Abstract
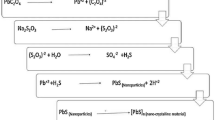
We have synthesized lead and lead sulfide nanoparticles embedded in a natural zeolite (clinoptilolite) matrix by a simple hydrothermal process. The process steps involve the partial removing of the natural cations in clinoptilolite, the ion-exchange process to enclose Pb ions and nanoparticles and finally a sulfuration process at different temperatures to obtain lead sulfide phases in the zeolite matrix. The samples were studied by X-ray diffraction, diffuse reflectance spectroscopy, energy dispersive X-ray spectroscopy, X-ray photon spectroscopy and transmission electron microscopy. The experimental results show the inclusion of three Pb species with different valence states after the Pb ion-exchange step, namely Pb2+, Pb4+, and Pb0. At the end of the process, two simultaneous lead sulfide crystalline phases, PbS (Galena) and PbS2 (tetragonal) were synthesized in the clinoptilolite matrix. The optical absorption spectra of the samples show the exciton absorption peaks typical of colloidal PbS nanoparticles. The average size of the PbS nanoparticles was about 10 nm and their crystalline structure was determined from diffraction electron patterns. The high-pressure phase PbS2 was also identified and its formation was attributed to the influence of the special conditions of clinoptilolite matrix as crystallization media to induce some selective nucleation process of this crystalline phase.








Similar content being viewed by others
References
Faghihian H, Ghannadi Marageh M, Kazemian H (1999) Appl Radiat Isot 50:660
Mondale KD, Carland RM, Aplan FF (1995) Min Eng 8:535
Cerjan Stefanovic S, Zabukovec Logar N, Margeta K, Novak Tusar N, Arcon I, Maver K, Kovac J, Kaucic V (2007) Microporous Mesoporous Mater 105:251
Dávila-Jiménez MM, Elizalde-González MP, Mattusch J, Morgenstern P, Pérez-Cruz MA, Reyes-Ortega Y, Wennrich R, Yee-Madeira H (2008) J Colloid Interface Sci 322:527
Erdem E, Karanipar N, Donat R (2004) J Colloid Interface Sci 280:309
Wang Y, Herron N (1987) J Phys Chem 91:257
Chen W, Wang ZG, Lin ZJ, Lin LY (1996) Solid State Commun 101:371
Herron N (1995) J Incl Phenom Macrocycl Chem 21:283
Suzuki S, Ryo M, Yamamoto T, Sakata T, Yanagida S, Wada Y (2007) J Mater Sci 42:5995. doi:10.1007/s10853-006-1127-z
Kadlecikova M, Breza J, Jesena K, Pastorkova K, Luptakova V, Kolmacka M, Vojackova A, Michalka M, Vavra I, Krizanova Z (2008) Appl Surf Sci 254:5073
Li F, Suna S, Jiang Y, Xia M, Suna M, Xue B (2008) J Hazard Mater 152:1037
Sohrabnezhad S, Pourahmad A, Sadjadi MS, Zanjanchi MA (2008) Mater Sci Eng C 28:202
Sadjadi MS, Pourahmad A, Sohrabnezhad S, Zare K (2007) Mater Lett 61:2923
Iacomi F, Vasile A, Polychroniadis EK (2003) Mater Sci Eng B 101:275
Martín-Palma RJ, Hernández-Vélez M, Díaz I, Villavicencio-García H, García-Poza MM, Martínez-Duart JM, Pérez-Pariente J (2001) Mater Sci Eng C 15:163
Sotelo-Lerma M, Quevedo-López MA, Orozco-Terán RA, Ramírez-Bon R, Espinoza-Beltrán FJ (1998) J Phys Chem Solids 59:145
Ochoa-Landín R, Flores-Acosta M, Ramírez-Bon R, Arizpe-Chávez H, Sotelo-Lerma M, Castillón-Barraza FF (2003) J Phys Chem Solids 64:2245
Flores-Acosta M, Sotelo-Lerma M, Arizpe-Chávez H, Castillón-Barraza FF, Ramírez-Bon R (2003) Solid State Commun 136:567
Xie R, Li D, Yang D, Jiang M (2007) J Mater Sci 42:1376. doi:10.1007/s10853-006-1211-4
Wise FW (2000) Acc Chem Res 33:773
Efros AIL, Efros AL (1982) Fiz Tekh Poluprov 16:1209
Espiau R, Bernas H (2005) J Appl Phys 98:104310
Wang Y, Suna A, Mahler W, Kasowski R (1987) J Chem Phys 87:7315
Asunskis DJ, Boloti IL, Hanley L (2008) J Phys Chem C 112:9555
Zhang Y, Xu Z (2008) Appl Phys Lett 93:083106
Kraus TD, Wise FW (1997) Phys Rev B 55:9860
Zhao X-S, Xu S-Y, Liang L-Y, Li T, Cauchi S (2007) J Mater Sci 42:4265. doi:10.1007/s10853-006-0679-2
Chen W, Wang Z, Lin Z, Qian J, Lin L (1990) Appl Phys Lett 68:91998
Leiggener C, Calzaferri G (2005) Chem A Eur J 11:7191
Kostic R, Romcevic M, Romcevic N, Klopotowski L, Kossut J, Kuljanin J, Comor MI, Nedeljkovic JM (2008) Opt Mater 30:1177
Rodríguez-Iznaga I, Gómez A, Rodríguez-Fuentes G, Benítez-Aguilar A, Serrano-Bayan J (2002) Microporous Mesoporous Mater 53:71
Perraki T, Orfanoudaki A (2004) Appl Clay Sci 25:9
Elaiopoulos K, Perraki Th, Grigoropoulou E (2008) Microporous Mesoporous Mater 112:441
Lee Y, Carr SW, Parised J (1998) Chem Mater 10:2561
Fuggle JC, Martensson N (1980) J Electron Spectrosc Relat Phenom 21:275
Liu Z, Guo B, Tay SW, Hong L, Zhang X (2008) J Power Sources 184:16
Ganguly P, Hedge MS (1988) Phys Rev B 37:5107
Soto G, Díaz JA, Machorro R, Reyes-Serrato A, de la Cruz W (2002) Mater Lett 52:29
Henglein A (1999) J Phys Chem B 103:9302
Henglein A, Holzwarth A, Mulvaney P (1992) J Phys Chem 96:8700
Mulvaney P (1996) Langmuir 12:788
Fernee MJ, Watt A, Warner J, Heckenberg N, Rubinsztein-Dunlop H (2004) Nanotech 15:1328
Fernee MJ, Watt A, Warner J, Cooper S, Heckenberg N, Rubinsztein-Dunlop H (2003) Nanotech 14:991
Cademartiri L, Montanari E, Calestani G, Migliori A, Guagliardi A, Ozin GA (2006) J Am Chem Soc 128:10337
Peterson JJ, Krauss TD (2006) Phys Chem Chem Phys 8:3851
Song Li L, Qu L, Wang L, Lu R, Peng X, Zhao Y, Li T (1997) Langmuir 13:6183
Flores-Acosta M, Pérez-Salas R, Sotelo-Lerma M, Castillón-Barraza FF, Ramírez-Bon R (2005) Adv Tech Mater Mater Process J 7:101
Pitcher MW, Cates E, Raboin L, Bianconi PA (2000) Chem Mater 12:1738
Wheeler KT, Walker D, Johnson MC (2007) Am J Sci 307:590
Acknowledgements
We thank the technical assistance of Jesús Antonio Díaz, J. E. Urbina, M. A. Hernández L. and E. Larios. F:F. Castillón-Barraza also acknowledges to CONACyT and DGAPA-UNAM for research grants 50547 and IN110208-3, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Román-Zamorano, J.F., Flores-Acosta, M., Arizpe-Chávez, H. et al. Structure and properties of lead and lead sulfide nanoparticles in natural zeolite. J Mater Sci 44, 4781–4788 (2009). https://doi.org/10.1007/s10853-009-3720-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-009-3720-4