Abstract
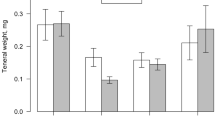
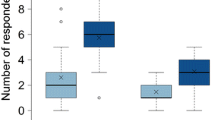
In dominant old-field plant species, genotypic variation in traits important for herbivorous insects may explain variation in insect species abundance. While the importance of plant genetic identity on arthropod abundance has been demonstrated, specific factors that drive genotype choice by insects remain largely unknown. Sixteen genotypes of the widely distributed plant species Solidago altissima were used to investigate the possible role of nutrients and terpene secondary metabolites in shaping the abundance of a common specialist aphid, Uroleucon nigrotuberculatum. Ramets were propagated in a greenhouse and then transferred to a natural field setting. After 76 days, aphid abundance was quantified and leaf tissue assayed for nutrients and terpenes. Aphids/g plant biomass significantly differed among genotypes, with a 30-fold difference observed among plant genotypes. Leaf nitrogen, C:N ratio and water did not vary among genotypes. Of eight terpenes quantified, five were influenced by plant genotype. Aphid abundance increased marginally with the concentration of the monoterpene β-pinene in leaf tissue (P = 0.056). A partial least squares analysis determined that nutritional chemicals did not explain aphid responses, while 49 % of the variation in aphid colonization among genotypes was explained by terpenes. This study is one of the first to demonstrate that variation in allelochemicals may be related to differences in the abundance of a key herbivore among genotypes of a plant species that exhibits large intraspecific genetic variation.





Similar content being viewed by others
References
Barber NA, Marquis RJ (2011) Leaf quality, predators, and stochastic processes in the assembly of a diverse herbivore community. Ecology 92:699–708
Bowers WS, Nishino C, Montgomery ME, Nault LR, Nielson MW (1977) Sesquiterpene progenitor, Germacrene A: an alarm pheromone in aphids. Science 196:680–681
Burkle LA, Souza L, Genung M, Crutsinger CM (2013) Plant genotype, nutrients, and G X E interactions structure floral visitor communities. Ecosphere 4:1–20
Cappuccino N (1988) Spatial patterns of goldenrod aphids and the response of enemies to patch density. Oecologia 76:607–610
Couture JJ, Lindroth RL (2012) Atmospheric change alters the performance of an invasive forest insect. Glob Chang Biol 18:3543–3557. doi:10.1111/gcb.1214
Couture JJ, Meehan TD, Lindroth RL (2012) Atmospheric change alters foliar quality of host trees and performance of two outbreak insect species. Oecologia 168:863–876. doi:10.1007/s00442-011-2139-1
Crutsinger GM, Collins MD, Fordyce JA, Gompert Z, Nice CC, Sanders NJ (2006) Plant genotypic diversity predicts community structure and governs an ecosystem process. Science 313:966–968
Crutsinger GM, Reynolds WN, Classen AT, Sanders NJ (2008) Disparate effects of plant genotypic diversity on foliage and litter arthropod communities. Oecologia 158:65–75. doi:10.1007/s00442-008-1130-y
Dungey HS, Potts BM, Whitham TG, Li HF (2000) Plant genetics affects arthropod community richness and composition: evidence from a synthetic eucalypt hybrid population. Evolution 54:1938–1946
Genung MA, Lessard J-P, Brown CB, Bunn WA, Cregger MA, Reynolds WM, Felker-Quinn E, Stevenson ML, Hartley AS, Crutsinger GM, Schweitzer JA, Bailey JK (2010) Non-additive effects of genotypic diversity increases floral abundance and abundance of floral visitors. PLoS One 5:e8711. doi:10.1371/journal.pone.0008711
Genung MA, Crutsinger GM, Bailey JK, Schweitzer JA, Sanders NJ (2012) Aphid and ladybird abundance depend on the interaction of spatial effects and genotypic diversity. Oecologia 168:167–174. doi:10.1007/s00442-011-2080-3
Gutiérrez C, Fereres A, Reina M, Cabrera R, González-Coloma A (1997) Behavioral and sublethal effects of structurally related lower terpenes on Myzus persciae. J Chem Ecol 23:1641–1650
Haddad NM, Haarstad J, Tilman D (2000) The effects of long-term nitrogen loading on grassland insect communities. Oecologia 124:73–84
Hair JF, Anderson RE, Tatham RL, Black WC (1984) Multivariate analysis: with readings, 3rd edn. MacMillan Publishing Company, New York
Hakes AS, Cronin JT (2011) Resistance and tolerance to herbivory in Solidago altissima (Asteraceae): genetic variability, costs, and selection for multiple traits. Am J Bot 98:1446–1455. doi:10.3732/ajb.1000485
Hersch-Green EI, Turley NE, Johnson MT (2011) Community genetics: what we have accomplished and where should we be going? Phil Trans R Soc B 366:1453–1460. doi:10.1098/rstb.2010.0331
Horner JD, Abrahamson WG (1992) Influence of plant genotype and environment on oviposition preference and offspring survival in a gallmaking herbivore. Oecologia 90:323–332
Hughes AR, Inouye BD, Johnson MT, Underwood N, Vellend M (2008) Ecological consequences of genetic diversity. Ecol Lett 11:609–623. doi:10.1111/j.1461-0248.2008.011179x
Hull-Sanders HM, Clare R, Johnson RH, Meyer GA (2007) Evaluation of the evolution of increased competitive ability (EICA) hypothesis: loss of defense against generalist but not specialist herbivores. J Chem Ecol 33:781–799. doi:10.1007/s10886-007-9252-y
Hull-Sanders HM, Johnson RH, Owen HA, Meyer GA (2009) Effects of polyploidy on secondary chemistry, physiology, and performance of native and invasive genotypes of Solidago gigantea (Asteraceae). Am J Bot 96:762–770. doi:10.3732/ajb.0800200
Johnson MJ (2008) Bottom-up effects of plant genotype on aphids, ants, and predators. Ecology 89:145–154
Johnson MJ, Agrawal AA (2005) Plant genotype and environment interact to shape a diverse arthropod community on evening primrose (Oenothera biennis). Ecology 86:874–885
Johnson MJ, Lajeunesse MJ, Agrawal AA (2006) Additive and interactive effects of plant genotypic diversity on arthropod communities and plant fitness. Ecol Lett 9:24–34. doi:10.1111/j.1461-0248.2005.00833.x
Johnson RH, Hull-Sander HM, Meyer GA (2007) Comparison of foliar terpenes between native and invasive Solidago giganteana. Biochem Syst Ecol 35:821–830. doi:10.1016/j.bse.2007.06.005
Johnson RH, Halitschke R, Kessler A (2010) Simultaneous analysis of tissue- and genotype-specific variation in Solidago altissima (Asteraceae) rhizome terpenoids, and the polyacetylene dehydromatricaria ester. Chemoecology 20:255–264. doi:10.1007/s00049-010-0055-
Kalemba D, Marschall H, Bradesi P (2001) Constituents of the essential oil of Solidago gigantea Ait. (giant goldenrod). Flavor Frag J 16:19–26
Keinänen M, Julkunen-Titto R, Mutikainen P, Walls M, Ovaska J, Vapaavuori E (1999) Trade-offs in phenolic metabolism of silver birth: effects of fertilization, defoliation and genotype. Ecology 80:1970–1986
Langenheim JH (1994) Higher plant terpenoids: a phytocentric overview of their ecological roles. J Chem Ecol 20:1223–1280
Maddox GD, Cappuccino N (1986) Genetic determination of plant susceptibility to an herbivorous insect depends on environmental context. Evolution 40:863–866
Maddox GD, Cook RE, Wimberger PH, Gardescu S (1989) Clone structure in four Solidago altissima (Asteraceae) populations: rhizome connections within genotypes. Am J Bot 76:318–326
Madritch MD, Hunter MD (2005) Phenotypic variation in oak litter influences short- and long-term nutrient cycling through litter chemistry. Soil Biol Biochem 37:319–327. doi:10.1016/j.soilbio.2004.08.002
Maffei ME (2010) Sites of synthesis, biochemistry and functional role of plant volatiles. S Afr J Bot 76:612–631
Müller M, Buchbauer G (2010) Essential oil components as pheromones: a review. Flavor Frag J. doi:10.1002/ffj.2055
Ohgushi T, Ando Y, Utsami S, Craig TP (2011) Indirect interaction webs on tall goldenrod: community consequences for herbivore-induced phenotypes and genetic variation in plants. J Plant Interact 6:147–150. doi:10.1080/17429145.2010.544768
Schädler M, Brandl R, Kempel A (2010) Host-plant genotype determines bottom-up effects in an aphid-parasitoid-predator system. Entomol Exp Appl 135:162–169. doi:10.1111/j.1570-7458.2010.00976.x
Semiz G, Heijari J, Isik K, Holopainen JK (2007) Variation in needle terpenoids among Pinus sylvestris L. (Pinaceae) provenances from Turkey. Biochem Syst Ecol 35:652–661. doi:10.1016/j.bse.2007.05.013
Tobias RD (1995) An introduction to partial least squares regression. SAS Institute, Cary, pp 1–8
Utsumi S, Ando Y, Craig TP, Ohgushi T (2011) Plant genotypic diversity increases population size of a herbivorous insect. Proc R Soc B 278:3108–3115. doi:10.1098/rspb.2011.0239
Whitham TG, Young WP, Martinsen GD, Gehring CA, Schweitzer JA, Shuster SM, Wimp GM, Fischer DG, Bailey JK, Lindroth RL, Woolbright S, Kuske CR (2003) Community and ecosystem genetics: a consequence of the extended phenotype. Ecology 84:559–573
Whitham TG, Gehring CA, Lamit LJ, Wojtowicz T, Evans LM, Keith AR, Smith DS (2012) Community specificity: life and afterlife effects of genes. Trends Plant Sci 17:271–281. doi:10.1016/j.tplants.2012.01.005
Wimp GM, Young WP, Woolbright SA, Martinsen GD, Kein P, Whitham TG (2004) Conserving plant genetic diversity for dependent animal communities. Ecol Lett 7:776–780. doi:10.1111/j.1461-0248.2004.00635.x
Wimp GM, Martinsen GD, Floate KD, Bangert RK, Whitham TG (2005) Plant genetic determinants of arthropod community structure and diversity. Evolution 59:61–69
Wimp GM, Wooley S, Bangert RK, Young WP, Martinsen GD, Kein P, Rehill B, Lindroth RL, Whitham TG (2007) Plant genetics predicts intra-annual variation in phytochemistry and arthropod community structure. Mol Ecol 16:5057–5069. doi:10.1111/j.1365-294X.2007.03544.x
Wold S (1984) The collinearity problem problem in linear regression. The partial least squares (pls) approach to generalized inverses. SIAM J Sci Stat Comput 5:735–743
Wold S, Sjöström M, Erikson L (2001) PLS-regression: a basic tool of chemometrics. Chemometr Intell Lab Syst 58:109–130
Acknowledgments
We thank the field crew at the Gilley Research Station, including Jessica, Eli, and Sophie Howells, Jeff Williams, Bryan Taylor, Peter Blum, and Mathew Swain. Special thanks to Drs. Jennifer Schweitzer and Joseph Bailey at the University of Tennessee-Knoxville for supplying the plants. We also acknowledge the contribution of colleagues and anonymous reviewers, which increased the quality of the manuscript. Support for work with gas chromatography came from Dr. Barkley Sive and David Farrar in the Department of Chemistry at Appalachian State University (ASU). The Cratis Williams Graduate School and Office of Student Research at ASU provided financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Williams, R.S., Avakian, M.A. Colonization of Solidago altissima by the Specialist Aphid Uroleucon nigrotuberculatum: Effects of Genetic Identity and Leaf Chemistry. J Chem Ecol 41, 129–138 (2015). https://doi.org/10.1007/s10886-015-0546-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-015-0546-1