Abstract
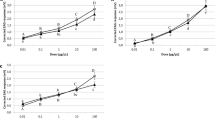
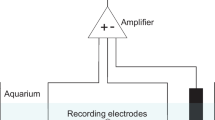
The essential oil constituents citral, geraniol, and eugenol have toxic or repellent properties that are utilized by a variety of organisms to deter natural enemies. Their mechanism of action is unknown, but some essential oils such as eugenol are claimed to act on insects by specific binding to octopamine receptors. We studied their effects on the isolated buccal ganglia of Planorbis corneus, having demonstrated that they caused cessation of feeding and death when added to the aquarium water (approximately 5 × 10−4 mol l−1). They abolished spike activity at 2 × 10−3 mol l−1 but at lower doses (threshold 5 × 10−5 mol l−1) they resembled octopamine in eliciting burst firing indicative of the fictive feeding sequence. However, the octopamine antagonists phentolamine (10−5 mol l−1), yohimbine (10−4 mol l−1; which blocked hyperpolarizing octopamine responses), and metoclopramide (10−4 mol l−1; which blocked depolarization) had no effect on any of the responses to the oils. The oils produced incomplete block of excitatory and inhibitory responses to octopamine even at high doses (10−3 mol l−1), and they had similar effects on responses to dopamine and acetylcholine. The oils (10−5–2 × 10−3 mol l−1) all increased the frequency of contractions of the isolated esophagus and progressively reduced their amplitude. The effects were similar to those of octopamine but were not blocked by phentolamine, metoclopramide, or prolonged exposure of the esophagus to octopamine. The results suggest a variety of actions that could contribute to toxic effects in molluscs, but there was no evidence for specific actions on octopamine receptors, either as agonists or antagonists. Evidence is presented that nonspecific depolarization produces their octopamine-like actions.






Similar content being viewed by others
References
Amin, G., Salehi Sourmaghi, M. H., Zahedi, M., Khanavi, M., and Samadi, N. 2005. Essential oil composition and antimicrobial activity of Oliveria decumbens. Fitoterapia 76:704–707.
Arshavsky, Y., Deliagina, T. G., Meizerov, E. S., Orlovsky, G. N., and Panchin, Y. 1988. Control of feeding movements in the freshwater snail Planorbis corneus. I. Rhythmical neurons of buccal ganglia. Exp. Brain Res. 70:310–322.
Benjamin, P. R. 1983. Gastropod feeding: behavioural and neural analysis of a complex multicomponent system, pp. 159–193, in A. Roberts, and B. Roberts (eds.). Neural Origin of Rhythmic Movements. Cambridge University Press, Cambridge.
Berry, M. S., and Cottrell, G. A. 1975. Excitatory, inhibitory and biphasic synaptic potentials mediated by an identified dopamine-containing neuron. J. Physiol. Lond. 244:589–612.
Bhat, S. V., Nagasampagi, B. A., and Sivakumar, M. 2005. Chemistry of Natural Products. Narosa Publishing House, New Delhi.
Brodin, P., and Røed, A. 1984. Effects of eugenol on rat phrenic nerve and phrenic nerve-diaphragm preparations. Arch. Oral Biol. 29:611–615.
Chadha, M. S., Eisner, T., Monro, A., and Meinwald, J. 1962. Defense mechanisms of arthropods-VII. Citronellal and citral in the mandibular gland secretion of the ant Acanthomyops claviger (Roger). J. Insect Physiol. 8:175–176.
Cheng, S.-S., Chang, H.-T., Chang, S.-T., Tsai, K.-H., and Chen, W.-J. 2003. Bioactivity of selected plant essential oils against the yellow fever mosquito Aedes aegypti larvae. Biores. Technol. 89:99–102.
Clark, T. E., and Appleton, C. C. 1997. The molluscicidal activity of Apodytes dimidiata E. Meyer ex Arn (Icacinaceae), Gardenia thunbergia L.f. (Rubiaceae) and Warburgia salutaris (Bertol. F.) Chiov. (Cannelaceae), three South African plants. J. Ethnopharm. 56:15–30.
Coats, J. R., Karr, L. L., and Drewes, C. D. 1991. Toxicity and neurotoxic effects of monoterpenoids in insects and earthworms, pp. 305–316, in P. A. Hedin (ed.). Naturally Occurring Pest Bioregulators. ACS Symposium Series 449. American Chemical Society, Washington DC.
De Souza, C. P., De Oliveira, A. B., Araujo, N., and Katz, N. 1991. Effects of eugenol and derivatives on Biomphalaria glabrata. Rev. Bras. Biol. 51:295–299.
Dyrynda, P. E. J. 1986. Defensive strategies of modular organisms. Phil. Trans. R. Soc. Lond. B 313:227–243.
Elliott, C. J. H., and Vehovszky, Á. 2001. Octopamine is not just for arthropods. Physiol. News 45:17–19.
Enan, E. E. 2001. Insecticidal activity of essential oils: octopaminergic sites of action. Comp. Biochem. Physiol. C 130:325–337.
Enan, E. E. 2005. Molecular and pharmacological analysis of an octopamine receptor from American cockroach and fruit fly in response to plant essential oils. Arch. Insect Biochem. Physiol. 59:161–171.
Evans, P. D., and Robb, S. 1993. Octopamine receptor subtypes and their modes of action. Neurochem. Res. 18:869–874.
Evans, P. D., and Maqueira, B. 2005. Insect octopamine receptors: a new classification scheme based on studies of cloned Drosophila G-protein coupled receptors. Invertebr. Neurosci. 5:111–118.
Evans, P. D., Robb, S., Cheek, T. R., Reale, V., Hannan, F. L., Swales, L. S., Hall, M., and Midgley, J. M. 1995. Agonist-specific coupling of G-protein coupled receptors to second messenger systems. Prog. Brain Res. 106:259–268.
Finney, D. J. 1952. Probit Analysis. Cambridge University Press, Cambridge.
Ghelardini, C., Galeotti, N., Salvatore, G., and Mawwanti, G. 1999. Local anaesthetic activity of the essential oil of Lavandula angustifolia. Planta Medica 65:700–703.
Hiripi, L., Vehovszky, Á., Juhos, S., and Elekes, K. 1998. An octopaminergic system in the CNS of the snails, Lymnaea stagnalis and Helix pomatia. Phil. Trans. R. Soc. Lond. B 353:1621–1629.
Huang, Y., Ho, S. H., Lee, H. -C., and Yap, Y. -L. 2002. Insecticidal properties of eugenol, isoeugenol and methyleugenol and their effects on nutrition of Sitophilus zeamais Motsch. (Coleoptera: Curculionidae) and Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J. Stored Products Res. 38:403–412.
Isman, M. B. 2000. Plant essential oils for pest and disease management. Crop Prot. 19:603–608.
Katerinopoulos, H. E., Pagona, G., Afratis, A., and Stratigakis, N. 2005. Composition and insect attracting activity of the essential oil of Rosmarinus officinalis. J. Chem. Ecol. 31:111–122.
Kostyukovsky, M., Rafaeli, A., Gileadi, C., Demchenko, N., and Shaaya, E. 2002. Activation of octopaminergic receptors by essential oil constituents isolated from aromatic plants: possible mode of action against insect pests. Pest Manag. Sci. 58:1101–1106.
Lahlou, M., and Berrada, R. 2001. Potential of essential oils in schistosomiasis control in Morocco. Int. J. Aroma. 11:87–96.
Mikich, S. B., Bianconi, G. V., Maia, B. H. L. N. S., and Teixeira, S. D. 2003. Attraction of the fruit-eating bat Carollia perspicillata to Piper gaudichaudianum essential oil. J. Chem. Ecol. 29:2379–2383.
Nakamura, Y., Miyamoto, M., Murakami, A., Ohigashi, H., Osawa, T., and Uchida, K. 2003. A phase II detoxification enzyme inducer from lemongrass: identification of citral and involvement of electrophilic reaction in the enzyme induction. Biochem. Biophys. Res. Commun. 302:593–600.
Ngoh, S. P., Choo, L. E. W., Pang, F. Y., Huang, Y., Kini, M. R., and Ho, S. H. 1998. Insecticidal and repellent properties of nine volatile constituents of essential oils against the American cockroach, Periplaneta americana. Pestic. Sci. 54:261–268.
O’reilly-Wapstra, J. M., Potts, B. M., Mcarthur, C., Davies, N. W., and Tilyard, P. 2005. Inheritance of resistance to mammalian herbivores and of plant defensive chemistry in a Eucalyptus species. J. Chem. Ecol. 31:519–537.
Pavela, R. 2005. Insecticidal activity of some essential oils against larvae of Spodoptera littoralis. Fitoterapia 76:691–696.
Price, D. N., and Berry, M. S. 2006. Comparison of effects of octopamine and insecticidal essential oils on activity in the nerve cord, foregut, and dorsal unpaired median neurons of cockroaches. J. Insect Physiol. 52:309–319.
Singh, S., Singh, V. K., and Singh, D. K. 1997. Molluscicidal activity of some common spice plants. Biol. Agric. Hortic. 14:237–249.
Szabadics, J., and Erdélyi, L. 2000. Pre- and postsynaptic effects of eugenol and related compounds on Helix pomatia L. Acta Biol. Hung. 51:265–273.
Vasudevan, P., Kashyap, S., and Sharma, S. 1997. Tagetes: a multipurpose plant. Biores. Tech. 62:29–35.
Vehovszky, Á., and Elliott, C. J. H. 2002. Heterosynaptic modulation by the octopaminergic OC interneurons increases the synaptic outputs of protraction phase interneurons (SO, N1L) in the feeding system of Lymnaea stagnalis. Neurosci. 115:483–494.
Vehovszky, Á., Elliott, C. J. H., Voronezhskaya, E. E., Hiripi, L., and Elekes, K. 1998. Octopamine: a new feeding modulator in Lymnaea. Phil. Trans. R. Soc. Lond. B 353:1631–1643.
Acknowledgements
DNP was supported by a studentship from the Department of Biological Sciences, School of the Environment and Society, Swansea University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Price, D.N., Berry, M.S. Neurophysiological Effects of Naturally Occurring Defensive Compounds on the Freshwater Snail Planorbis corneus: Comparison with Effects in Insects. J Chem Ecol 34, 994–1004 (2008). https://doi.org/10.1007/s10886-008-9511-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9511-6