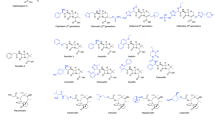
Abstract
Mycotoxins are fungal natural products that are toxic to vertebrate animals including humans. Microbes have been identified that enzymatically convert aflatoxin, zearalenone, ochratoxin, patulin, fumonisin, deoxynivalenol, and T-2 toxin to less toxic products. Mycotoxin-degrading fungi and bacteria have been isolated from agricultural soil, infested plant material, and animal digestive tracts. Biotransformation reactions include acetylation, glucosylation, ring cleavage, hydrolysis, deamination, and decarboxylation. Microbial mycotoxin degrading enzymes can be used as feed additives or to decontaminate agricultural commodities. Some detoxification genes have been expressed in plants to limit the pre-harvest mycotoxin production and to protect crop plants from the phytotoxic effects of mycotoxins. Toxin-deficient mutants may be useful in assessing the role of mycotoxins in the ecology of the microorganisms.







Similar content being viewed by others
References
Abbas HK, Gelderblom WC, Cawood ME, Shier WT (1993) Biological activities of fumonisins, mycotoxins from Fusarium moniliforme, in jimsonweed (Datura stramonium L.) and mammalian cell cultures. Toxicon 31:345–353
Abbas HK, Tanaka T, Duke SO, Porter JK, Wray EM, Hodges L, Sessions AE, Wang E, Merrill AH Jr, Riley RT (1994) Fumonisin- and AAL-toxin-induced disruption of sphingolipid metabolism with accumulation of free sphingoid bases. Plant Physiol 106:1085–1093
Abrunhosa L, Paterson RRM, Venâncio A (2010) Biodegradation of ochratoxin A for food and feed decontamination. Toxins 2:1078–1099
Alberts JF, Engelbrecht Y, Steyn PS, Holzapfel WH, Van Zyl WH (2006) Biological degradation of aflatoxin B1 by Rhodococcus erythropolis cultures. Int J Food Microbiol 109:121–126
Alberts JF, Gelderblom WC, Botha A, Van Zyl WH (2009) Degradation of aflatoxin B1 by fungal laccase enzymes. Int J Food Microbiol 135:47–52
Alexander NJ (2008) The TRI101 story: engineering wheat and barley to resist Fusarium head blight. World Mycotoxin J 1:31–37
Alexander NJ, McCormick SP, Hohn TM (2002) The identification of the Saccharomyces cerevisiae gene AYT1(ORF-YLLO63c) encoding an acetyltransferase. Yeast 19:1425–1430
Alexander NJ, McCormick SP, Ziegenhorn SL (1999) Phytotoxicity of selected trichothecenes using Chlamydomonas reinhardtii as a model system. Nat Toxins 7:265–269
Baldwin NC, Bycroft BW, Dewick PM, Gilbert J (1986) Metabolic conversions of trichothecene mycotoxins: biotransformation of 3-acetyldeoxynivalenol into fusarenon-X. Z. Naturforsch C 41:845–850
Beeton S, Bull AT (1989) Biotransformation and detoxification of T-2 toxin by soil and freshwater bacteria. Appl Environ Microbiol 55:190–197
Benedetti R, Nazzi F, Locci R, Firrao G (2006) Degradation of fumonisin B1 by a bacterial strain isolated from soil. Biodegradation 17:31–38
Bennett JW, Klich M (2003) Mycotoxins. Clin Microbiol Rev 16:497–516
Berthiller F, Crews C, Dall-Asta C, De Saeger S, Haesaert G, Karlovsky P, Oswald IP, Seefelder W, Speijers G, Stroka J (2013) Masked mycotoxins: a review. Mol Nutr Food Res 57:165–186
Blackwell BA, Gilliam JT, Savard ME, Miller JD, Duvick JP (1999) Oxidative deamination of hydrolyzed fumonisin B1 (AP1) by cultures of Exophiala spinifera. Nat Toxins 7:31–38
Bocarov-Stamcic AS, Stankovic SZ, Levic JT, Salma NM, Pantic VR, Barnic SS (2011) In vitro degradation of diacetoxyscirpenol and T-2 toxin by use of Mucor racemosus Fresen. f. racemonsus isolate. Proc Natl Sci 121:51–59
Bu’Lock JD (1980) Mycotoxins as secondary metabolites. In: Steyn PS (ed) The biosynthesis of mycotoxins. Academic, New York, pp 1–16
Castagnero M, Canadas D, Vrabcheva T, Petrova-Bocharova T, Chernozemsky IN, Pfohl-Leszkowwicz A (2006) Balkan endemic nephropathy: role of ochratoxins A through biomarkers. Mol Nutr Food Res 50:519–529
Castoria R, Mannina L, Durán-Patrón R, Maffei F, Sobolev AP, De Felice DV, Pinedo-Rivilla C, Ritieni A (2011) Conversion of the mycotoxin patulin to the less toxic desoxypatulinic acid by the biocontrol yeast Rhodosporidium kratochvilovae strain LS11. J Agric Food Chem 59:11571–11578
Ciegler A (1983) Do mycotoxins function in ecological processes? J Food Saf 5:23–30
Ciegler A, Lillehoj EB, Peterson RE, Hall HH (1966) Microbial detoxification of aflatoxin. Appl Microbiol 14:934–939
Coelho AR, Celli MG, Sataque Ono EY, Hoffman FL, Pagnocca FC, Garcia S, Sabino M, Harada K-I, Wosiacki G, Hirooka EY (2008) Patulin biodegradation using Pichia ohmeri and Saccharomyces cerevisiae. World Mycotoxin J 1:325–331
Coelho AR, Levy RM, Hoffman FL, Taniwaki MH, Kmelmmeier C, Pagnocca FC, Hirooka EY (2006) Potential bio-control of patulin producing Penicillium expansum in post-harvest fruits using antagonistic yeasts. In: Njapau H, Trujillo S, Van Egmond HP, Park DL (eds) Mycotoxins and phycotoxins: advances in determination, toxicology and exposure management. Wageningen Academic Publishers, Wageningen, pp 249–257
Cole RJ, Kirksey JW, Blankenship BR (1972) Conversion of Aflatoxin B1 to isomeric hydroxy compounds by Rhizopus spp. J Agric Food Chem 20:1100–1102
Côté L-M, Dahlem AM, Yoshizawa T, Swanson SP, Buck WB (1986) Excretion of deoxynivalenol and its metabolite in milk, urine, and feces of lactating dairy cows. J Dairy Sci 69:2416–2423
Cotty PJ, Mellon JE (2006) Ecology of aflatoxin producing fungi and biocontrol of aflatoxin contamination. Mycotoxin Res 22:110–117
Desjardins AE, McCormick SP, Appell M (2007) Structure-activity relationships of trichothecene toxins in an Arabidopsis thaliana leaf assay. J Agric Food Chem 55:6487–6492
Desjardins AE, Munkvold GP, Plattner RD, Proctor RH (2002) FUM1-a gene required for fumonisin biosynthesis but not for maize ear rot and ear infection by Gibberella moniliformis in field tests. Mol Plant Microbe Interact 15:1157–1164
Desjardins AE, Proctor RH, Bai G, McCormick SP, Shaner G, Beuchley G, Hohn TM (1996) Reduced virulence of trichothecene-non-producing mutants of Gibberella zeae in wheat field tests. Mol Plant Microbe Interact 9:775–781
Detroy RW, Hesseltine CW (1970) Aflatoxicol: structure of a new transformation product of aflatoxin B1. Can J Biochem 48:830–832
Devi P, Naik CG, Rodreigues C (2006) Biotransformation of citrinin to decarboxycitrinin using an organic solvent-tolerant marine bacterium, Moraxella sp. MB1. Mar Biotechnol 8:129–138
Duvick J (2001) Prospects for reducing fumonisin contamination of maize through genetic modification. Environ Health Perspect 109(Suppl. 2):337–342
Duvick J and Rood T (1998) Zearalenone detoxification compositions and methods. US Patent 5846812, Pioneer Hi-Bred International, 8 Dec 1998
Duvick J, Rood T, Maddox J, and Wang X (1998) Fumonisin detoxification enzymes. US Patent 5716820, Pioneer Hi-Bred International, 10 Feb 1998
Duvick J and Rood TA (2000) Beauvericin detoxification method using bacteria. US Patent 6126934, Pioneer Hi-Bred International, 3 Oct 2000
Duvick JP, Maddox JR, and Gilliam JT (2003) Compositions and methods for fumonisin detoxification. US Patent 6538177, Pioneer Hi-Bred International 25 March 2003
Ehrlich KC, Cotty PJ (2004) An isolate of Aspergillus flavus used to reduce aflatoxin contamination in cottonseed has a defective polyketide synthase gene. Appl Microbiol Biotechnol 65:473–478
El-Nezami HS, Chrevatidis A, Ariola S, Salminen S, Mykkänen H (2002) Removal of common Fusarium toxins in vitro by strains of Lactobacillus and Propionibacterium. Food Addit Contam 19:680–686
El-Sharkawy S, Abul-Hajj YJ (1987) Microbial transformation of zearalenone, I. Formation of zearalenone-4-O-β-glucoside. J Nat Prod 50:520–521
El-Sharkawy S, Abul-Hajj YJ (1988a) Microbial cleavage of zearalenone. Xenobiotica 18:365–371
El-Sharkawy S, Abul-Hajj YJ (1988b) Microbial transformation of zearalenone. 2. Reduction, hydroxylation, and methylation products. J Org Chem 53:515–519
El-Sharkawy S, Selim MI, Afifi MS, Halaweich FT (1991) Microbial transformation of zearalenone to a zearalenone sulfate. Appl Environ Microbiol 57:549–552
Eriksen GS, Pettersson H, Lundh T (2004) Comparative cytotoxicity of deoxynivalenol, nivalenol, their acetylated derivatives and de-epoxy metabolites. Food Chem Toxicol 42:619–624
Falardeau J, Wise C, Novitsky L, and Avis TJ (2013) Ecological and mechanistic insights into the direct and indirect antimicrobial properties of Bacillus subtilis lipopeptides on plant pathogens. J Chem Ecol, this issue
Fliege R, Metzler M (2000) Electrophilic properties of patulin. N-Acetylcysteine and glutathione adducts. Chem Res Toxicol 13:373–381
Fuchs E, Binder, Heidler D, Krska R (2002) Structural characterization of metabolites after the microbial degradation of type A trichothecenes by the bacterial strain BBSH 797. Food Addit Contam 19:379–386
Gaffoor I, Trail F (2006) Characterization of two polyketide synthase genes involved in zearalenone biosynthesis in Gibberella zeae. Appl Environ Microbiol 72:1793–1799
Gelderblom WCA, Jaskiewicz K, Marasas WFO, Thiel PG, Horak RM, Vleggaar R, Kriek NP (1988) Fumonisin—novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl Environ Microbiol 54:1806–1811
Gelderblom WCA, Abel S, Smuts CM, Marnewick J, Marasas WFO, Lemmer ER, Ramljak D (2001) Fumonisin-induced hepatocarcinogenesis: mechanisms related to cancer initiation and promotion. Environ Health Perspect 109:291–300
González-Osnaya L, Soriano JM, Moltó JC, Mañes J (2007) Exposure to patulin from consumption of apple-based products. Food Addit Contam 24:1268–1274
Guan S, He J, Young JC, Zhu H, Li X-Z, Ji C, Zhou T (2009) Transformation of trichothecene mycotoxins by microorganisms from fish digesta. Aquaculture 290:290–295
Guan S, Ji C, Zhou T, Li J, Ma Q, Niu T (2008) Aflatoxin B1 degradation by Stenotrophomonas maltophilia and other microbes selected using coumarin medium. Int J Mol Sci 9:1489–1503
Hartinger D, Schwartz H, Hametner C, Schatzmayr G, Haltrich D, Moll W-D (2011) Enzyme characteristics of aminotransferase FUMI of Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1. Appl Microbiol Biotechnol 91:757–768
Haskard CA, El-Nezami HS, Kankaanpää PE, Salminen S, Ahokas JT (2001) Surface binding of aflatoxin B1 by lactic acid bacteria. Appl Environ Microbiol 67:3086–3091
He C, Fan Y, Liu G, Zhang H (2008) Isolation and identification of a strain of Aspergillus tubingensis with deoxynivalenol biotransformation capability. Int J Mol Sci 9:2366–2375
Heinl S, Hartinger D, Thamhesl M, Schatzmayr G, Moll W-D, Grabherr R (2011) An aminotransferase from bacterium ATCC 55552 deaminates hydrolyzed fumonisin B1. Biodegradation 22:25–30
Heinl S, Hartinger D, Thamhesl M, Vekiruc E, Krska R, Schatzmayr G, Moll W-D, Grabherr R (2010) Degradation of fumonisin B1 by the consecutive action of two bacterial enzymes. J Biotechnol 145:120–129
Hohn TM, Beremand PD (1989) Isolation and nucleotide sequence of a sesquiterpene cyclase gene from the trichothecene-producing fungus Fusarium sporotrichioides. Gene 79:131–138
Hormisch D, Kohring W-W, Giffhorm F, Kroppenstedt RM, Stackebrandt E, Färber P, Holzapfel WH (2004) Mycobacterium fluoranthenivorans sp. nov., a fluoranthene and aflatoxin B1 degrading bacterium from contaminated soil of a former coal gas plant. Syst Appl Microbiol 27:653–660
Igawa T, Takahashi-Ando N, Ochiai N, Ohsato S, Shimizu T, Kudo T, Yamaguchi I, Kimura M (2007) Reduced contamination by the Fusarium mycotoxin zearalenone in maize kernels through genetic modification with a detoxification gene. Appl Environ Microbiol 73:1622–1629
Ikunaga Y, Sato I, Grond S, Numaziri N, Yashida S, Yamaya H, Hiradate S, Hasegawa M, Toshima H, Koitabashi M, Ito M, Karlovsky P, Tsushima S (2011) Nocardioides sp. strain WSN05-2, isolated from a wheat field, degrades deoxynivalenol, producing the novel intermediate 3-epi-deoxynivalenol. Appl Microbiol Biotechnol 89:419–427
Islam R, Zhou T, Young JC, Goodwin PH, Pauls KP (2012) Aerobic and anaerobic de-epoxydation of mycotoxin deoxynivalenol by bacteria originating from agricultural soil. World J Microbiol Biotechnol 28:7–13
Jackson LK, Ciegler A (1978) Production and analysis of citrinin in corn. Appl Environ Microbiol 36:408–411
Janzen DH (1977) Why fruits rot, seeds mold, and meat spoils. Am Nat 111:691–713
Juhasz AL, Stanley GA, Britz ML (2003) Microbial degradation and detoxification of high molecular weight polycyclic aromatic hydrocarbons by Stenophomonas maltophilia strain VUN 10,003. Lett Appl Microbiol 30:396–401
Kakeya H, Takahashi-Ando N, Kimura M, Onose R, Yamaguchi I, Osada H (2002) Biotransformation of the mycotoxin, zearalenone, to a non-estrogenic compound by a fungal strain of Clonostachys sp. Biosci Biotechnol Biochem 66:2723–2726
Karlovsky P, Edmund HC III, Gilliam JT, Maddox JR (2004) Compositions and methods of zearalenone detoxification. US Patent 6812380, Pioneer Hi-Bred International
Karlovsky P (2008) Secondary metabolites in soil ecology. In: Varma A (ed) Secondary metabolites in soil ecology, vol 14, Soil biology series. Springer, Dodrecht, pp 1–19
Kamimura H (1986) Conversion of zearalenone to zearalenone glycoside by Rhizopus sp. Appl Environ Microbiol 52:515–519
Keyser Z, Vismer Z (1999) The antifungal effect of fumonisin B1 on Fusarium and other fungal species. S Afr J Sci 95:455–458
Khatibi PA, Newmister SA, Rayment I, McCormick SP, Alexander NJ, Schmale DG III (2011) Bioprospecting for trichothecene-3-O-acetyltransferases in the fungal genus Fusarium yields functional enzymes that vary in their ability to modify the mycotoxin deoxynivalenol. Appl Environ Microbiol 77:1162–1170
Kimura M, Kaneko I, Komiyama M, Takatsuki A, Koshino H, Yoneyama K, Yamaguchi I (1998a) Trichothecene 3-O-acetyltransferase protects both the producing organism and transformed yeast from related mycotoxins. J Biol Chem 273:1654–1661
Kimura M, Shingu Y, Yoneyama K, Yamaguchi I (1998b) Features of Tri101, the trichothecene 3-O-acetyltransferase gene, related to the self-defense mechanism in Fusarium graminearum. Biosci Biotechnol Biochem 62:1033–1036
King RR, McQueen RE, Levesque D, Greenhalgh R (1984) Transformation of deoxynivalenol (vomitoxin) by rumen microorganisms. J Agric Food Chem 32:1181–1183
Kuiper-Goodman T, Scott PM (1989) Risk assessment of the mycotoxin ochratoxin A. Biomed Environ Sci 2:179–248
Kurtzman CP (2007) Blastobotrys americana sp. nov., Blastobotrys illinoisensis sp. nov., Blastobotrys malaysiensis sp. nov., Blastobotrys muscicola sp. nov., Blastobotrys peoriensis sp. nov. and Blastobotrys raffinosifermentans sp. nov., novel anamorphic yeast species. Int J Syst Evol Microbiol 57:1154–1162
Lapalikar GV, Taylor MC, Warden AC, Scott C, Russell RJ, Oakeshott JG (2012) F420H2-dependent degradation of aflatoxin and other furanocoumarins is widespread throughout the Actinomycetales. PLoS One 7:e30114
Lattanzio VMT, Visconti A, Haidukowski M, Pascale M (2011) Identification and characterization of new Fusarium masked mycotoxins, T2 and HT2 glycosyl derivatives, in naturally contaminated wheat and oats by liquid chromatography-high resolution mass spectrometry. J Mass Spectrom 47:466–475
Lee LS, Dunn JJ, Delucca AJ, Ciegler A (1981) Role of lactone ring of aflatoxin B1 in toxicity and mutagenicity. Experientia 37:16–17
Levy RM, Hayashi L, Carreiro SC, Pagnocca FC, Hirooka EY (2002) Inhibition of mycotoxigenic Penicillium sp. and patulin biodegradation by yeast strains. Rev Bras Armazenamento 27:41–47
Li X-Z, Zhu C, Lange CFM, Zhou T, He J, Yu H, Gong J, Young JC (2011) Efficacy of detoxification of deoxynivalenol-contaminated corn by Bacillus sp. LS100 in reducing the adverse effects of the mycotoxin on swine growth performance. Food Addit Contam 28:894–901
Liu D-L, Yao D-S, Liang R, Ma L, Cheng W-Q, Gu L-Q (1998) Detoxification of aflatoxin B1 by enzymes isolated from Armillariella tabescens. Food Chem Toxicol 36:563–574
Mann R and Rhem HJ (1976) Degradation products from aflatoxin B1 by Corynebacterium rubrum, Aspergillus niger, Trichoderma viride and Mucor ambiguus. Eur J Appl Microbiol: 2:297–306
McCormick SP, Alexander NJ, Trapp SE, Hohn TM (1999) Disruption of TRI101, the gene encoding trichothecene 3-O-acetyltransferase, from Fusarium sporotrichioides. Appl Environ Microbiol 65:5252–5256
McCormick SP, Price NPJ, Kurtzman CP (2012) Glucosylation and other biotransformations of T-2 toxin by yeasts of the Trichomonascus clade. Appl Environ Microbiol 78:8694–8702
McCormick SP, Stanley AM, Stover NA, Alexander NJ (2011) Trichothecenes: from simple to complex mycotoxins. Toxins 3:802–814
Megharaj M, Garthwaite I, Thiele JH (1997) Total biodegradation of the oestrogenic mycotoxin zearalenone by a bacterial culture. Lett Appl Microbiol 24:329–333
Merrill AH Jr, Van Echten G, Wang E, Sandhoff K (1993) Fumonisin B1 inhibits sphingosine (sphinganine) N-aceyltransferase and de novo sphingolipid biosynthesis in cultured neurons in situ. J Biol Chem 268:27299–27306
Middelhoven WJ, Kurtzman CP (2003) Relation between phylogeny and physiology in some ascomycetous yeasts. Antonie Van Leeuwenhoek 83:69–74
Moake MM, Padilla-Zakour OI, Worobo RW (2005) Comprehensive review of patulin control methods in food. Compr Rev Food Sci Food Saf 4:8–21
Molnar O, Schatzmayr G, Fuchs E, Prillinger H (2004) Trichosporon mycotoxinivorans sp. nov., a new yeast species useful in biological detoxification of various mycotoxins. Syst Appl Microbiol 27:661–671
Moss MO, Long MT (2002) Fate of patulin in the presence of the yeast Saccharomyces cerevisiae. Food Addit Contam 19:387–399
Motomura M, Toyomasu T, Mizuno K, Shinozawa T (2003) Purification and characterization of an aflatoxin degradation enzyme from Pleurotus ostreatus. Microbiol Res 158:237–242
Myburg RB, Dutton MF, Chuturgoon AA (2002) Cytotoxicity of fumonisin B1, diethylnitrosamine, and catechol on the SNO esophageal cancer cell line. Environ Health Perspect 110:813–815
Niu G, Siegel J, Schuler M, Berenbaum MR (2009) Comparative toxicity of mycotoxins to navel orangeworm (Amyelois transitella) and corn earworm (Helicoverpa zea). J Chem Ecol 35:951–957
O’Callaghan J, Caddick MX, Dobson AD (2003) A polyketide synthase gene required for ochratoxin A biosynthesis in Aspergillus ochraceus. Microbiology 149:3485–3491
Ohsato S, Ochiai-Fukuda T, Nishiuchi T, Takahashi-Ando N, Koizumi S, Hamamoto H, Kudo T, Yamaguchi I, Kimura M (2007) Transgenic rice plants expressing trichothecene 3-O-acetyltransferase show resistance to the Fusarium phytotoxin deoxynivalenol. Plant Cell Rep 26:531–538
Palencia E, Torres O, Hagler W, Meredith FI, Williams LD, Riley RT (2003) Total fumonisins are reduced in tortillas using the traditional nixtamalization method of Mayan communities. J Nutr 133:3200–3203
Proctor RH, Brown DW, Plattner RD, Desjardins AE (2003) Co-expression of 15 contiguous genes delineates a fumonisin biosynthetic gene cluster in Gibberella moniliformis. Fungal Genet Biol 38:237–249
Puel O, Galtier P, Oswald IP (2010) Biosynthesis and toxicological effects of patulin. Toxins 2:613–631
Rasmussen TB, Skindersoe ME, Bjarnsholt T, Phipps RK, Christensen KB, Jensen PO, Anmdersen JB, Koch B, Larsen TO, Hentzer M, Eberl L, Hoiby N, Givskov M (2005) Identity and effects of quorum-sensing inhibitors produced by Penicillium species. Microbiology 151:1325–1340
Ricelli A, Baruzzi F, Solfrizzo M, Morea M, Fanizzi FP (2007) Biotransformation of patulin by Gluconobacter oxydans. Appl Environ Microbiol 73:785–793
Riley RT, Goeger DE (1992) Cylopiazonic acid: speculation on its function in fungi. In: Bhatnagar D, Lillehoj EB, Arora DK (eds) Handbook of applied mycology, vol 5, Mycotoxins in Ecological Systems. Marcel Dekker, New York, pp 385–402
Rohlfs M, Albert M, Keller NP, Kempken (2010) Secondary chemicals protect mould from fungivory. Biol Lett 3:523–525
Ross PF, Rice LG, Osweiler GD, Nelson PE, Richard JL, Wilson TM (1992) A review and update of animal toxicoses associated with fumonisin-contaminated feeds and production of fumonisins by Fusarium isolates. Mycopathologia 117:109–114
Sanzani SM, Reverberi M, Punelli M, Ippolito A, Fanelli C (2012) Study on the role of patulin on pathogenicity and virulence of Penicillium expansum. Int J Food Microbiol 15:1157–1164
Sato I, Ito M, Ishizaka M, Ikunaga Y, Sato Y, Yoshida S, Koitabashi M, Tsushima S (2012) Thirteen novel deoxynivalenol-degrading bacteria are classified within two genera with distinct degradation mechanisms. FEMS Microbiol Lett 327:110–117
Schatzmayr G, Heidler D, Fuchs E, Nitsch S, Mohnl M, Täubel M, Loibner AP, Braun R, Binder E-M (2003) Investigation of different yeast strains for the detoxification of ochratoxin A. Mycotoxin Res 19:124–128
Schmidt-Heydt M, Graf E, Stoll D, Geisen R (2012) The biosynthesis of ochratoxin A by Penicillium as one mechanism for adaptation to NaCl rich foods. Food Microbiol 29:233–241
Schneweis I, Meyer K, Engelhardt G, Bauer J (2002) Occurrence of zearalenone-4-β-D-glucopyranoside in wheat. J Agric Food Chem 50:1736–1738
Scott PM, Kennedy B, Van Walbeek W (1972) Desoxypatulinic acid from a patulin-producing strain of Penicillium patulum. Experientia 28:1252
Sekiguchi J, Shimamoto T, Yamada Y, Gaucher GM (1983) Patulin biosynthesis: enzymatic and nonenzymatic transformations of the mycotoxin (E)-ascladiol. Appl Environ Microbiol 45:1939–1942
Shima JS, Takahashi Y, Iawi Y, Fujimoto H, Yamazaki M, Ochi K (1997) Novel detoxification of the trichothecene mycotoxin deoxynivalenol by a soil bacterium isolated by enrichment culture. Appl Environ Microbiol 63:3825–3830
Shimizu T, Kinoshita H, Ishihara S, Sakai K, Nagai S, Nihira T (2005) Polyketide synthase gene responsible for citrinin biosynthesis in Monascus purpureus. Appl Environ Microbiol 71:3453–3457
Smiley RD, Draughon FA (2000) Preliminary evidence that degradation of aflatoxin B1 by Flavobacterium aurantiacum is enzymatic. J Food Prot 63:415–418
Stinson EE, Osman SF, Huhtanen CN, Bills DD (1978) Disappearance of patulin during alcoholic fermentation of apple juice. Appl Environ Microbiol 36:620–622
Størmer FC, Høiby EA (1996) Citrinin, ochratoxin A and iron. Possible implications for their biological function and induction of nephropathy. Mycopathologia 134:103–107
Suzuki T, Takeda M, Tanabe H (1971) A new mycotoxin produced by Aspergillus clavatus. Chem Pharm Bull 19:1786–1788
Swanson SP, Nicoletti J, Rood HD Jr, Buck WB, Coté LM, Yashizawa T (1987a) Metabolism of three trichothecene mycotoxins, T-2 toxin, diacetoxyscirpenol and deoxynivalenol, by bovine rumen microorganisms. J Chromatogr Biomed Appl 414:335–342
Swanson SP, Rood HD Jr, Berhrens JC, Sanders PE (1987b) Preparation and characterization of the deepoxy trichothecenes: deepoxy HT-2, deepoxy T-2 triol, deepoxy T-2 tetraol, deepoxy 15-monoacetoxyscirpenol, and deepoxyscirpentriol. Appl Environ Microbiol 53:2821–2826
Takahashi-Ando N, Kimura M, Kakeya H, Osada H, Yamaguchi I (2002) A novel lactonohydrolase responsible for the detoxification of zearalenone: enzyme purification and gene cloning. Biochem J 365:1–6
Takahashi-Ando N, Ohsato S, Shibata T, Hamamoto H, Yamaguchi I, Kimura M (2004) Metabolism of zearalenone by genetically modified organisms expressing the detoxification gene from Clonostachys rosea. Appl Environ Microbiol 70:3239–3245
Taylor MC, Jackson CJ, Tattersall DB, French N, Peat TS, Newman J, Briggs LJ, Lapalikar GV, Campbell PM, Scott C, Russell RV, Oakeshott JG (2010) Identification and characterization of two families of F420H2-dependent reductases from Mycobacteria that catalyse aflatoxin degradation. Mol Microbiol 78:561–575
Teniola OD, Addo PA, Brost IM, Färber P, Jany K-D, Alberts JF, Van Zyl WH, Steyn PS, Holzapfel WH (2005) Degradation of aflatoxin B1 by cell-free extracts of Rhodococcus erythropolis and Mycobacterium fluoranthenivorans sp. nov. DSM44556. Int J Food Microbiol 105:111–117
Tokai T, Fujimura M, Inoue H, Aoki T, Ohta K, Shibata T, Yamaguchi I, Kimura M (2005) Concordant evolution of trichothecene 3-O-acetyltransferase and an rDNA species phylogeny of trichothecene-producing and non-producing fusarium and other ascomycetous fungi. Microbiology 151:509–519
Ueno Y, Nakayama K, Ishii K, Tashiro F, Minoda Y, Omori T, Komagata K (1983) Metabolism of T-2 toxin in Curtobacterium sp. strain 114–2. Appl Environ Microbiol 46:120–127
Utermark J, Karlovsky P (2007) Role of zearalenone lactonase in protection of Gliocladium roseum from fungitoxic effects of the mycotoxin zearalenone. Appl Environ Microbiol 73:637–642
Vekiru E, Hametner C, Mittbauer R, Rechhaler J, Adam G, Schatzmayr G, Krska R, Schuhmacher R (2010) Cleavage of zearalenone by Trichosporon mycotoxinivorans to a novel nonestrogenic metabolite. Appl Environ Microbiol 76:2353–2359
Wallen L, Lyons AJ, Pridham TG (1980) Antimicrobial activity of patulin derivatives: a preliminary report. J Antibiot 33:767–769
Wegst W, Lingens F (1983) Bacterial degradation of ochratoxin A. FEMS Microbiol Lett 17:341–344
Wicklow DT, Dowd PF, Alfatafta AA, Gloer JB (1996) Ochratoxin A: an antiinsectan metabolite from the sclerotia of Aspergillus carbonarius NRRL 369. Can J Microbiol 42:1100–1103
Williams JH, Phillips TD, Jolly PE, Stiles JK, Jolly CM, Aggarwal D (2004) Human aflatoxicosis in developing countries: a review of toxicology, exposure, potential health consequences, and interventions. Am J Clin Nutr 80:1106–1122
White S, O’Callaghan J, Dobson AD (2006) Cloning and molecular characterization of Penicillium expansum genes up-regulated under conditions permissive for patulin biosynthesis. FEMS Microbiol Lett 255:17–26
Young JC, Zhou T, Yu H, Zhu H, Gong J (2007) Degradation of trichothecene mycotoxins by chicken intestinal microbes. Food Chem Toxicol 45:136–143
Yu H, Zhou T, Gong J, Young C, Su Z, Li X-Z, Zhu H, Tsao R, Yang R (2010) Isolation of deoxynivalenol-transforming bacteria from the chicken intestines using the approach of PCR-DGGE guided microbial selection. BMC Microbiol 10:182
Yu J, Chang P-K, Erlich KC, Cary JW, Bhatnagar D, Cleveland TE, Payne GA, Linz JE, Woloshuk CP, Bennett JW (2004) Clustered pathway genes in aflatoxin biosynthesis. Appl Environ Microbiol 70:1253–1262
Zeng R, Niu G, Wen Z, Schuler M, Berenbaum MR (2006) Toxicity of aflatoxin B1 to Helicoverpa zea and bioactivation by cytochrome P450 monooxygenases. J Chem Ecol 32:1459–1471
Zhou T, He J, Gong J (2008) Microbial transformation of trichothecene mycotoxins. World Mycotoxin J 1:23–30
Zinedine A, Soriano JM, Molto JC, Man J (2007) Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem Toxicol 45:1–18
Zou Z-Y, He Z-F, Li H-J, Han P-F, Meng Z, Zhang Y, Zhou F, Ouyang K-P, Chen X-Y, Tang J (2007) In vitro removal of deoxynivalenol and T-2 toxin by lactic acid bacteria. Food Sci Biotechnol 21:1677–1683
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McCormick, S.P. Microbial Detoxification of Mycotoxins. J Chem Ecol 39, 907–918 (2013). https://doi.org/10.1007/s10886-013-0321-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-013-0321-0