Abstract
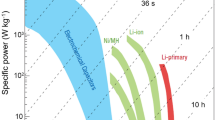
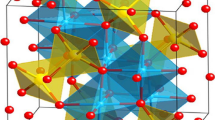
Hybrid supercapacitors are successfully introduced to reduce the gap between high-capacity battery electrodes and high-power capacitor electrodes in case of electrochemical energy storage devices. Niobium pentoxide (Nb2O5) has attracted great interest for hybrid supercapacitors because of its moderate capacity and excellent cycle performance. However, its low electronic conductivity is still a major problem. Carbon is usually incorporated to address this limitation. Here, we report the Nb2O5 nanoparticle networks to facilitate electronic transport via continuous connection of materials. Additionally, the high surface area of the nanoparticles is maintained. The Nb2O5 nanoparticle network was synthesized using a simple solvothermal reaction in organic media. The materials characterization was performed using X-ray diffraction analysis, and scanning and transmission electron microscopies. The charge storage mechanism of the synthesized Nb2O5 material was investigated by cyclic voltammetry. In galvanostatic charge–discharge tests, the synthesized Nb2O5 nanoparticle network electrode exhibited stable cycle performance and remarkable rate capability without carbon incorporation.





Similar content being viewed by others
References
Winter M, Brodd RJ (2004) What are batteries, fuel cells, and supercapacitors? Chem Rev 104:4245–4270
Amatucci GG, Badway F, Du Pasquier A, Zheng T (2001) An asymmetric hybrid nonaqueous energy storage cell. J Electrochem Soc 148:A930–A939
Du Pasquier A, Plitz I, Menocal S, Amatucci G (2003) A comparative study of Li-ion battery, supercapacitor and nonaqueous asymmetric hybrid devices for automotive applications. J Power Sources 115:171–178
Cheng L, Liu H-J, Zhang J-J, Xiong H-M, Xia Y-Y (2006) Nanosized Li4Ti5O12 prepared by molten salt method as an electrode material for hybrid electrochemical supercapacitors. J Electrochem Soc 153:A1472–A1477
Naoi K, Naoi W, Aoyagi S, J-i Miyamoto, Kamino T (2013) New generation “nanohybrid supercapacitor”. Acc Chem Res 46:1075–1083
Kim J-H, Kim J-S, Lim Y-G, Lee J-G, Kim Y-J (2011) Effect of carbon types on the electrochemical properties of negative electrodes for Li-ion capacitors. J Power Sources 196:10490–10495
Kim J, Young C, Lee J et al (2017) Nanoarchitecture of MOF-derived nanoporous functional composites for hybrid supercapacitors. J Mater Chem A 5:15065–15072
Brezesinski K, Wang J, Haetge J et al (2010) Pseudocapacitive contributions to charge storage in highly ordered mesoporous group V transition metal oxides with iso-oriented layered nanocrystalline domains. J Am Chem Soc 132:6982–6990
Augustyn V, Come J, Lowe MA et al (2013) High-rate electrochemical energy storage through Li+ intercalation pseudocapacitance. Nat Mater 12:518–522
Lubimtsev AA, Kent PRC, Sumpter BG, Ganesh P (2013) Understanding the origin of high-rate intercalation pseudocapacitance in Nb2O5 crystals. J Mater Chem A 1:14951–14956
Viet AL, Reddy MV, Jose R, Chowdari BVR, Ramakrishna S (2010) Nanostructured Nb2O5 polymorphs by electrospinning for rechargeable lithium batteries. J Phys Chem C 114:664–671
Wen H, Liu Z, Wang J, Yang Q, Li Y, Yu J (2011) Facile synthesis of Nb2O5 nanorod array films and their electrochemical properties. Appl Surf Sci 257:10084–10088
Wang X, Li G, Chen Z et al (2011) High-performance supercapacitors based on nanocomposites of Nb2O5 nanocrystals and carbon nanotubes. Adv Energy Mater 1:1089–1093
Kong L, Zhang C, Zhang S et al (2014) High-power and high-energy asymmetric supercapacitors based on Li+-intercalation into a T–Nb2O5/graphene pseudocapacitive electrode. J Mater Chem A 2:17962–17970
Kong L, Zhang C, Wang J, Qiao W, Ling L, Long D (2015) Free-standing T–Nb2O5/graphene composite papers with ultrahigh gravimetric/volumetric capacitance for Li-ion intercalation pseudocapacitor. ACS Nano 9:11200–11208
Lim E, Kim H, Jo C et al (2014) Advanced hybrid supercapacitor based on a mesoporous niobium pentoxide/carbon as high-performance anode. ACS Nano 8:8968–8978
Lim E, Jo C, Kim H et al (2015) Facile synthesis of Nb2O5@carbon core–shell nanocrystals with controlled crystalline structure for high-power anodes in hybrid supercapacitors. ACS Nano 9:7497–7505
Shi C, Xiang K, Zhu Y, Chen X, Zhou W, Chen H (2017) Nb2O5 nanospheres/surface-modified graphene composites as superior anode materials in lithium ion batteries. Ceram Int 43:6232–6238
Kang SH, Park C-M, Lee J, Kim J-H (2015) Electrochemical lithium storage kinetics of self-organized nanochannel niobium oxide electrodes. J Electroanal Chem 746:45–50
Kim K, Kim M-S, Cha P-R, Kang SH, Kim J-H (2016) Structural modification of self-organized nanoporous niobium oxide via hydrogen treatment. Chem Mater 28:1453–1461
Yan L, Rui X, Chen G, Xu W, Zou G, Luo H (2016) Recent advances in nanostructured Nb-based oxides for electrochemical energy storage. Nanoscale 8:8443–8465
Zhai J, Wu Y, Zhao X, Yang Q (2017) Facile preparation of flower-like hierarchical Nb2O5 microspheres self-assembled by nanorod for high-power anodes in advanced hybrid supercapacitor. J Alloy Compd 715:275–283
Kong L, Cao X, Wang J, Qiao W, Ling L, Long D (2016) Revisiting Li+ intercalation into various crystalline phases of Nb2O5 anchored on graphene sheets as pseudocapacitive electrodes. J Power Sources 309:42–49
Kim K, Woo S-G, Jo YN, Lee J, Kim J-H (2017) Niobium oxide nanoparticle core–amorphous carbon shell structure for fast reversible lithium storage. Electrochim Acta 240:316–322
Zhang S, Wu J, Wang J, Qiao W, Long D, Ling L (2018) Constructing T–Nb2O5@Carbon hollow core-shell nanostructures for high-rate hybrid supercapacitor. J Power Sources 396:88–94
Zhang J, Chen H, Sun X et al (2017) High intercalation pseudocapacitance of free-standing T–Nb2O5 nanowires@carbon cloth hybrid supercapacitor electrodes. J Electrochem Soc 164:A820–A825
Qinglin D, Mengjiao L, Junyong W, Kai J, Zhigao H, Junhao C (2018) Free-anchored Nb2O5@graphene networks for ultrafast-stable lithium storage. Nanotechnology 29:185401
Aegerter MA (2001) Sol–gel niobium pentoxide: a promising material for electrochromic coatings, batteries, nanocrystalline solar cells and catalysis. Sol Energy Mater Sol Cells 68:401–422
Parker AJ (1969) Protic-dipolar aprotic solvent effects on rates of bimolecular reactions. Chem Rev 69:1–32
Liang X, Wang X, Zhuang J, Chen Y, Wang D, Li Y (2006) Synthesis of nearly monodisperse iron oxide and oxyhydroxide nanocrystals. Adv Funct Mater 16:1805–1813
Duan X, Mei L, Ma J, Li Q, Wang T, Zheng W (2012) Facet-induced formation of hematite mesocrystals with improved lithium storage properties. Chem Commun 48:12204–12206
Pawar RC, Um JH, Kang S, Yoon W-S, Choe H, Lee CS (2017) Solvent-polarity-induced hematite (α-Fe2O3) nanostructures for lithium-ion battery and photoelectrochemical applications. Electrochim Acta 245:643–653
Lindström H, Södergren S, Solbrand A et al (1997) Li+ ion insertion in TiO2 (anatase). 2. Voltammetry on nanoporous films. J Phys Chem B 101:7717–7722
Wang J, Polleux J, Lim J, Dunn B (2007) Pseudocapacitive contributions to electrochemical energy storage in TiO2 (anatase) nanoparticles. J Phys Chem C 111:14925–14931
Zhu K, Wang Q, Kim J-H, Pesaran AA, Frank AJ (2012) Pseudocapacitive lithium-ion storage in oriented anatase TiO2 nanotube arrays. J Phys Chem C 116:11895–11899
Kim J-H, Zhu K, Kim JY, Frank AJ (2013) Tailoring oriented TiO2 nanotube morphology for improved Li storage kinetics. Electrochim Acta 88:123–128
Abdur R, Kim K, Kim J-H, Lee J (2015) Electrochemical behavior of manganese oxides on flexible substrates for thin film supercapacitors. Electrochim Acta 153:184–189
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) Grant funded by the Korean Government (2017H1A2A1043359, 2015R1A5A7037615, 2016M3C1B5906958, and 2016R1C1B1014015). This work was also supported by the National Research Council of Science and Technology of Korea (R&D Convergence Program, CAP-16-08-KITECH).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, K., Hwang, J., Seo, H. et al. Surface-controlled Nb2O5 nanoparticle networks for fast Li transport and storage. J Mater Sci 54, 2493–2500 (2019). https://doi.org/10.1007/s10853-018-3010-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-018-3010-0