Abstract
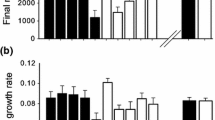
The evolution of increased competitive ability (EICA) hypothesis predicts that invasive plant species may escape their specialized natural enemies in their introduced range and subsequently evolve with a decrease in investment in anti-herbivore chemical defenses relative to native conspecifics. We compared the chemical profile of 10 populations of US native and 20 populations of European invasive Solidago gigantea. To test for differences in inducibility between native and invasive populations, we measured secondary chemistry in both damaged and undamaged plants. We also performed bioassays with three specialist and two generalist insect herbivores from four different feeding guilds. There was no evidence that invasive populations had reduced concentrations of sesquiterpenes, diterpenes, or short-chain hydrocarbons (SCH), although significant variation among populations was detected. Sesquiterpene and diterpene concentrations were not influenced by damage to the host plant, whereas SCH concentrations were decreased by damage for both native and invasive plants. Performance of the three specialist insects was not affected by the continental origin of the host plant. However, larval mass of the generalist caterpillar Spodoptera exigua was 37% lower on native plants compared to invasive plants. The other generalist insect, a xylem-tapping spittlebug that occurs on both continents, performed equally well on native and invasive plants. These results offer partial support for the defense predictions of the EICA hypothesis: the better growth of Spodoptera caterpillars on European plants shows that some defenses have been lost in the introduced range, even though our measures of secondary chemistry did not detect differences between continents. Our results show significant variation in chemical defenses and herbivore performance across populations on both continents and emphasize the need for analysis across a broad spatial scale and the use of multiple herbivores.



Similar content being viewed by others
References
Agrawal, A. A., Kotanen, P. M., Mitchell, C. E., Power, A. G., Godsoe, W., and Klironomos, J. 2005. Enemy release? An experiment with congeneric plant pairs and diverse above- and belowground enemies. Ecology 86:2979–2989.
Anthonsen, T., Henderson, M. S., Martin, A., Murray, R. D. H., McCrindle, R., and McMaster, D. 1973. Constituents of Solidago species. Part IV. Solidagoic acids A and B, Diterpenoids from Solidago gigantea var. serotina. Can. J. Chem. 51:1332–1345.
Baldwin, I. T., Halitschke, R., Paschold, A., von Dahl, C. C., and Preston, C. A. 2006. Volatile signaling in plant–plant interactions: “Talking Trees” in the genomics era. Science 311:812–815.
Barbosa, P. 1991. Plant pathogens and non-vector herbivores, pp. 341–382, in P. Barbosa, V. A. Krischik, and C. G. Jones (eds.). Microbial Mediation of Plant–Herbivore Interactions. John Wiley and Sons, New York.
Biere, A., Marak, H. B., and van Damme, J. M. M. 2004. Plant chemical defense against herbivores and pathogens: generalize defense or trade-offs? Oecologia 140:430–441.
Blossey, B. and Nötzold, R. 1995. Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J. Ecol. 83:887–889.
Bossdorf, O., Schroder, S., Prati, D., and Auge, H. 2004. Palatability and tolerance to simulated herbivory in native and introduced populations of Alliaria petiolata (Brassicaceae). Am. J Bot. 91:856–862.
Cappuccino, N. and Carpenter, D. 2005. Invasive exotic plants suffer less herbivory than non-invasive exotic plants. Biol. Lett. 1:435–438.
Carpenter, D. and Cappuccino, N. 2005. Herbivory, time since introduction and the invasiveness of exotic plants. J. Ecol. 93:315–321.
Cipollini, D., Mbagwu, J., Barto, K., Hillstrom, C., and Enright, S. 2005. Expression of constitutive and inducible chemical defenses in native and invasive populations of Alliaria petiolata. J. Chem. Ecol. 31:1255–1267.
Cooper-Driver, G. A. and LeQuesne, P. W. 1987. Diterpenoids as insect antifeedants and growth inhibitors: role in Solidago species, pp. 334–550, in G. R. Waller (ed.). Allelochemicals: Role in Agriculture and Forestry. American Chemical Society Publication.
D’Antonio, C. M. and Kark, S. 2002. Impacts and extent of biotic invasions in terrestrial ecosystems. Trends Ecol. Evol. 17:202–204.
Elton, C. S. 1958. The Ecology of Invasions by Animals and Plants. Methuen, London, UK.
Genton, B. J., Kotanen, P. M., Cheptou, P.-O., Adolphe, C., and Shykoff, J. A. 2005. Enemy release but no evolutionary loss of defense in a plant invasion: an inter-continental reciprocal transplant experiment. Oecologia 146:404–414.
Greenberg, S. M., Sappington, T. W., Legaspi, B. C., Liu, T.-X., and Sétamou, M. 2001. Feeding and life history of Spodoptera exigua (Lepidoptera : Noctuidae) on different host plants. Ann. Entomol. Soc. Am. 94:566–575.
Hansen, S. O., Hattendorf, J., Wittenberg, R., Reznik, S. Y., Nielsen, C., Raven, H. P., and Nentwig, W. 2006. Phytophagous insects of giant hogweed Heracleum mantagazzianum (Apiaceae) in invaded areas of Europe and in its native area of the Caucasus. Eur. J. Entomol. 103:387–395.
Harborne, J. B. 1993. Introduction to Biochemistry. Academic Press, New York.
Henderson, M. S., McCrindle, R., and McMaster, D. 1973. Constituents of Solidago species. Part V. Non-acidic diterpenoids form Solidago gigantea var. serotina. Can. J. Chem. 51:1346–1358.
Hinz, H. L. and Schwarzlaender, M. 2004. Comparing invasive plants from their native and exotic range: what can we learn for biological control. Weed Technol. 18:1533–1541.
Jakobs, G., Weber, E., and Edwards, P. J. 2004. Introduced plants of the invasive Solidago gigantea (Asteraceae) are larger and grow denser than conspecifics in the native range. Divers. Distrib. 10:11–19.
Jobin, A., Schaffner, U., and Nentwig, W. 1996. The structure of the phytophagous insect fauna on the introduced weed Solidago altissima in Switzerland. Entomol. Exp. Appl. 79:33–42.
Joshi, J. and Vrieling, K. 2005. The enemy release and EICA hypothesis revisited: incorporating the fundamental difference between specialist and generalist herbivores. Ecol. Lett. 8:704–714.
Karban, R. and Baldwin, I. T. 1997. Induced Responses to Herbivory. University of Chicago Press, Chicago.
Keane, R. M. and Crawley, M. J. 2002. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 17:164–170.
Krischik, V. A. 1991. Specific or generalized plant defense: reciprocal interactions between herbivores and pathogens, pp. 309–340, in P. Barbosa, V. A. Krischik, C. G. Jones (eds.). Microbial Mediation of Plant–Herbivore Interactions. John Wiley and Sons, New York.
Krischik, V. A., Goth, R. W., and Barbosa, P. 1991. Generalized plant defense—effects on multiple species. Oecologia 85:562–571.
Leger, E. A. and Forister, M. L. 2005. Increased resistance to generalist herbivores in invasive populations of the California poppy (Eschscholzia californica). Divers. Distrib. 11:311–317.
LeQuesne, P. W., Cooper-Driver, G. A., Villani, M., Do, M. N., Morrow, P. A. and Tonkyn, D. A. 1986. Biologically active diterpenoids of Solidago species–plant–insect interactions, pp. 271–282, in Atta-ur-Rahman and P. W. LeQuensne (eds.). New Trends in Natural Products Chemistry, Vol. 26. Overseas Publishers Association, Amsterdam.
Lewis, W. J., Jones, R. L., Nordlund, D. A., and Sparks, A. N. 1975. Kairomones and their use for management of entomophagous insects: I. Evaluation for increasing rates of parasitization by Trichogramma spp. in the field. J. Chem. Ecol. 1:343–347.
Mack, R. N., Simberloff, D., Lonsdale, W. M., Evans, H., Clout, M., and Bazzaz, F. A. 2000. Biotic Invasions: Causes, epidemiology, global consequences, and control. Ecol. Appl. 10:689–710.
Maron, J. L. and Vilà, M. 2001. When do herbivores affect plant invasions? Evidence for the natural enemies and biotic resistance hypothesis. Oikos 95:361–373.
Maron J. L., Vilà, M., and Arnason, J. 2004a. Loss of enemy resistance among introduced populations of St. John’s Wort (Hypericum perforatum). Ecology 85:2343–3253.
Maron, J. L., Vilà, M., Bommarco, R., Elmendorf, S., and Beardsley, P. 2004b. Rapid evolution of an invasive plant. Ecol. Monogr. 74:261–280.
McBrien, H., Harmsen, R., and Crowder, A. 1983. A case of insect grazing affecting plant succession. Ecology 64:1035–1039.
Meyer, G. A. 1993. A comparison of the impacts of leaf and sap-feeding insects on growth and allocation of goldenrod. Ecology 74:1101–1116.
Meyer, G. A. and Root, R. B. 1993. Effects of herbivorous insects and soil fertility on reproduction of goldenrod. Ecology 74:1117–1128.
Meyer, G., Clare, R., and Weber, E. 2005. An experimental test of the evolution of increased competitive ability hypothesis in goldenrod, Solidago gigantea. Oecologia 144:299–307.
Mitchell, C. E. and Power, A. G. 2003. Release of invasive plants from fungal and viral pathogens. Nature 421:625–627.
Müller-Schärer, H., Schaffner, U., and Steinger, T. 2004. Evolution in invasive plants: implication for biological control. Trends Ecol. Evol. 19:417–422.
Neter, J., Kutner, M. H., Nachtsheim, C. J., and Wasserman, W. 1996. Applied Linear Statistical Models. 4th ed. McGraw-Hill, Boston, Massachusetts.
Padmavathi, C. and Paul, A. V. N. 1998. Saturated hydrocarbons as kairomonal source for the egg parasitoid, Trichogramma chilonis Ishii (Hym., Trichogrammatidae). J. Appl. Entomol. 122:29–32.
Paxton, R. J. and Tengo, J. 2001. Doubly duped males: the sweet and sour of the orchid’s bouquet. Trends Ecol. Evol. 16:167–169.
Pilson, D. 1992. Aphid distribution and the evolution of goldenrod resistance. Evolution 46:1358–1372.
Pimentel, D., Lach, L., Zuniga, R., and Morrison, D. 2000. Environmental and economic costs of nonindigenous species in the United States. BioScience 50:53–65.
Root, R. B. and Cappuccino, N. 1992. Patterns in population change and the organization of the insect community associated with goldenrod. Ecol. Monogr. 62:393–420.
SAS Institute. 2001. SAS. Version 8.2. SAS Institute, Cary, North Carolina, USA.
Siemann, E. and Rogers, W. E. 2003a. Reduced resistance of invasive varieties of the alien tree Sapium sebiferum to a generalist herbivore. Oecologia 135:451–457.
Siemann, E. and Rogers, W. E. 2003b. Increased competitive ability of an invasive tree may be limited by an invasive beetle. Ecol. Appl. 13:1503–1507.
Stastny, M., Schaffner, U., and Elle, E. 2005. Do vigour of introduced populations and escape from specialist herbivores contribute to invasiveness? J. Ecol. 93:27–37.
Tandon, P. L. and Bakthavatsalam, N. 2005. Electro-physiological and olfactometric responses of Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) and Tricogramma chilonis Ishii (Hymenoptera: Trichogrammatidae) to volatiles of trap crops—Tagetes erecta Linnaeus and Solanum viarum Dunal. J. Biol. Control 19:9–16.
Van Kleunen, M., Ramponi, G., and Schmid, B. 2004. Effects of herbivory simulated by clipping and jasmonic acid on Solidago canadensis. Basic Appl. Ecol. 5:173–181.
Vilà, M., Maron, J. L., and Marco, L. 2005. Evidence for the enemy release hypothesis in Hypericum perforatum. Oecologia 142:474–479.
Weaver, C. R. and King, D. R. 1954. Meadow spittlebug. Ohio Agricultural Experiment Station, Wooster, OH. Research Bulletin no. 741.
Weber, E. 1998. The dynamics of plant invasions: a case study of three exotic goldenrod species (Solidago L.) in Europe. J. Biogeogr. 25:147–154.
Weber, E. and Schmid, B. 1998. Latitudinal population differentiation in two species of Solidago (Asteraceae) introduced in Europe. Am. J. Bot. 85:1110–1121.
Willis, A. J., Thomas, M. B., and Lawton, J. H. 1999. Is the increased vigor of invasive weeds explained by a trade-off between growth and herbivore resistance? Oecologia 120:632–640.
Wolfe, L. M. 2002. Why alien invaders succeed: support for the escape-from-enemy hypothesis. Am. Nat. 160:705–711.
Acknowledgements
We thank Terry Bott and Lou Nelson for help in the garden. Special thanks to Jane Clare and Stephen Sanders. Two anonymous reviewers provided comments that improved the manuscript. Financial support was provided by NSF grant DEB-0315430 to G. Meyer and DEB-035127 to R. Johnson.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hull-Sanders, H.M., Clare, R., Johnson, R.H. et al. Evaluation of the Evolution of Increased Competitive Ability (EICA) Hypothesis: Loss of Defense Against Generalist but not Specialist Herbivores. J Chem Ecol 33, 781–799 (2007). https://doi.org/10.1007/s10886-007-9252-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-007-9252-y