Abstract
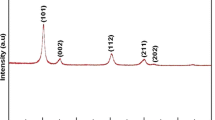
The new phosphates ZnxMg0.5+xE2−x(PO4)3 (E = Ti, Zr) were synthesized by the sol–gel method followed by heat treatment. Obtained samples were characterized with X-ray, IR, DTA and microprobe electron analyses. The ZnxMg0.5+xTi2−x(PO4)3 solid solution (0 ≤ x ≤ 0.5, kosnarite-type, sp. gr. R\(\bar{3}\)) is stable up to 1323 K. The ZnxMg0.5+xZr2−x(PO4)3 solid solution (0 ≤ x ≤ 0.5, Sc2(WO4)3-type, sp. gr. P21/n) is thermally unstable above 1273 K. The thermal expansion of ZnxMg0.5+xE2−x(PO4)3 solid solutions was studied in the temperature range 298–1073 K. The magnitudes of average volume thermal expansion coefficients vary from 1 × 10−5 to 5 × 10−6 K−1. The composition regulation of these medium- and high-expanding materials allows you to change the characteristics of thermal expansion in the required direction.
Graphic abstract











Similar content being viewed by others
References
Naqash S, Tietz F, Guillon O. Synthesis and characterization of equimolar Al/Y-substituted NASICON solid solution Na1+2x+yAlxYxZr2−2xSiyP3−yO12. Solid State Ion. 2018;319:13–21. https://doi.org/10.1016/j.ssi.2018.01.048.
Pet’kov VI. Complex phosphates formed by metal cations in oxidation states I and IV. Russ Chem Rev. 2012;81:606–37. https://doi.org/10.1070/RC2012v081n07ABEH004243.
Brownfield ME, Foord EE, Sutley SJ, Botinelly T. Kosnarite, KZr2(PO4)3 a new mineral from Mount Mica and Black Mountain, Oxford County, Maine. Am Mineral. 1993;78:653–6.
Wang Y, Zhou Y, Song Y, Yang L, Liu F. Mechanical and thermal expansion studies on Ca0.5Sr0.5Zr4-xTixP6O24 ceramics. Ceram Int. 2018;44:16698–702. https://doi.org/10.1016/j.ceramint.2018.06.097.
Ivanov-Shits AK, Murin IV. Ionica solid. V. 1. St. Petersburg: St Petersburg State University; 2001. 616 p. (in Russian)
Lu X, Wang S, Xiao R, Shi S, Li H, Chen L. First-principles insight into the structural fundamental of super ionic conducting in NASICON MTi2(PO4)3 (M = Li, Na) materials for rechargeable batteries. Nano Energy. 2017;41:626–33. https://doi.org/10.1016/j.nanoen.2017.09.044.
Pet’kov VI, Orlova AI, Kazantsev GN, Samoilov SG, Spiridonova ML. Thermal expansion in the Zr and 1-, 2-valent complex phosphates of NaZr2(PO4)3 (NZP) structure. J Therm Anal Cal. 2001;66:623–32. https://doi.org/10.1023/A:1013145807987.
Asabina EA, Orekhova NV, Ermilova MM, Pet’kov VI, Glukhova IO, Zhilyaeva NA, Yaroslavtsev AB. Synthesis and catalytic properties of M05(1 + x)FexTi2 − x(PO4)3 (M = Co, Ni, Cu; 0 ≤ x ≤ 2) for methanol conversion reactions. Inorg Mater. 2015;51:793–8. https://doi.org/10.1134/S002016851508004X.
Ilin AB, Ermilova MM, Orekhova NV, Cretin M, Yaroslavtsev AB. Conversion of aliphatic C1–C2 alcohols on In-, Nb-, Mo-doped complex lithium phosphates and HZr2(PO4)3 with NASICON-type structure. J Alloys Compd. 2018;748:583–90. https://doi.org/10.1016/j.jallcom.2018.03.099.
Pet’kov VI, Lavrenov DA, Sukhanov MV, Kovalsky AM, Borovikova EY. Sol-gel synthesis and structural manganese-zirconium (titanium) phosphates. Russ J Inorg Chem. 2019;64:170–8. https://doi.org/10.1134/S0036023619020165.
Pet’kov VI, Orlova AI, Sukhanov MV, Zharinova MV, Kurazhkovskaya VS. Synthesis and characterization of M1/3Nb5/3(PO4)3 (M = Mg, Mn Co, Ni, Cu or Zn) compounds with the sodium zirconium phosphate framework structure. J Mater Sci Lett. 2002;21:513–6. https://doi.org/10.1023/A:1015355228098.
Jaeger C, Barth S, Feltz A, Scheler G. Studies of the structure of mixed crystals of the system Nal+2xMgxZr2-x(PO4)3 by SIP-MAS –NMR. Physica Stat Solidi (a). 1987;102:791–802. https://doi.org/10.1002/pssa.2211020242.
Feltz A, Barth S. Preparation and conductivity behaviour of Na3MIIZr(PO4)3, (MII:Mn, Mg, Zn). Solid State Ion. 1983;9–10:817–21. https://doi.org/10.1016/0167-2738(83)90094-2.
Asabina E, Pet’kov V, Mayorov P, Lavrenov D, Schelokov I, Kovalsky A. Synthesis, structure and thermal expansion of the phosphates M0.5+xM′xZr2−x(PO4)3 (M, M′–metals in oxidation state +2). Pure Appl Chem. 2017;89:523–34. https://doi.org/10.1515/pac-2016-1005.
Pyatenko Yu A, Voronkov AA, Pudovkina ZV (1976) Mineralogical crystal chemistry of titanium. M.: Nauka. 155 p. (in Russian).
Rietveld HM. Line profiles of neutron powder-diffraction peaks for structure refinement. Acta Crystallogr. 1967;22:151. https://doi.org/10.1107/S0365110X67000234.
Kim YI, Izumi F. Structure refinements with a new version of the Rietveld-refinement program RIETAN. J Ceram Soc Jpn. 1994;102:401–4. https://doi.org/10.2109/jcersj.102.401.
Izumi F. Rietveld analysis programs Rietan and Premos and special applications. In: Young RA, editor. The Rietveld method. Oxford: Oxford University Press; 1993. p. 236–53.
Pet’kov VI, Dmitrienko AS, Sukhanov MV, Kovalskii AM. New approach of synthesis of phosphate–sulfates with NZP-type structure. J Therm Anal Calorim. 2017;127:2093–9. https://doi.org/10.1007/s10973-016-5724-z.
Pet’kov VI, Dmitrienko AS, Bokov AI. Thermal expansion of phosphate–sulfates of eulytite structure. J Therm Anal Calorim. 2018;133:199–205. https://doi.org/10.1007/s10973-017-6676-7.
Sadovnikov SI, Gusev AI. Thermal expansion, heat capacity and phase transformations in nanocrystalline and coarse-crystalline silver sulfide at 290–970 K. J Therm Anal Calorim. 2018;131:1155–64. https://doi.org/10.1007/s10973-017-6691-8.
Pet’kov VI, Shipilov AS, Dmitrienko AS, Alekseev AA. Characterization and controlling thermal expansion of materials with kosnarite- and langbeinite-type structures. J Ind Eng Chem. 2018;57:236–43. https://doi.org/10.1016/j.jiec.2017.08.029.
Acknowledgements
The present work was performed at the Lobachevsky State University of Nizhni Novgorod with the financial support of the Russian Foundation for Basic Research (Project No. 18-29-12063).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pet’kov, V.I., Lavrenov, D.A. & Kovalsky, A.M. Synthesis, characterization and thermal expansion of the zinc-containing phosphates with the mineral-like framework structures. J Therm Anal Calorim 139, 1791–1798 (2020). https://doi.org/10.1007/s10973-019-08624-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-08624-8