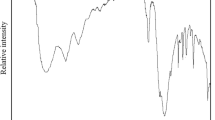
The crystal structure of manganese sulfate templated by 1,4-diaza-bicyclo[2.2.2]octane (abbreviated dabco), (C6H14N2)[Mn(H2O)6](SO4)2, was investigated using single crystal X-ray diffraction data. It crystallises in the monoclinic system (space group P21/c) with the following unit-cell parameters: a = 12.1392(2) Å, b = 12.3117(2) Å, c = 12.2765(2) Å, β = 104.607(1)°, V = 1775.47(5) Å3 and Z = 4. The structure has been solved using direct methods and refined by least-squares analysis [R 1 = 0.0381, wR 2 = 0.1082]. The crystal structure of the title compound is built from isolated [Mn(H2O)6]2+ octahedral cations, 1,4-diaza-bicyclo[2.2.2]octandiium cations (C6H14N2)2+ and sulfate anions (SO4)2− connected by a three-dimensional hydrogen-bond network. The thermal decomposition of the precursor, studied by thermogravimetry and temperature-dependent X-ray powder diffraction, proceeds through four stages giving rise to the mixture of Mn2O3and Mn3O4.






Similar content being viewed by others
References
Cheetham, A.K.; Férey, G.; Loiseau, T. Angew. Chem., Int. Ed. 1999, 38, 3269.
Gier, T.E.; Stucky, G.D. Nature 1991, 349, 508; Férey, G. J. Fluorine Chem. 1995, 72, 187; Khan, M.I.; Meyer, L.M.; Haushalter, R.C.; Schweitzer, A.L.; Zubieta, J.; Dye, J.L. Chem. Mater. 1996, 8, 43.
Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry and Use; Wiley and Sons: London, 1974; Clearfield A. Chem. Rev. 1988, 88, 125; Venuto, P.B. Microporous Mater. 1994, 2, 297.
Khan, M.I.; Cevik, S.; Doedens, R.J. Inorg. Chim. Acta 1999, 292, 112.
Choudhury, A., Krishnamoorthy, J.; Rao, C.N.R. Chem. Commun. 2001, 2610.
Bull, I.; Wheatley, P.S.; Lightfoot, P.; Morris, R.E.; Sastre, E.; Wright, P.A. Chem. Commun. 2002, 1180.
Rao, C.N.R.; Behera, J.N.; Dan, M. Chem. Soc. Rev. 2006, 35, 375.
Bataille, T.; Louër, D. J. Mater. Chem. 2002, 12, 3487.
Bataille, T.; Louër, D. J. Solid State Chem. 2004, 177, 1235.
Thomas, P.M.; Norquist, A.J.; Doran, M.B.; O’Hare, D. J. Mater. Chem. 2003, 13, 88.
Norquist, A.J.; Thomas, P.M.; Doran, M.B.; O’Hare, D. Chem. Mater. 2002, 14, 5179.
Doran, M.B.; Norquist, A.J.; O’Hare, D. Chem. Commun. 2002, 2946.
Norquist, A.J.; Doran, M.B.; Thomas, P.M.; O’Hare, D. Dalton Trans. 2003, 1168.
Bataille, T. Acta Crystallogr. 2003, C59, m459.
Pan, J.-X.; Yang, G.-Y.; Sun, Y.-Q. Acta Crystallogr. 2003, E59, m286.
Fleck, M.; Bohaty, L.; Tillmanns, E. Solid State Sci. 2004, 6, 469.
Rekik, W.; Naïli, H.; Mhiri, T.; Bataille, T. Acta Crystallogr. 2005, E61, m629.
Rekik, W.; Naïli, H.; Bataille, T.; Roisnel, T.; Mhiri, T. Inorg. Chim. Acta 2006, 359, 3954.
Rekik, W.; Naïli, H.; Bataille, T.; Mhiri, T. J. Organomet. Chem. 2006, 691, 4725.
Naïli, H.; Rekik, W.; Bataille, T.; Mhiri, T. Polyhedron 2006, in press.
Zhao, Y.-J.; Li, X.-H.; Wang, S. Acta Crystallogr. 2005, E61, m671.
Held, P. Acta Crystallogr. 2003, E59, m197.
Nonius. Kappa CCD Program Software, Nonius BV, Delft, The Netherlands, 1998.
Otwinowski, Z.; Minor, W.; Carter, C.W.; Sweet, R.M. (Eds.). Methods in Enzymology; Academic Press: New York, 1997; vol. 276, p. 307.
De Meulenaer, J.; Tompa, H. Acta Crystallogr. 1965, 19, 1014.
Farrugia, L.J. J. Appl. Crystallogr. 1999, 32, 837.
Sheldrick, G.M. SHELXS-97, Program for Crystal Structure Solution; University of Göttingen: Germany, 1997.
Sheldrick, G.M. SHELXL-97, Program for Crystal Structure Refinement; University of Göttingen: Germany, 1997.
Paul, G.; Choudhury, A.; Rao, C.N.R. Chem. Mater. 2003, 15, 1174.
Brown, I.D. J. Appl. Crystallogr. 1996, 29, 479.
Jayaraman, K.; Choudhury, A.; Rao, C.N.R. Solid State Sci. 2002, 4, 413.
Paul, G.; Choudhury, A.; Nagarajan, R.; Rao, C.N.R. Inorg. Chem. 2003, 42, 2004.
Zaki, M.I.; Hasan, M.A.; Pasupulety, L.; Kumari, K. Thermochim. Acta 1997, 303, 171.
Acknowledgment
Grateful thanks are expressed to Dr. T. Roisnel (Centre de Diffractométrie X, UMR CNRS 6226) and Mr. G. Marsolier for their assistance in single-crystal and powder X-ray diffraction data collection, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Material CCDC 620298 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Rights and permissions
About this article
Cite this article
Rekik, W., Naïli, H., Mhiri, T. et al. A new dabco templated metal sulfate, (C6H14N2)[Mn (H2O)6](SO4)2. Chemical preparation, hydrogen-bonded structure and thermal decomposition. J Chem Crystallogr 37, 147–155 (2007). https://doi.org/10.1007/s10870-006-9170-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-006-9170-9