Abstract
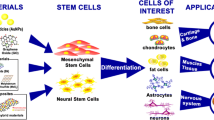
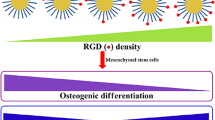
Gold nanoparticles (AuNPs) have been extensively studied for applications in biomedical sciences. AuNPs have exceptional advantages in cells studies include good biocompatibility and very low cytotoxicity. The eco-friendly synthesis of AuNPs with various physical characteristics is simple, and led to the fabrication of nanomaterials showing different biological functions routing cellular physiology. A growing number of recent studies focused on the promoting effect of AuNPs on stem cells osteogenic differentiation. This includes the ability of the AuNP to activate Wnt/β-catenin pathway, ERK/MAPK pathway and p38 MAPK pathway, resulting in the activation of the transcription factors for the osteogenic differentiation. This technology represents a promising therapeutic strategy to heal patients with bone injury, by regenerating bone cells using stem cells therapy. In this review, recent applications of AuNPs in stem cells osteogenic differentiation, along with the related inducing mechanism are discussed. We also provide recent updates on the AuNP synthesis methods.



Similar content being viewed by others
References
J. Li, J. En, J. Li, J. Zhang, X. Wang, N. Kawazoe, and G. Chen (2016). Gold Nanoparticle Size and Shape Influence on Osteogenesis of Mesenchymal Stem Cells. Nanoscale 8, (15), 7992–8007.
A. Alkilany, S. Lohse, and C. Murphy (2012). The Gold Standard: Gold Nanoparticle Libraries to Understand the Nano–bio Interface. Acc. Chem. Res. 46, (3), 650–661.
L. Ricles, S. Nam, and E. Trevino (2014). A Dual Gold Nanoparticle System for Mesenchymal Stem Cell Tracking. J. Mater. Chem. 2, (46), 8220–8230.
Y. S. Zhang, Y. Wang, L. Wang, Y. Wang, X. Cai, C. Zhang, L. V. Wang, and Y. Xia (2013). Labeling Human Mesenchymal Stem Cells with Gold Nanocages for in Vitro and in Vivo Tracking by Two-Photon Microscopy and Photoacoustic Microscopy. Theranostics 3, (8), 532–543.
W. Ko, D. Heo, H. Moon, S. Lee, and M. Bae (2015). The Effect of Gold Nanoparticle Size on Osteogenic Differentiation of Adipose-Derived Stem Cells. J. Colloid Interface Sci. 438, 68–76.
A. Higuchi, S.H. Kao, Q.D. Ling, Y.M. Chen, H.F. Li, A.A. Alarfaj, M.A. Munusamy, K. Murugan, S.C. Chang, H.C. Lee, S.T. Hsu, S. Suresh Kumar, and A. Umezawa (2015). Long-Term Xeno-Free Culture of Human Pluripotent Stem Cells on Hydrogels with Optimal Elasticity. Scientific Reports, 5, 18136.
I. Peng, C. Yeh, Y. Lu, S. Muduli, Q. Ling, and A. Alarfaj (2016). Continuous Harvest of Stem Cells via Partial Detachment from Thermoresponsive Nanobrush Surfaces. Biomaterials 76, 76–86.
K. Russell, D. Phinney, M. Lacey, and B. Barrilleaux (2010). In Vitro High-capacity Assay to Quantify the Clonal Heterogeneity in Trilineage Potential of Mesenchymal Stem Cells Reveals a Complex Hierarchy of Lineage. Stem Cells 28, (4), 788–798.
A. Higuchi, S. Kumar, Q. Ling, and A. Alarfaj (2017). Polymeric Design of Cell Culture Materials That Guide the Differentiation of Human Pluripotent Stem Cells. Prog. Polym. Sci. 65, 83–126.
D. Chen, L. Chen, Q. Ling, M. Wu, and C. Wang (2014). Purification of Human Adipose-Derived Stem Cells from Fat Tissues Using PLGA/silk Screen Hybrid Membranes. Biomaterials 35, (14), 4278–4287.
C. Yi, D. Liu, C.-C. C. Fong, J. Zhang, and M. Yang (2010). Gold Nanoparticles Promote Osteogenic Differentiation of Mesenchymal Stem Cells through p38 MAPK Pathway. ACS Nano 4, (11), 6439–6448.
T. Pan, W. Song, H. Gao, T. Li, X. Cao, S. Zhong, and Y. Wang (2016). MiR-29b-Loaded Gold Nanoparticles Targeting to the Endoplasmic Reticulum for Synergistic Promotion of Osteogenic Differentiation. ACS Appl. Mater. Interfaces. 8, (30), 19217–19227.
S. S. Y. Choi, M. M. S. Song, P. P. D. Ryu, A. A. T. N. Lam, S.-W. Joo, and S. Y. Lee (2015). Gold Nanoparticles Promote Osteogenic Differentiation in Human Adipose-Derived Mesenchymal Stem Cells through the Wnt/β-Catenin Signaling Pathway. Int. J. Nanomed. 10, 4383.
D. Zhang, D. Liu, J. Zhang, C. Fong, and M. Yang (2014). Gold Nanoparticles Stimulate Differentiation and Mineralization of Primary Osteoblasts through the ERK/MAPK Signaling Pathway. Mater. Sci. Eng., C 42, 70–77.
C. Bennett, K. Longo, and W. Wright (2005). Regulation of Osteoblastogenesis and Bone Mass by Wnt10b. Proc. Natl. Acad. Sci. U.S.A. 102, (9), 3324–3329.
R. M. Salasznyk, R. F. Klees, M. K. Hughlock, and G. E. Plopper (2004). ERK Signaling Pathways Regulate the Osteogenic Differentiation of Human Mesenchymal Stem Cells on Collagen I and Vitronectin. Cell Commun. Adhes. 11, (5–6), 137–153.
J. J. Li, N. Kawazoe, and G. Chen (2015). Gold Nanoparticles with Different Charge and Moiety Induce Differential Cell Response on Mesenchymal Stem Cell Osteogenesis. Biomaterials 54, 226–236.
X. Ji, X. Song, J. Li, Y. Bai, and W. Yang (2007). Size Control of Gold Nanocrystals in Citrate Reduction: The Third Role of Citrate. J. Am. Chem. Soc. 129, (45), 13939–13948.
A. Madu, P. Njoku, and G. Iwuoha (2011). Synthesis and Characterization of Gold Nanoparticles Using 1-Alkyl, 3-Methyl Imidazolium Based Ionic Liquids. Int. J. Phys. Sci. 6, (4), 635–640.
K. Mallick, Z. Wang, and T. Pal (2001). Seed-Mediated Successive Growth of Gold Particles Accomplished by UV Irradiation: A Photochemical Approach for Size-Controlled Synthesis. J. Photochem. Photobiol. A. 140, (1), 75–80.
K. Okitsu, Y. Mizukoshi, T. Yamamoto, and Y. Maeda (2007). Sonochemical Synthesis of Gold Nanoparticles on Chitosan. Mater. Lett. 61, (16), 3429–3431.
J. Turkevich, P. Stevenson, and J. Hillier (1951). A Study of the Nucleation and Growth Processes in the Synthesis of Colloidal Gold. Discuss. Faraday Soc. 11, 55–75.
M. Wuithschick, A. Birnbaum, S. Witte, and M. Sztucki (2015). Turkevich in New Robes: Key Questions Answered for the Most Common Gold Nanoparticle Synthesis. ACS Nano 9, (7), 7052–7071.
J. Li, R. Cai, N. Kawazoe, and G. Chen (2015). Facile Preparation of Albumin-Stabilized Gold Nanostars for the Targeted Photothermal Ablation of Cancer Cells. J. Mater. Chem. B 3, (28), 5806–5814.
Y. Xiang, X. Wu, D. Liu, L. Feng, and K. Zhang (2008). Tuning the Morphology of Gold Nanocrystals by Switching the Growth of 110 Facets from Restriction to Preference. J. Phys. Chem. 112, (9), 3203–3208.
S. Eustis and M. El-Sayed (2006). Why Gold Nanoparticles Are More Precious than Pretty Gold: Noble Metal Surface Plasmon Resonance and Its Enhancement of the Radiative and Nonradiative Properties. Chem. Soc. Rev. 35, (3), 209–217.
Y. Li, L. Feng, X. Shi, X. Wang, Y. Yang, K. Yang, T. Liu, G. Yang, and Z. Liu (2014). Surface Coating-Dependent Cytotoxicity and Degradation of Graphene Derivatives: Towards the Design of Non-Toxic, Degradable Nano-Graphene. Small 10, (8), 1544–1554.
L. Qi, Y. Guo, J. Luan, D. Zhang, and Z. Zhao (2014). Folate-Modified Bexarotene-Loaded Bovine Serum Albumin Nanoparticles as a Promising Tumor-Targeting Delivery System. J. Mater. Chem. B 2, (47), 8361–8371.
P. Quaresma, L. Soares, L. Contar, and A. Miranda (2009). Green Photocatalytic Synthesis of Stable Au and Ag Nanoparticles. Green Chem. 11, (11), 1889–1893.
P. Kuppusamy, S. Ichwan, and N. Parine (2015). Intracellular Biosynthesis of Au and Ag Nanoparticles Using Ethanolic Extract of Brassica Oleracea L. and Studies on Their Physicochemical and Biological Properties. J. Environ. Sci. 29, 151–157.
V. Gopinath, S. Priyadarshini, D. MubarakAli, M.F. Loke, N. Thajuddin, N.S. Alharbi, T. Yadavalli, and M. V. J. Alagiri (2016). Anti-Helicobacter Pylori, Cytotoxicity and Catalytic Activity of Biosynthesized Gold Nanoparticles: Multifaceted Application. Arab. J. Chem. https://doi.org/10.1016/j.arabjc.2016.02.005.
M. Ateeq, M. Shah, N. ul Ain, S. Bano, and I. Anis (2015). Green Synthesis and Molecular Recognition Ability of Patuletin Coated Gold Nanoparticles. Biosens. Bioelectron. 63, 499–505.
J. Subramaniam, K. Murugan, and C. Panneerselvam (2016). Multipurpose Effectiveness of Couroupita guianensis-Synthesized Gold Nanoparticles: High Antiplasmodial Potential, Field Efficacy against Malaria Vectors and Synergy. Environ. Sci. Pollut. Res. 23, (8), 7543–7558.
K. Tahir, S. Nazir, B. Li, A. Khan, Z. Khan, and P. Gong (2015). Nerium oleander Leaves Extract Mediated Synthesis of Gold Nanoparticles and Its Antioxidant Activity. Mater. Lett. 156, 198–201.
B. Paul, B. Bhuyan, D. Purkayastha, M. Dey, and S. Dhar (2015). Green Synthesis of Gold Nanoparticles Using Pogestemon benghalensis (B) O. Ktz. Leaf Extract and Studies of Their Photocatalytic Activity in Degradation of Methylene. Mater. Lett. 148, 37–40.
K. Murugan, G. Benelli, and C. Panneerselvam (2015). Cymbopogon citratus-Synthesized Gold Nanoparticles Boost the Predation Efficiency of Copepod Mesocyclops Aspericornis against Malaria and Dengue Mosquitoes. Exp. Parasitol. 153, 129–138.
N. Sharma, A. Pinnaka, and M. Raje (2012). Exploitation of Marine Bacteria for Production of Gold Nanoparticles. Microb. cell 11, (1), 86.
S. Rajasree and T. Suman (2012). Extracellular Biosynthesis of Gold Nanoparticles Using a Gram Negative Bacterium Pseudomonas fluorescens. Asian Pac. J. Trop. Dis. 2, S796–S799.
M. Agnihotri, S. Joshi, A. R. Kumar, and S. K. Zinjarde (2009). Biosynthesis of Gold Nanoparticles by the Tropical. Mater. Lett. 63, (15), 1231–1234.
P. Manivasagan, J. Venkatesan, and K. Kang (2015). Production of α-Amylase for the Biosynthesis of Gold Nanoparticles Using Streptomyces Sp. MBRC-82. Int. J. Biol. Macromol. 72, 71–78.
R. Bhambure, M. Bule, and N. Shaligram (2009). Extracellular Biosynthesis of Gold Nanoparticles Using Aspergillus niger—its Characterization and Stability. Chem. Eng. Technol. 32, (7), 1036–1041.
J. Kimling, M. Maier, B. Okenve, and V. Kotaidis (2006). Turkevich Method for Gold Nanoparticle Synthesis Revisited. J. Phys. Chem. 110, (32), 15700–15707.
S. Mathews, P. Gupta, R. Bhonde, and S. Totey (2011). Chitosan Enhances Mineralization during Osteoblast Differentiation of Human Bone Marrow-derived Mesenchymal Stem Cells, by Upregulating the Associated Genes. Cell Prolif. 44, (6), 537–549.
R. Muzzarelli (2011). Chitosan Composites with Inorganics, Morphogenetic Proteins and Stem Cells, for Bone Regeneration. Carbohydr. Polym. 83, (4), 1433–1445.
J. Deng, H. Zheng, X. Zheng, M. Yao, Z. Li, and C. Gao (2016). Gold Nanoparticles with Surface-Anchored Chiral Poly (Acryloyl-l(d)-Valine) Induce Differential Response on Mesenchymal Stem Cell Osteogenesis. Nano Res. 9, (12), 3683–3694.
D. Zhang, D. Liu, J. Zhang, C. Fong, and M. Yang (2014). Gold Nanoparticles Stimulate Differentiation and Mineralization of Primary Osteoblasts through the ERK/MAPK Signaling Pathway. Mater. Sci. Eng. 42, 70–77.
R. Fodde and T. Brabletz (2007). Wnt/β-Catenin Signaling in Cancer Stemness and Malignant Behavior. Curr. Opin. Cell Biol. 19, (2), 150–158.
Z. Li, W. Liao, Q. Zhao, T. Huan, and P. Feng (2014). Effect of Cbfa1 on Osteogenic Differentiation of Mesenchymal Stem Cells under Hypoxia Condition. Int. J. Clin. Exp. Med. 7, (3), 540.
B. M. Strem, K. C. Hicok, M. Zhu, I. Wulur, Z. Alfonso, R. E. Schreiber, and J. K. Fraser (2005). H. M. Multipotential Differentiation of Adipose Tissue-Derived Stem Cells. J. Med. 54, (3), 132–141.
T. Matsushita and S. Murakami The ERK MAPK Pathway in Bone and Cartilage Formation (In Protein Kinases, InTech, 2012).
M. Cargnello and P. Roux (2011). Activation and Function of the MAPKs and Their Substrates, the MAPK-Activated Protein Kinases. Microbiol. Mol. Biol. 75, (1), 50–83.
W. Zhang and H. Liu (2002). MAPK Signal Pathways in the Regulation of Cell Proliferation in Mammalian Cells. Cell Res. 12, (1), 9.
J. McCain (2013). The MAPK (ERK) Pathway: Investigational Combinations for the Treatment of BRAF-Mutated Metastatic Melanoma. Pharm. Ther. 38, (2), 96.
P. Shapiro (1998). Activation of the MKK/ERK Pathway during Somatic Cell Mitosis: Direct Interactions of Active ERK with Kinetochores and Regulation of the Mitotic 3F3/2 Phosphoantigen. J. Cell Biol. 142, (6), 1533–1545.
P. Ducy, R. Zhang, V. Geoffroy, A. L. Ridall, and G. Karsenty (1997). Osf2/Cbfa1: A Transcriptional Activator of Osteoblast Differentiation. Cell 89, (5), 747–754.
C. Ge, G. Xiao, D. Jiang, and R. T. Franceschi (2007). Critical Role of the Extracellular Signal–regulated kinase–MAPK Pathway in Osteoblast Differentiation and Skeletal Development. J. Cell Biol. 176, (5), 709–718.
A. Verma and F. Stellacci (2010). Effect of Surface Properties on Nanoparticle–Cell Interactions. Small 6, (1), 12–21.
J. F. Hillyer and R. M. Albrecht (2017). Gastrointestinal Persorption and Tissue Distribution of Differently Sized Colloidal Gold Nanoparticles. J. Pharm. Sci. 90, (12), 1927–1936.
S. Katz, R. Boland, and G. Santillan (2006). Modulation of ERK 1/2 and p38 MAPK Signaling Pathways by ATP in Osteoblasts: Involvement of Mechanical Stress-Activated Calcium Influx, PKC and Src Activation. Int. J. Biochem. 38, (12), 2082–2091.
G.-Y. Jung, Y.-J. Park, and J.-S. Han (2011). Mediation of Rac1 Activation by Kindlin-2: An Essential Function in Osteoblast Adhesion, Spreading, and Proliferation. J. Cell. Biochem. 112, (9), 2541–2548.
X. Li, N. Udagawa, K. Itoh, K. Suda, Y. Murase, T. Nishihara, and T. Suda (2002). T. N. p38 MAPK-Mediated Signals Are Required for Inducing Osteoclast Differentiation but Not for Osteoclast Function. Endocrinology 143, (8), 3105–3113.
T. Zarubin and J. Han (2005). Activation and Signaling of the p38 MAP Kinase Pathway. Cell Res. 15, (1), 11.
G. Benelli and C. Lukehart (2017). Special Issue: Applications of Green-Synthesized Nanoparticles in Pharmacology, Parasitology and Entomology. J. Clust. Sci. 28, (1), 1–2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Rights and permissions
About this article
Cite this article
Xiang, Z., Wang, K., Zhang, W. et al. Gold Nanoparticles Inducing Osteogenic Differentiation of Stem Cells: A Review. J Clust Sci 29, 1–7 (2018). https://doi.org/10.1007/s10876-017-1311-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-017-1311-0