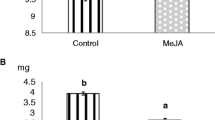
Abstract
The up-regulation of plant defense-related toxins or metabolic enzyme binding proteins often leads to a negative effect on herbivorous insects. These negative effects can manifest themselves at three points: changes in food ingestion, post-ingestive-changes, and post-digestive changes. Many studies have related the decrease in herbivore growth and/or survival with expression of chemicals that interfere with post-digestive effects such as the anti-nutritive effects of protease inhibitors. Nevertheless, it is unclear whether such compounds impact herbivores via earlier ingestive processes. We addressed this question by using a jasmonate-deficient mutant (Def-1), a jasmonate-overexpressor mutant (Prosystemin or Prosys), and wild-type tomato in three trials with 5th instar Trichoplusia ni. Decreases in relative growth rate (RGR) confirmed that T. ni fed on the Prosys plants developed poorly compared to those feeding on Def-1 plants (larvae on wild-types were intermediate). Preingestive and postingestive processes contributed to this effect. Total food ingested and the consumptive index were 25% lower on Prosys plants compared to Def-1 plants. Post-ingestive processes, measured by approximate digestibility, were 62% greater on Prosy plants. Post-digestive efficiency measured by conversion of ingested and digested food (ECI and ECD) decreased on Prosys plants two-fold compared to Def-1 plants. This post-digestive interference correlated well with the 2-fold decrease in activity of digestive enzymes, serine proteases, in Prosys-fed T. ni compared to those on Def-1 plants. No difference in detoxifying enzyme activity was detected.



Similar content being viewed by others
References
Abdeen, A., Virgós, A., Olivella, E., Villanueva, J., Avilés, X., Gabarra, R., and Prat, S. 2005. Multiple insect resistance in transgenic tomato plants over-expressing two families of plant proteinase inhibitors. Plant Mol. Biol. 57:189–202.
Baker, J. E., Fabrick, J. A., and Zhu, K. Y. 1998. Characterization of esterases in malathion-resistant and susceptible strains of the pteromalid parasitoid Anisopteromalus calandre. Insect Biochem. Mol. Biol. 28:1039–1050.
Barbehenn, R. V., Jones, C. P., Yip, L., Tran, L., and Constabel, C. P. 2007. Limited impact of elevated levels of polyphenol oxidase on tree-feeding caterpillars: assessing individual plant defenses with transgenic poplar. Oecologia 154:129–140.
Bi, J. L., Felton, G. W., Murphy, J. B., Howles, P. A., Dixon, R. A., and Lamb, C. J. 1997. Do plant phenolics confer resistance to specialist and generalist insect herbivores? J. Agric. Food Chem. 45:4500–4504.
Boughton, A. J., Hoover, K., and Felton, G. W. 2005. Methyl jasmonate application induces increased densities of glandular trichomes on tomato. J. Chem. Ecol. 31:2211–2216.
Bradford, M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochem. 72:248–254.
Broadway, R. M. 1995. Are insects resistant to plant proteinase inhibitors? J. Insect Physiol. 2:107–116.
Broadway, R. M., Duffey, S. S., Pearce, G., and Ryan, C. A. 1986. Plant proteinase inhibitors: a defense against herbivorous insects? Entomol. Exp. Appl. 41:33–38.
Broadway, R. M., and Colvin, A. A. 1992. Influence of cabbage proteinase inhibitors in situ on the growth of larval Trichoplusia ni and Pieris rapae. J. Chem. Ecol. 18:1009–1024.
Chen, H., Wilkerson, C. G., Kuchar, J. A., Phinney, B. S., and Howe, G. A. 2005. Jasmonate-inducible plant enzymes degrade essential amino-acids in the herbivore midgut. Proc. Natl. Acad. Sci. USA 102:19237–19242.
Chougule, N. P., Doyle, E., Fitches, E., and Gatehouse, J. A. 2008. Biochemical characterization of midgut digestive proteases from Mamestra brassicae (cabbage moth; Lepidoptera: Noctuidae) and effect of soybean Kunitz inhibitor (SKTI) in feeding assays. J. Insect Physiol. 54:563–572.
Duffey, S. S., and Stout, M. J. 1996. Anti-nutritive and toxic components of plant defense against insects. Arch. Insect Biochem. Physiol. 32:3–37.
Elliger, C. A., Wong, Y., Chan, B. G., and Waiss, A. C. 1981. Growth inhibitors in tomato (Lycopersicon) to tomato fruitworm (Heliothis zea). J. Chem. Ecol. 7:753–758.
Farmer, E. E., and Ryan, C. A. 1992. Octadecanoid precursors of jasmonic acid activate the synthesis of wound-inducible proteinase inhibitors. Plant Cell 4:129–134.
Francis, F., Vanhaelen, N., and Haubruge, E. 2005. Glutathione S-transferases in the adaptation to plant secondary metabolites in the Myzus persicae aphid. Arch. Insect Biochem. Physiol. 58:166–174.
Green, T. R., and Ryan, C. A. 1972. Wound-induced proteinase inhibitor in plant leaves: a possible defense mechanism against insects. Science 175:776–777.
Hägele, B. F., and Rowell-rahier, M. 1999. Dietary mixing in three generalist herbivores: nutrient complementation or toxin dilution? Oecologia 119:521–533.
Halitschke, R., and Baldwin, I. T. 2005. Jasmonates and related compounds in plant-insect interactions. J. Plant Growth Regul. 23:238–245.
Hegedus, D., Baldwin, D., O’grady, M., Braun, L., Gleddie, S., Sharpe, A., Lydiate, D., and Erlandson, M. 2003. Midgut proteases from Mamestra configurata (Lepidoptera: Noctuidae) larvae: characterization, cDNA cloning, and expressed sequence tag analysis. Arch. Insect Biochem. Physiol. 53:30–47.
Howe, G. A., Lighter, J., Browse, J., and Ryan, C. A. 1996. An octadecanoid pathway mutant (JL5) of tomato is compromised in signaling for defense against insect attack. Plant Cell 8:2067–2077.
Howe, G. A., and Ryan, C. A. 1999. Suppressors of systemin signaling identify genes in the tomato wound response pathway. Genetics 153:1411–1421.
Jongsma, M. A., and Bolter, C. 1997. The adaptation of insects to plant protease inhibitors. J. Insect Physiol. 10:885–895.
Karban, R., and Myers, J. H. 1989. Induced plant response to herbivory. Ann. Rev. Ecol. System. 20:331–348.
Lee, S. S. T., and Scott, J. G. 1989. An improved method for preparation, stabilization and storage of house fly (Diptera: Muscidae) microsomes. J. Econ. Entomol. 82:1559–1563.
Li, C., Williams, M. M., Loh, Y.-T., Lee, G. I., and Howe, G. A. 2002. Resistance of cultivated tomato to cell content-feeding herbivores is regulated by the octadecanoid-signalling pathway. Plant Physiol. 130:494–503.
Li, H., Oppert, B., Higgins, R. A., Huang, F., Zhu, K. Y., and Buschmann, L. L. 2004. Comparative analysis of proteinase activities of Bacillus thuringiensis-resistant and—susceptible Ostrinia nubilalis (Lepidoptera: Crambidae). Insect Biochem. Mol. Biol. 34:753–762.
Lightner, J., Pearce, G., Ryan, C. A., and Browse, J. 1993. Isolation of signaling mutants of tomato (Lycopersicon esculentum). Mol. Gen. Genom. 241:595–601.
Mahanil, S., Attajarusit, J., Stout, M. J., and Thipyang, P. 2008. Overexpression of tomato polyphenol oxidase increases resistance to common cutworm. Plant Science 174:456–466.
Mcgurl, B., Orozco-cardeas, M., Pearce, G., and Ryan, C. A. 1994. Overexpression of the prosystemin gene in transgenic tomato plants generates a systemic signal that constitutively induces proteinase-inhibitor synthesis. Proc. Natl. Acad. Sci. USA 91:9799–9802.
Raubenheimer, D., and Simpson, S. J. 1992. Analysis of covariance: an alternative to nutritional indices. Entomol. Exp. Appl. 62:221–231.
Rayapuram, C., and Baldwin, I. T. 2006. Using nutritional indices to study LOX-3 dependent insect resistance. Plant Cell Environ. 29:1585–1594.
Ryan, C. A. 2000. The systemin signaling pathway: differential activation of plant defensive genes. Biochim. Biophys. Acta 1477:112–121.
Schoonhoven, L. M., Van loon, J. J. A., and Dicke, M. 2005. Insect-plant biology, 2nd Edition. Oxford University Press, Oxford.
Slansky, F., and Scriber, J. M. 1985. Food consumption and utilization, pp. 88–151, in G. A. Kerkut and L. I. Gilbert (eds.). Comprehensive Insect Physiology Biochemistry and Pharmacology. Volume 4. Regulation: Digestion, Nutrition, Excretion. Pergamon, New York.
Stout, M. J., and Duffey, S. S. 1996. Characterization of induced resistance in tomato plants. Entomol. Exp. Appl. 79:273–283.
Thaler, J. S., Farag, M. A., Paré, P. W., and Dicke, M. 2002. Jasmonate-deficient plants have reduced direct and indirect defences against herbivores. Ecology Lett. 5:764–774.
Wadleigh, R. W., and Yu, S. 1987. Glutathione transferase activity of fall armyworm larvae toward α,β-unsaturaqted carbonyl allelochemicals and its induction by allelochemicals. Insect Biochem. 17:759–764.
Wasternack, C. 2007. Jasmonates: An update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann. Bot. 100:681–697.
Yu, S. J. 1982. Host plant induction of glutathione S-transferase in the fall armyworm. Pestic. Biochem. Physiol. 18:101–106.
Yu, S. J. 1984. Interactions of allelochemicals with detoxification enzymes of insecticide-susceptible and resistant fall armyworms. Pestic. Biochem. Physiol. 22:60–68.
Acknowledgements
Thank you to C. Leichter and A. Tuccillo for technical assistance, and Drs. P. Wang, S. Behmer, and G. Howe for advice and the tomato varieties. Funding was provided by NSERC (IS), National Research Initiative of the USDA Cooperative State Research, Education and Extension Service, Grant Number # 2006-35302-17431 (JST), and the Daljit and Elaine Sarkaria Professorship (JGS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Scott, I.M., Thaler, J.S. & Scott, J.G. Response of a Generalist Herbivore Trichoplusia ni to Jasmonate-Mediated Induced Defense in Tomato. J Chem Ecol 36, 490–499 (2010). https://doi.org/10.1007/s10886-010-9780-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-010-9780-8