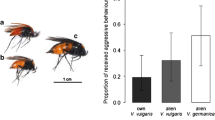
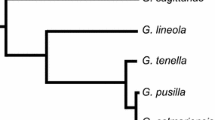
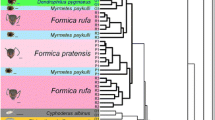
Abstract
Semiochemicals influence many aspects of insect behavior, including interactions between parasites and their hosts. We studied the chemical recognition system of bumblebees (Bombus) by examining the cuticular hydrocarbon cues of 14 species, including five species of social parasites, known as cuckoo bees (subgenus Psithyrus). We found that bumblebees possess species-specific alkene positional isomer profiles that are stable over large geographical regions and are mimicked by three host-specific cuckoo parasites. In three host-cuckoo associations where mimicry is poor, possibly due to recent host shifts, these cuckoos produce dodecyl acetate a known chemical repellent that allows the cuckoos to invade their host colonies. Our findings indicate cuckoos use two chemical mechanisms, mimicry and repellents, to invade their hosts, and this may reflect different stages of an ongoing dynamic arms race.






Similar content being viewed by others
References
Alford, D. V. 1975. Bumblebees. London: Davis-Poynter. 352 pp.
Ayasse, M., Marlovits, T., Tengö, J., Taghizadeh, T., and Francke, W. 1995. Are there pheromonal dominance signals in the bumblebee Bombus hypnorum L (Hymenoptera, Apidae)? Apidologie 26:163–180.
Ayasse, M., Birnbaum, J., Tengö, Van Doorn, A., Taghizadeh, T., and Francke, W. 1999. Caste- and colony-specific chemical signals on eggs of the bumble bee, Bombus terrestris. Chemoecology 9:119–126.
Blažytė-Čereškienė, L., and Būda, V. 2007. Ability of honey bees to detect and recognise isomers of cresol. Ekologija 53:16–21.
Byers, J. A. 2006. Pheromone component patterns of moth evolution revealed by computer analysis of the Pherolist. J. Anim. Ecol. 75:399–407.
Carlson, D. A., Roan, C-S., Yost, R. A., and Hector, J. 1989. Dimethyl disulfide derivatives of long chain alkenes, alkadienes, and alkatrienes for gas chromatography/mass spectrometry. Anal. Chem. 61:1564–1571.
Carvell, C., Rothery, P., Pywell, R. F., and Heard, M. S. 2008. Effects of resource availability and social parasite invasion on field colonies of Bombus terrestris. Eco. Entomol. 33:321–327.
Châline, N., Sandoz, J. C., Martin, S. J., Ratnieks, F. L. W., and Jones, G. R. 2005. Learning and discrimination of individual cuticular hydrocarbons by honey bees (Apis mellifera). Chem. Senses 30:327–333.
D’Ettorre, P., Errard, C., Ibarra, F., Francke, W., and Hefetz, A. 2000. Sneak in or repel your enemy: dufour’s gland repellent as a strategy for successful usurpation in the slave-maker Polyergus rufescens. Chemoecology 10:35–142.
Dronnet, S., Simon, X., Verhaeghe, J. C., Rasmont, P., and Errard, C. 2005. Bumblebee inquilinism in Bombus (Fernaldaepsithyrus) sylvestris (Hymenoptera, Apidae): behavioural and chemical analyses of host-parasite interactions. Apidologie 36:59–70.
Edwards, E. 1980. Social Wasps: Their Biology and Control. Rentokil Libr., East Grinstead. 398 pp.
Fisher, R. M. 1983. Recognition of host nest odour by the bumble bee social parasite Psithyrus ashtoni (Hymenoptera: Apidae), J. N. Y. Entomol. Soc. 91:503–507.
Fisher, R. M. 1987. Queen-worker conflict and social parasitism in bumblebees. Anim. Behav. 35:1026–1036.
Fisher, R. M. 1988. Observations on the behaviours of three European cuckoo bumble species. Insect. Soc. 35:341–354.
Fisher, R. M., and Sampson, B. J. 1992. Morphological specializations of the bumble bee social parasite Psithyrus ashtoni. Can. Entomol. 124:69–77.
Fisher, R. M., Greenwood, D. R., and Shaw, G. J. 1993. Host recognition and the study of a chemical basis for attraction by cuckoo bumble bees (Hymenoptera: Apidae). J. Chem. Ecol. 19:771–786.
Free, J. B., and Butler, C. G. 1959. Bumblebees. London. 208 pp.
Frehn, E., and Schwammberger, K. H. 2001. Social parasitism of Psithyrus vestalis in Free-foraging Colonies of Bombus terrestris (Hymenoptera: Apidae). Entomol. Gen. 25:103–105.
Graham, R. A., Brand, J. M., and Markovetz, A. J. 1979. Decyl acetate synthesis in the ant Formica schaufussi (Hymenoptera: Formicidae). Insect Biochem. 9:331–333.
Greene, M. J., and Gordon, D. M. 2007. Structural complexity of chemical recognition cues affects the perception of group membership in the ants Linephithema humile and Aphaenogaster cockerelli. J. Exp. Biol. 210:897–905.
Hoffer, E. 1888. Die schmarotzerhummeln Steiermarks. Lebensgeschichte und beschreibung derselben.Mitt. naturw. Ver. Steirermark, 25:82–158.
Küpper, G., and Schwammberger, K. H. 1995. Social parasitism in bumble bees (Hymenoptera, Apidae): observations of Psithyrus sylvestris in Bombus pratorum nests. Apidologie 26:245–254.
Lanne, B. S., Bergström, G., Wassgren, A-B., and Törnbäck, B. 1987. Biogenetic patterns of straight chain marking compounds in male bumblebee. Comp. Biochem. Physiol. 88B: 631–636.
Lenoir, A., D’Ettorre, P., and Errard, C., 2001. Chemical ecology and social parasitism in ants. Annu. Rev. Entomol. 46:573–599.
Martin, S. J., and Drijfhout, F. P. 2009a. How reliable is the analysis of complex cuticular hydrocarbon profiles by multi-variate statistical methods? J. Chem. Ecol. 35:375–382.
Martin, S. J., and Drijfhout, F. P. 2009b. Nestmate and task cues are influenced and encoded differently within ant cuticular hydrocarbon profiles. J. Chem. Ecol. 35:368–374.
Martin, S. J., Vitikainen, E., Helanterä, H., and Drijfhout, F. P. 2008a. Chemical basis of nestmate recognition in the ant Formica exsecta. Proc. R. Soc. B. 275:1271–1278.
Martin, S. J., Helanterä, H., and Drijfhout, F. P. 2008b. Colony-specific hydrocarbons identify nest mates in two species of Formica ant. J. Chem Ecol. 34:1072–1080.
Martin, S. J., Helanterä, H., and Drijfhout, F. P. 2008c. Evolution of Species-Specific Cuticular Hydrocarbon Patterns in Formica Ants. Bio. J. Linn. Soc. 95:131–140.
Morgan, E. D. 2004. Biosynthesis in Insects. Royal Society of Chemistry, Cambridge.
Oldham, N. J., Billen, J., and Morgan, E. D. 1994. On the similarity of the Dufour gland secretion and the cuticular hydrocarbons of some bumblebees. Physiol. Entomol. 19:115–123.
Ozaki, M., Wada-Katsumata, A., Fujikawa, K., Iwasaki, M., Yokohari, F., Satoji, Y., Nisimura, T., and Yamaoka. R. 2005. Ant nest mate and non-nest mate discrimination by a chemosensory sensillium. Science 309:311–315.
Pamilo, P., Varvio-aho, S-L., and Pekkarinen, A. 1984. Genetic variation in bumblebees (Bombus, Psithyrus) and putative sibling species of Bombus lucorum. Hereditas 101:245–251.
Regnier, T. E., and Wilson, E. O. 1971. Chemical communication and ‘propaganda’ in slave-making ants. Science 172:267–269.
Reinig, W. F. 1935. On the variation of Bombus lapidarius L. and its cuckoo, Psithyrus rupestris Fabr., with notes on mimetic similarity. J. Genetics 30:321–356.
Richards, O. W. 1927. The specific characters of the British humblebees (Hymenoptera). Trans. Entomol. Soc. Lond. 75:233–268.
Rouault, J., Capy, P., and Jallon, J-M. 2001. Variations of male cuticular hydrocarbons with geoclimatic variables: an adaptative mechanism in Drosophila melanogaster? Genetica 110:117–130.
Sladen, F. W. L. 1912. The Humble-bee, Its Life History and How to Domesticate it, with Descriptions of All the British Species of Bombus and Psithyrus. London: MacMillan. 283 p. [Republished in 1989, Woonton (Herefordshire): Logaston Press. 273 p.]
Sramkova, A., and Ayasse, M. 2008. Psithyrus females do possess wax glands. Insect. Soc. 55:404–406.
Sramkova, A., and Ayasse, M. 2009. Chemical ecology involved in invasion success of the cuckoo bumblebee Psithyrus vestalis and in survival of workers of its host Bombus terrestris. Chemoecology 19:55–62.
Symonds, M. R. E., and Elgar, M. A. 2004. The mode of pheromone evolution: evidence from bark beetles. Proc. R. Soc. B. 271:839–846.
Tengö, J., Hefetz, A., Bertsch, A., Schmitt, U., Lübke, G., and Francke, W. 1991. Species specificity and complexity of Dufour’s gland secretion of bumblebees. Comp. Biochem. Physiol. 99B: 641–646.
Tsuneoka, Y., and Akino, T. 2009. Repellent effect on host Formica workers of queen Dufour’s gland secretion of the obligatory social parasite ant, Polyergus samurai (Hymenoptera: Formicidae). Appl. Entomol. Zool. 44:133–141.
Urbanová, K., Halík, J., Hovorka, O., Kindl, J., and Valterová, I. 2004. Marking pheromones of the cuckoo bumblebee males (Hym, Apoidea, Bombus): compositions of labial gland secretions of six species found in the Czech Republic. Biochem. Sys. Ecol. 32:1025–1045.
Van Honk, C. G. J., Roseler, P. F., Velthuis, H., and Malotaux, M. 1981.The onquest of a Bombus terrestris colony by a Psithyrus vestalis female. Apidologie 12:57–67.
Wagner, D., Tissot, M., and Gordon, D. M. 2001. Task-related environment alters the cuticular hydrocarbon composition of Harvester ants. J. Chem. Ecol. 27:1805–1819.
Williams, P. H. 1998. An annotated checklist of bumble bees with an analysis of patterns of description (Hymenoptera: Apidae, Bombini). Bull. Nat. His. Mus. (Entomol.) 67:79–152.
Williams, P. H. 2007. Bumble bees - and their decline in Britain, pp. 161–176, in NL Carreck and TW Johnson (eds.). Aspects of sociality in insects. Central Association of Bee-Keepers: Ilford.
Williams, P. H. 2008. Do the parasitic Psithyrus resemble their host bumblebees in colour pattern? Apidologie 39:637–649.
Williams, P. H., Cameron S. A., Hines, H. M., Cederberg, B., and Rasmont, P. 2008. A simplified subgeneric classification of the bumblebees (genus Bombus). Apidologie 39:46–74.
Wilson, E. O. 1971. The Insect Societies. Harvard Univ. Press, Cambridge, Mass, 548 pp.
Zimma, B. O., Ayasse, M., Tengö, J., Ibarra, F., Schulz, C., and Francke, W. 2003. Do social parasitic bumblebees use chemical weapons? (Hymenoptera, Apidae). J. Comp. Physiol. A 189:769–775.
Acknowledgments
Thanks go to Matthew Heard of CEH Wallingford, Dave Goulson of Stirling University, and Maggie Frankum of Leicester for providing additional Psithyrus queens. Also, thanks to Roger Butlin, Paul Brakefield, and Duncan Jackson of Sheffield University for detailed comments, in addition to the helpful comments by two anonymous referees. Funding was provided by NERC (NE/C512310/1 and NE/F018355/1).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental figure 1
The proportion of n-alkanes extracted from the wing and Dufour’s gland samples for each species of bumblebee. The top two graphs are for the non-parasitic Bombus species, while the bottom two inverted graphs shows the data for the corresponding Psithyrus-cuckoo species. The arrows indicate alternative hosts. The error bars represent one SD. (PPT 81 kb)
Rights and permissions
About this article
Cite this article
Martin, S.J., Carruthers, J.M., Williams, P.H. et al. Host Specific Social Parasites (Psithyrus) Indicate Chemical Recognition System in Bumblebees. J Chem Ecol 36, 855–863 (2010). https://doi.org/10.1007/s10886-010-9805-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-010-9805-3