Abstract
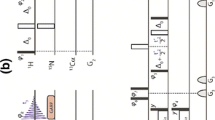
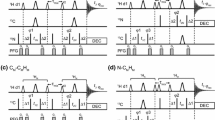
Two novel HSQC-IPAP approaches are proposed to achieve α/β spin-state editing simultaneously for 13C and 15N in a single NMR experiment. The pulse schemes are based on a time-shared (TS) 2D 1H,13C/1H,15N-HSQC correlation experiment that combines concatenated echo elements for simultaneous J(CH) and J(NH) coupling constants evolution, TS evolution of 13C and 15N chemical shifts in the indirect dimension and heteronuclear α/β-spin-state selection by means of the IPAP principle. Heteronuclear α/β-editing for all CH n (n = 1–3) and NH n (1–2) multiplicities can be achieved in the detected F2 dimension of a single TS-HSQC-F2-IPAP experiment. On the other hand, an alternative TS-HSQC-F1-IPAP experiment is also proposed to achieve α/β-editing in the indirect F1 dimension. Experimental and simulated data is provided to evaluate these principles in terms of sensitivity and performance simultaneously on backbone and side-chain CH, CH2, CH3, NH, and NH2 spin systems in uniformly 13C/15N-labeled proteins and in small natural-abundance peptides.
Similar content being viewed by others
References
Andersson P, Weigelt J, Otting G (1998) Spin-state selection filters for the measurement of heteronuclear one-bond coupling constants. J Biomol NMR 12:435–441
Boelens R, Burgering M, Fogh RH and Kaptein R (1994) J Biomol NMR 4:201–213
Carlomagno T, Peti W, Griesinger C (2000) A new method for the simultaneous measurement of magnitude and sign of 1DCH and 1DHH dipolar couplings in methylene groups. J Biomol NMR 17:99–109
Ding K, Gronenborn AM (2003) Sensitivity-enhanced 2D IPAP, TROSY/anti-TROSY, and E.COSY experiments: Alternatives for measuring dipolar 15N1HN couplings. J Magn Reson 163:208–214
Farmer II BT (1991) Simultaneous [13C,15N]-HMQC, A pseudo-triple-resonance Experiment. J Magn Reson 93:635–641
Fehér K, Berger S, Kövér KE (2003) Accurate determination of small one-bond heteronuclear residual dipolar couplings by F1 coupled HSQC modified with a G-BIRD(r) module. J Magn Reson 163:340–346
Frueh DP, Arthanari H, Wagner G (2005) Unambiguous Assignment of NMR Protein Backbone Signals with a Time-shared Triple-resonance Experiment. J Biomol NMR 33:187–196
Giesen AW, Bae LC, Barrett CL, Chyba JA, Chaykovsky MM, Cheng M-C, Murray JH, Oliver EJ, Sullivan SM, Brown JM, Dahlquist FW, Homans SW (2001) Measurement of one-bond 1Hα-13Cα couplings in backbone-labelled proteins. J Biomol NMR 19:255–260
Hall JB, Dayie KT, Fushman D (2003) Direct measurement of the 15N CSA/dipolar relaxation interference from coupled HSQC spectra. J Biomol NMR 26:181–186
Hall JB, Fushman D (2003) Direct measurement of the transverse and longitudinal 15N chemical shift anisotropy-dipolar cross-correlation rate constants using 1H-coupled HSQC spectra. Magn Reson Chem 41:837–842
Ishii Y, Markus MA, Tycko R (2001) Controlling residual bipolar couplings in high-resolution NMR of proteins by strain induced alignment in a gel. J Biomol NMR 21:141–151
Kay LE, Keifer P, Saarinen T (1992) Pure absorption gradient-enhanced heteronuclear single-quantum correlation spectroscopy with improved sensitivity. J Am Chem Soc 114:10663–10665
Kim S, Szyperski T (2003) GFT NMR, A new approach to rapidly obtain precise high-dimensional NMR spectral information. J Am Chem Soc 125:1385–1393
Kontaxis G, Bax A (2001) Multiplet component separation for measurement of methyl 13C-1H dipolar couplings in weakly aligned proteins. J Biomol NMR 20:77–82
Kövér KE, Batta G (2004) More line narrowing in TROSY by decoupling of long-range couplings: shift correlation and 1JNC coupling constant measurements. J Magn Reson 170:184–190
Mariani M, Tessari M, Boelens R, Vis H, Kaptein R (1994) Assignment of the Protein Backbone from a Single 3D, N-15, C-13, Time-Shared HxyH Experiment. J Magn Reson Series B 104:294–297
Meissner A, Duus JO, Sørensen OW (1997) Integration of spin-state-selective excitation into 2D NMR correlation experiments with heteronuclear ZQ/DQ π rotations for 1J(XH)-resolved E.COSY-type measurement of heteronuclear coupling constants in proteins. J Biomol NMR 10:89–94
Miclet E, O’Neil-Cabello E, Nikonowicz EP, Live D, Bax A (2003) 1H-1H dipolar couplings provide a unique probe of RNA backbone structure. J Am Chem Soc 125:15740–15741
Miclet E, Williams Jr DC, Clore GM, Bryce DL, Boisbouvier J, Bax A (2004) Relaxation-optimized NMR spectroscopy of methylene groups in proteins and nucleic acids. J Am Chem Soc 126:10560–10570
Nolis P, Espinosa JF, Parella T (2006) Optimum spin-state selection for all multiplicities in the acquisition dimension of the HSQC experiment. J Magn Reson 180:39–50
Ottiger M, Delaglio F, Bax A (1998) Measurement of J and dipolar couplings from simplified two-dimensional NMR spectra. J Magn Reson 131:373–378
Parella T, Gairí M (2004) Simultaneous recording of spin-state-selective NMR spectra for different InS spin systems. J Am Chem Soc 126:9821–9826
Permi P (2002) A spin-state-selective experiment for measuring heteronuclear one-bond and homonuclear two-bond couplings from an HSQC-type spectrum. J Biomol NMR 22:27–35
Sattler M, Maurer M, Schleucher J, Griesinger C (1995) A simultaneous 15N,1H and 13C,1H-HSQC with sensitivity enhancement and a heteronuclear echo. J Biomol NMR 5:97–102
Schleucher J, Schwendinger M, Sattler M, Schmidt P, Schedletzky O, Glaser SJ, Sørensen OW, Griesinger C (1994) A general enhancement scheme in heteronuclear multidimensional NMR employing pulsed field gradients. J Biomol NMR 4:301–306
Sørensen MD, Meissner A, Sørensen OW (1997) Spin-state-selective coherence transfer via intermediate states of two-spin coherence in IS spin systems. Application to E.COSY-type measurement of J coupling constants. J Biomol NMR 10:181–186
Sørensen MD, Meissner A, Sørensen OW (1999) 13C natural abundance S3E and S3CT experiments for measurement of J coupling constants between 13Cα or 1Hα and other protons in a protein. J Magn Reson 137:237–242
Sørensen OW (1990) Aspects and prospects of multidimensional time-domain spectroscopy. J Magn Reson 89:210–216
Szyperski T, Yeh DC, Sukumaran DK, Moseley HN, Montelione GT (2002) Reduced Dimensionality NMR spectroscopy for high-throughput protein resonance assignment. Proc Natl Acad Sci USA 99:8009–8014
Uhrin D, Bramham J, Winder SJ, Barlow PN (2000) Simultaneous CT-13C and VT-15N chemical shift labelling. Application to 3D NOESY-CH3NH and 3D 13C,15N HSQC-NOESY-CH3NH. J Biomol NMR 18:253–259
Acknowledgments
This work was supported by MCYT (project BQU2003-01677). The authors are grateful to the Servei de Ressonància Magnètica Nuclear, UAB, for allocating instrument time to this project and to Dr. W. Bermel for providing us a TS pulse program code. P.N thanks “Generalitat de Catalunya” for a predoctoral grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nolis, P., Parella, T. Simultaneous α/β spin-state selection for 13C and 15N from a time-shared HSQC-IPAP experiment. J Biomol NMR 37, 65–77 (2007). https://doi.org/10.1007/s10858-006-9104-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10858-006-9104-z