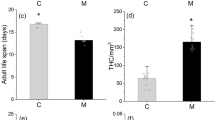
Abstract
The quality of available food may affect insect herbivores directly (via growth and survivorship) and/or indirectly (by modifying insect vulnerability to parasitoids and pathogens). We examined the relationship between different phenolic compounds, belonging to various phenolic groups, in Betula pubescens spp. czerepanovii (mountain birch) foliage and the larval performance of the geometrid Epirrita autumnata (autumnal moth). Direct effects on insect performance were described by pupal weight, developmental rate, and survivorship; indirect effects were described by the encapsulation rate of an implant inserted into the insect hemocoel, a commonly used way to describe insect immune defense. We found profound differences in the effects of different phenolic categories: several individual hydrolyzable tannins were associated positively with larval performance but negatively with level of immune defense, whereas flavonoid glycosides were inversely related to larval survival but showed no association with the larvae immune defense.

Similar content being viewed by others
References
Agrawal, A. A. 1999. Induced responses to herbivory in wild radish: Effects on several herbivores and plant fitness. Ecology 80:1713.
Auerbach, M. and Alberts, J. D. 1992. Occurrence and performance of the aspen blotch miner, Phyllonorycter salicifoliella, on three host-tree species. Oecologia 89:1.
Ayres, M. P., Clausen, T. P., Maclean, S. F. Jr., Redman, A. M., and Reichardt, P. B. 1997. Diversity of structure and antiherbivore activity in condensed tannins. Ecology 78:1696.
Benz, G. 1974. Negative rückkoppelung durch raum—und nahrungskonkurrenz sowie zyklishe veränderung der nahrungsgrundlage als regelprinzip in der populationsdynamik des grauen lärchenwiklers, Zeiraphera diniana (guenée) (Lep. Tortricidae). Z. Angew. Entomol. 76:196.
Berryman, A. A. 2002. Population Cycles. The Case for Trophic Interactions. Oxford University Press, New York.
Bjorkman, C. and Larsson, S. 1991. Pine sawfly defense and variation in host plant resin acids—a trade-off with growth. Ecol. Entomol. 16:283.
Bylund, H. 1995. Long-term interactions between the autumnal moth and mountain birch: The roles of resources, competition. natural enemies, and weather. Dissertation, Sveriges Lantbruks Universitet.
Cook, S. P., Webb, R. E., Podgwaite, J. D., and Reardon, R. C. 2003. Increased mortality of gypsy moth Lymantria dispar (L.) (Lepidoptera: Lymantriidae) exposed to gypsy moth nuclear polyhedrosis virus in combination with the phenolic glycoside salicin. J. Econ. Entomol. 96:1662.
Cory, J. S. and Hoover, K. 2006. Plant-mediated effects in insect-pathogen interactions. Tree 21:278.
Dixon, P. M. 2001. The bootstrap and the jackknife—describing the precision of ecological indices, pp. 267, in S. M. Scheiner and J. Gurevitch (eds.). Design and Analysis of Ecological Experiments. Chapman Hall, New York.
Feeny, P. 1970. Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth caterpillars. Ecology 51:565.
Felton, G. W., Duffey, S. S., Vail, P. V., Kaya, H. K., and Manning, J. 1987. Interaction of nuclear polyhedrosis virus with catechols: Potential incompatibility for host-plant resistance against noctuid larvae. J. Chem. Ecol. 13:947.
Gallardo, F., Boethel, D. J., Fuxa, J. R., and Richter, A. 1990. Susceptibility of Heliothis zea (Boddie) larvae to Nomuraea rileyi (Farlow) Samson: Effects of α-tomatine at the third trophic level. J. Chem. Ecol. 16:1751.
Gorman, M. J., Cornel, A. J., Collins, F. H., and Paskewitz, S. M. 1996. A shared genetic mechanism for melanotic encapsulation of CM-Sephadex beads and the malaria parasite, Plasmodium cynomolgi B, in the mosquito Anopheles gambiae. Exp. Parasitol. 84:380.
Haukioja, E. 2003. Putting the insect into the birch–insect interaction. Oecologia 136:161.
Haukioja, E. 2005a. Plant defenses and population fluctuations of forest defoliators: Mechanism-based scenarios. Ann. Zool. Fenn. 42:313.
Haukioja, E. 2005b. Tree defenses against insects, pp. 279, in E. Bent and S. Tuzun (eds.). Multigenic and Induced Systemic Resistance in Plants. Kluwer, Academic Publishers, New York.
Haukioja, E. and Neuvonen, S. 1985. Induced long-term resistance of birch foliage against defoliators: Defensive or incidental? Ecology 66:1303.
Haukioja, E., Ossipov, V., and Lempa, K. 2002. Interactive effects of leaf maturation and phenolics on consumption and growth of a geometrid moth. Entomol. Exp. Appl. 104:125.
Havill, N. P. and Raffa, K. F. 2000. Compound effects of induced plant responses on insect herbivores and parasitoids: Implications for tritrophic interactions. Ecol. Entomol. 25:171.
Hilker, M. and Schulz, S. 1994. Composition of larval secretion of Chrysomela lapponica (Coleoptera, Chrysomelidae) and its dependence on host-plant. J. Chem. Ecol. 20:1075.
Holton, M. K., Lindroth, R. L., and Nordheim, E. V. 2003. Foliar quality influences tree-herbivore-parasitoid interactions: Effects of elevated CO2, O3, and plant genotype. Oecologia 137:233.
Hoover, K., Yee, J. L., Schultz, C. M., Rocke, D. M., Hammock, B. D., and Duffey, S. S. 1998. Effects of plant identity and chemical constituents on the efficacy of a baculovirus against Heliothis virescens. J. Chem. Ecol. 24:221–252.
Hunter, M. D. and Schultz, J. C. 1993. Induced plant defenses breached? phytochemical induction protects an herbivore from disease. Oecologia 94:195.
Kaitaniemi, P., Ruohomäki, K., Ossipov, V., Haukioja, E., and Pihlaja, K. 1998. Delayed induced changes in the biochemical composition of host plant leaves during an insect outbreak. Oecologia 116:182.
Kallio, P. and Mäkinen, Y. 1978. Vascular flora of Inari Lapland. 4. Betulaceae. Rep. Kevo Subarctic Res. Stat. 14:38.
Kapari, L., Haukioja, E., Rantala, M. J., and Ruuhola, T. 2006. Defoliating insect immune defense interacts with induced plant defense during a population outbreak. Ecology 87:291.
Karban, R. and Baldwin, I. T. 1997. Induced Responses to Herbivory. The University Chicago Press, Chicago.
Kause, A., Ossipov, V., Haukioja, E., Lempa, K., Hanhimäki, S., and Ossipova, S. 1999a. Multiplicity of biochemical factors determining quality of growing birch leaves. Oecologia 120:102.
Kause, A., Saloniemi, I., Haukioja, E., and Hanhimäki, S. 1999b. How to become large quickly: Quantitative genetics of growth and foraging in a flush feeding lepidopteran larva. J. Evol. Biol. 12:471.
Keating, S. T., Yendol, W. G., and Schultz, J. C. 1988. Relationship between susceptibility of gypsy moth larvae (Lepidoptera: Lymantriidae) to a baculovirus and host plant foliage constituents. Environ. Entomol. 17:952.
Keating, S. T., Hunter, M. D., and Schultz, J. C. 1990. Leaf phenolic inhibition of gypsy moth nuclear polyhedrosis virus. J. Chem. Ecol. 16:1445.
Klemola, T., Ruohomäki, K., Andersson, T., and Neuvonen, S. 2004. Reduction in size and fecundity of the autumnal moth, Epirrita autumnata, in the increase phase of a population cycle. Oecologia 141:47.
Lindroth, R. L., Hwang, S., and Osier, T. L. 1999. Phytochemical variation in quaking aspen: Effects on gypsy moth susceptibility to nuclear polyhedrosis virus. J. Chem. Ecol. 25:1331.
Ludlum, C. T., Felton, G. W., and Duffey, S. S. 1991. Plant defenses: Chlorogenic acid and polyphenol oxidase enhance toxicity of Bacillus thuringiensis subsp. kurstaki to Heliothis zea. J. Chem. Ecol. 17:217.
Ode, P. J., Berenbaum, M. R., Zangerl, A. R., and Hardy, I. C. W. 2004. Host plant, host plant chemistry and the polyembryonic parasitoid Copidosoma sosares: Indirect effects in a tritrophic interaction. Oikos 104:388.
Ojala, K., Julkunen-Tiitto, R., Lindström, L., and Mappes, J. 2005. Diet affects the immune defence and life-history traits of an arctiid moth Parasemia plantaginis. Evol. Ecol. Res. 7:1153.
Ossipov, V., Haukioja, E., Ossipova, S., Hanhimäki, S., and Pihlaja, K. 2001. Phenolic and phenolic-related factors as determinants of suitability of mountain birch leaves to an herbivorous insect. Biochem. Syst. Ecol. 29:223.
Ossipova, S., Ossipov, V., Haukioja, E., Loponen, J., and Pihlaja, K. 2001. Proanthocyanidins from mountain birch leaves: Quantification and properties. Phytochem. Anal. 12:128.
Parry, D., Herms, D. A., and Mattson, W. J. 2003. Responses of an insect folivore and its parasitoids to multiyear experimental defoliation of aspen. Ecology 84:1768.
Paskewitz, S. and Riehle, M. A. 1994. Response of plasmodium refractory and susceptible strains of Anopheles gambiae to inoculated Sephadex beads. Dev. Comp. Immunol. 18:369.
Rantala, M. J. and Roff, D. A. 2005. An analysis of trade-offs in immune function, body size and development time in the Mediterranean field cricket, Gryllus bimaculatus. Funct. Ecol. 19:323.
Riipi, M., Ossipov, V., Lempa, K., Haukioja, E., Koricheva, J., Ossipova, S., and Pihlaja, K. 2002. Seasonal changes in birch leaf chemistry: Are there trade-offs between leaf growth and accumulation of phenolics? Oecologia 130:380.
Riipi, M., Haukioja, E., Lempa, K., Ossipov, V., Ossipova, S., and Pihlaja, K. 2004. Ranking of individual mountain birch trees in terms of leaf chemistry: seasonal and annual variation. Chemoecology 14:31.
Roth, S., Knorr, C., and Lindroth, R. L. 1997. Dietary phenolics affects performance of the gypsy moth (Lepidoptera: Lymantriidae) and its parasitoid Cotesia melanoscela (Hymenoptera: Braconidae). Environ. Entomol. 26:668.
Ruohomäki, K. 1994. Larval parasitism in outbreaking and non-outbreaking populations of Epirrita autumnata (Lepidopteran, Geometridae). Entomol. Fenn. 5:27.
Ruohomäki, K., Tanhuanpää, M., Ayres, M. P., Kaitaniemi, P., Tammaru, T., and Haukioja, E. 2000. Causes of cyclicity of Epirrita autumnata (Lepidoptera, Geometridae): Grandiose theory and tedious practice. Popul. Ecol. 42:211.
Salminen, J., Ossipov, V., Loponen, J., Haukioja, E., and Pihlaja, K. 1999. Characterisation of hydrolysable tannins from leaves of Betula pubescens by high-performance liquid chromatography-mass spectrometry. J. Chromatogr. A. 864:283.
Salminen, J., Ossipov, V., Haukioja, E., and Pihlaja, K. 2001. Seasonal variation in the content of hydrolysable tannins in leaves of Betula pubescens. Phytochemistry 57:15.
Sas Institute. 1990. SAS/STAT User’s Guide, 6th Version. SAS Institute, Cary, North Carolina, USA.
Tenow, O. 1972. The outbreaks of Oporinia autumnata Bkh. and Operophtera spp. (Lep.; Geometridae) in the Scandinavian mountain chain and northern Finland 1862–1968. Dissertation, University of Uppsala.
Vainio, L., Hakkarainen, H., Rantala, M. J., and Sorvari, J. 2004. Individual variation in immune function in the ant Formica exsecta; effects of the nest, body size and sex. Evol. Ecol. 18:75.
Young, S. Y., Yang, J. G., and Felton, G. W. 1995. Inhibitory effects of dietary tannins on the infectivity of a nuclear polyhedrosis virus to Helicoverpa zea (Noctuidae: Lepidoptera). Biol. Cont. 5:145.
Acknowledgements
We thank the staff of the Kevo Subartic Research Station for making this study possible. Irma Saloniemi and Kari Saikkonen gave helpful comments on the manuscript. Ellen Valle checked the language. The study was supported financially by the Academy of Finland: project 212712 (EH), 206144 (TR), 201073 (VO), and 207294 (MJR). The experiments presented comply with the current laws of Finland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haviola, S., Kapari, L., Ossipov, V. et al. Foliar Phenolics are Differently Associated with Epirrita autumnata Growth and Immunocompetence. J Chem Ecol 33, 1013–1023 (2007). https://doi.org/10.1007/s10886-007-9271-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-007-9271-8