Abstract
Purpose
Activated PI3Kδ syndrome 1 is a primary immunodeficiency disease, usually caused by heterozygous mutations in PIK3CD. We aimed to identify the cause of homozygous mutation at c.G3061A (p.E1021K) in a patient and the effect of allele dose in this mutation.
Methods
Genomic DNA from the parent-child trio was analyzed by next-generation sequencing. We performed phenotypic analyses in the patient and in Pik3cdE1024K+/+ mice.
Results
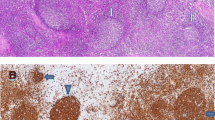
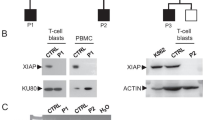
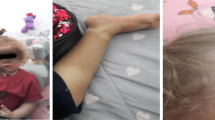
The patient was a girl harboring a homozygous mutation for p.E1021K in PIK3CD. At the age of 2 months, she began experiencing respiratory tract infections and lymphoproliferation, accompanied by bronchiectasis and extensive atelectasis in the lungs. She suffered from Haemophilus influenzae and Cytomegalovirus infections and experienced restricted growth and development. Whole-exome sequencing showed a region that included PIK3CD, with loss of heterozygosity (LOH) in chromosome 1 of the patient. The patient had not inherited any allele from her father in the LOH region. Copy number variation analysis showed no changes in the patient’s father and the patient. Ultra-deep sequencing of genomic DNA from the patient’s mother showed that the mutant allele frequency for c.G3061A was 1.64%. Thus, the presence of segmental maternal uniparental disomy and maternal gonosomal mosaicism resulted in the homozygous mutation. Lymphadenopathy, differentiation of activated T cells, and follicular B cells lymphopenia were found to be more prominent in Pik3cdE1024+/+ mice than in Pik3cdE1024+/− mice.
Conclusion
This report showed the coexistence of uniparental disomy and mosaicism in PIK3CD. Some immunological features were seen to be allele dose-dependent in the presence of p.E1021K mutation.



Similar content being viewed by others
References
Angulo I, Vadas O, Garçon F, Banham-Hall E, Plagnol V, Leahy TR, et al. Phosphoinositide 3-kinase δ gene mutation predisposes to respiratory infection and airway damage. Science. 2013;342:866–71.
Lucas CL, Chandra A, Nejentsev S, Condliffe AM, Okkenhaug K. PI3Kδ and primary immunodeficiencies. Nat Rev Immunol. 2016;16:702–14.
Han JM, Patterson SJ, Levings MK. The role of the PI3K signaling pathway in CD4+ T cell differentiation and function. Front Immunol. 2012;3:1–12.
Coulter TI, Chandra A, Bacon CM, Babar J, Curtis J, Screaton N, et al. Clinical spectrum and features of activated phosphoinositide 3-kinase δ syndrome: a large patient cohort study. J Allergy Clin Immunol. 2017;139:597–606.e4.
Jamee M, Moniri S, Zaki-Dizaji M, Olbrich P, Yazdani R, Jadidi-Niaragh F, et al. Clinical, immunological, and genetic features in patients with activated PI3Kδ syndrome (APDS): a systematic review. Clin Rev Allergy Immunol. 2019.
Wang Y, Wang W, Liu L, Hou J, Ying W, Hui X, et al. Report of a Chinese cohort with activated Phosphoinositide 3-kinase δ syndrome. J Clin Immunol. 2018;38:854–63.
Michalovich D, Nejentsev S. Activated PI3 kinase delta syndrome: from genetics to therapy. Front Immunol. 2018;9:1–6.
Lougaris V, Baronio M, Moratto D, Tampella G, Gazzurelli L, Facchetti M, et al. A novel monoallelic gain of function mutation in p110δ causing atypical activated phosphoinositide 3-kinase δ syndrome (APDS-1). Clin Immunol. 2019;200:31–4.
Heurtier L, Lamrini H, Chentout L, Deau MC, Bouafia A, Rosain J, et al. Mutations in the adaptor-binding domain and associated linker region of p110δ cause activated PI3K-δ. syndrome 1 (APDS1). Haematologica. 2017;102:e278–81.
Rae W, Gao Y, Ward D, Mattocks CJ, Eren E, Williams AP. A novel germline gain-of-function variant in PIK3CD. Clin Immunol. 2017;181:29–31.
Takeda AJ, Zhang Y, Dornan GL, Siempelkamp BD, Jenkins ML, Matthews HF, et al. Novel PIK3CD mutations affecting N-terminal residues of p110δ cause activated PI3Kδ syndrome (APDS) in humans. J Allergy Clin Immunol. 2017;140:1152–1156.e10.
Dornan GL, Siempelkamp BD, Jenkins ML, Vadas O, Lucas CL, Burke JE. Conformational disruption of PI3Kδ regulation by immunodeficiency mutations in PIK3CD and PIK3R1. Proc Natl Acad Sci U S A. 2017;114:1982–7.
Ding Y, Zhou L, Xia Y, Wang W, Wang Y, Li L, et al. Reference values for peripheral blood lymphocyte subsets of healthy children in China. J Allergy Clin Immunol. 2018;142:970–973.e8.
Harel T, Yoon WH, Garone C, Gu S, Coban-Akdemir Z, Eldomery MK, et al. Recurrent De novo and Biallelic variation of ATAD3A, encoding a mitochondrial membrane protein, results in distinct neurological syndromes. Am J Hum Genet. 2016;99:831–45.
Liu H, Tang XL, Liu JR, Li HM, Zhao SY. Clinical and genetic analysis for activated PI3K-δ syndrome by PIK3CD gene mutation. Chinese J Pediatr. 2016;54:698–702.
Soler-Palacín P, Garcia-Prat M, Martín-Nalda A, Franco-Jarava C, Rivière JG, Plaja A, et al. LRBA deficiency in a patient with a novel homozygous mutation due to chromosome 4 segmental uniparental isodisomy. Front Immunol. 2018;9:1–6.
Yamazawa K, Ogata T, Ferguson-Smith AC. Uniparental disomy and human disease: an overview. Am J Med Genet Part C Semin Med Genet. 2010;154:329–34.
Bravo García-Morato M, Nevado J, González-Granado LI, Sastre Urgelles A, Rodríguez Pena R, Ferreira CA. Chronic granulomatous disease caused by maternal uniparental isodisomy of chromosome 16. J Allergy Clin Immunol Pract. 2017;5:1146–8.
Mensa-Vilaró A, Bravo García-Morato M, de la Calle-Martin O, Franco-Jarava C, Martínez-Saavedra MT, González-Granado LI, et al. Unexpected relevant role of gene mosaicism in patients with primary immunodeficiency diseases. J Allergy Clin Immunol. 2019;143:359–68.
Spinner NB, Conlin LK. Mosaicism and clinical genetics. Am J Med Genet Part C Semin Med Genet. 2014;166:397–405.
Puck JM, Pepper AE, Bedard PM, Laframboise R. Female germ line mosaicism as the origin of a unique IL-2 receptor γ-chain mutation causing X-linked severe combined immunodeficiency. J Clin Invest. 1995;95:895–9.
Pang X, Zheng X, Kong X, Chai Y, Wang Y, Qian H, et al. A homozygous MITF mutation leads to familial Waardenburg syndrome type 4. Am J Med Genet Part A. 2019;179:243–8.
Soga K, Ishikawa K, Furuya T, Iida T, Yamada T, Ando N, et al. Gene dosage effect in spinocerebellar ataxia type 6 homozygotes: a clinical and neuropathological study. J Neurol Sci. 2017;373:321–8.
Fabrizi GM, Pellegrini M, Angiari C, Cavallaro T, Morini A, Taioli F, et al. Gene dosage sensitivity of a novel mutation in the intracellular domain of P0 associated with Charcot-Marie-tooth disease type 1B. Neuromuscul Disord. 2006;16:183–7.
Edwards N, Olinger E, Adam J, Kelly M, Schiano G, Ramsbottom SA, et al. A novel homozygous UMOD mutation reveals gene dosage effects on uromodulin processing and urinary excretion. Nephrol Dial Transplant. 2017;32:1994–9.
Madsen RR, Knox RG, Pearce W, Lopez S, Mahler-Araujo B, McGranahan N, et al. Oncogenic PIK3CA promotes cellular stemness in an allele dose-dependent manner. Proc Natl Acad Sci U S A. 2019;116:8380–9.
Lucas CL, Kuehn HS, Zhao F, Niemela JE, Deenick EK, Palendira U, et al. Dominant-activating germline mutations in the gene encoding the PI(3)K catalytic subunit p110δ result in T cell senescence and human immunodeficiency. Nat Immunol. 2014;15:88–97.
Acknowledgments
We thank the patients and their families for taking part in the study. We also thank the doctors, nurses, and other health care providers at the Children’s Hospital of Chongqing Medical University. We thank the staffs of Chigene (Beijing) Translational Medical Research Center Co. Ltd. for providing next-generation sequencing, analyzing genetic diseases, and discussion.
Funding
This work was supported by the Natural Science Foundation of China (81974255).
Author information
Authors and Affiliations
Contributions
Y. W., X. C., Q. Y., and W. T. performed the experiments and analyzed the data. Y. J. and L. Zhou provided help in performing the research. Y. A., Z. Z., and X. T. managed the patients. Y. W. and X. Z. wrote the manuscript. X. Z. designed the study and provided the financial support.
Corresponding author
Ethics declarations
Informed consent was obtained from all the participants. This study was conducted in accordance with the tenets of the Declaration of Helsinki and was approved by the ethics committee of Chongqing Medical University. All animal work was reviewed and approved by the Institutional Animal Care and Usage Committee of the Children’s Hospital of Chongqing Medical University.
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Y., Chen, X., Yang, Q. et al. E1021K Homozygous Mutation in PIK3CD Leads to Activated PI3K-Delta Syndrome 1. J Clin Immunol 40, 378–387 (2020). https://doi.org/10.1007/s10875-020-00749-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-020-00749-y