q. Drmosh, Appl. Surf. Sci. 258, 6982–6986 (2012)
M. Allen, D. Willits, M. Young, T. Douglas, Inorg. Chem 42, 6300–6305 (2003)
H. Wang, A.L. Rogach, Chem. Mater. 26, 123–133 (2013)
A. Shalan, I. Osama, M. Rashad, I. Ibrahim, J. Mater. Sci. 25, 303–310 (2014)
S.S. Lekshmy, K. Joy, J. Sol–Gel Sci. Technol. 67, 29–38 (2013)
S. Han, B. Jang, T. Kim, S.M. Oh, T. Hyeon, Adv. Funct. Mater. 15, 1845–1850 (2005)
C. Nayral, E. Viala, P. Fau, F. Senocq, J.C. Jumas, A. Maisonnat, B. Chaudret, Methods 4082, 4090 (2000)
L. Jiang, G. Sun, Z. Zhou, S. Sun, Q. Wang, S. Yan, H. Li, J. Tian, J. Guo, B. Zhou, J. Phys. Chem. B 109, 8774–8778 (2005)
P.V. Viet, C.M. Thi, L.V. Hieu, J. Nanomater. 2016, 6 (2016)
J. Ba, J. Polleux, M. Antonietti, M. Niederberger, Adv. Mater. 17, 2509–2512 (2005)
J. Zhang, L. Gao, J. Solid State Chem. 177, 1425–1430 (2004)
D. Chen, L. Gao, J. Colloid Interface Sci. 279, 137–142 (2004)
S. Habibzadeh, A. Kazemi-Beydokhti, A.A. Khodadadi, Y. Mortazavi, S. Omanovic, M. Shariat-Niassar, Chem. Eng. J. 156, 471–478 (2010)
E. Najafi, M. Kheirkhahi, M.M. Amini, S.W. Ng, J. Inorg. Organomet. Polym. Mater. 23, 1015–1022 (2013)
M.M. Amini, E. Najafi, P.H. Poor, P. Karami, B. Mohammadi, A. Olyaei, S.W. Ng, J. Inorg. Organomet. Polym. Mater. 25, 1137–1150 (2015)
M.S. Park, G.X. Wang, Y.M. Kang, D. Wexler, S.X. Dou, H.K. Liu, Angew. Chem. 119, 764–767 (2007)
L.-Y. Jiang, X.-L. Wu, Y.-G. Guo, L.-J. Wan, J. Phys. Chem. C 113, 14213–14219 (2009)
M.A. Hossain, G. Yang, M. Parameswaran, J.R. Jennings, Q. Wang, J. Phys. Chem. C 114, 21878–21884 (2010)
L. Nejati-Moghadam, A. Esmaeili Bafghi-Karimabad, M. Salavati-Niasari, H. Safardoust, J. Nanostruct. 5, 47–53 (2015)
S. Karthikeyan, A. Elango, A. Prathima, Indian J. Chem. Technol. 21, 73 (2014)
S. Tazikeh, A. Akbari, A. Talebi, E. Talebi, Mater. Sci. Pol. 32, 98–101 (2014)
M. Choudhary, N.K. Singh, R. Dwivedi, V. Mishra, J. Mater. Sci. 24, 752–757 (2013)
A. Kar, S. Kundu, A. Patra, J. Phys. Chem. C 115, 118–124 (2010)
X.W. Lou, C. Yuan, L.A. Archer, Small 3, 261–265 (2007)
H.C. Chiu, C.S. Yeh, J. Phys. Chem. C 111, 7256–7259 (2007)
G. Sakai, N.S. Baik, N. Miura, N. Yamazoe, Sens. Actuators B 77, 116–121 (2001)
J. Zhang, L. Gao, Mater. Chem. Phys. 87, 10–13 (2004)
V.A. Sakkas, M.A. Islam, C. Stalikas, T.A. Albanis, J. Hazard. Mater. 175, 33–44 (2010)
S. Chatterjee, D.S. Lee, M.W. Lee, S.H. Woo, Bioresour. Technol. 100, 2803–2809 (2009)
M. Ghaedi, M.N. Biyareh, S.N. Kokhdan, S. Shamsaldini, R. Sahraei, A. Daneshfar, S. Shahriyar, Mater. Sci. Eng. C 32, 725–734 (2012)
R. Begum, K. Naseem, Z.H. Farooqi, J. Sol–Gel Sci. Technol. 77, 497–515 (2016)
Z.H. Farooqi, A. Ijaz, R. Begum, K. Naseem, M. Usman, M. Ajmal, U. Saeed, Polym. Compos. (2016). doi:10.1002/pc.23980
Acknowledgements
Authors are highly grateful to the Department of Chemistry, University of Agriculture, Faisalabad Pakistan for assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khalid, M.U., Khan, S.R. & Jamil, S. Morphologically Controlled Synthesis of Cubes like Tin Oxide Nanoparticles and Study of its Application as Photocatalyst for Congo Red Degradation and as Fuel Additive. J Inorg Organomet Polym 28, 168–176 (2018). https://doi.org/10.1007/s10904-017-0687-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-017-0687-5