Abstract
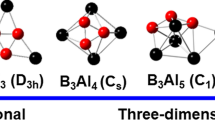
Geometrical and electronic properties of Asn (n = 2–15) neutrals, cations and anions have been investigated using the density functional method of B3LYP. Berny structural optimization and frequency analyses are performed with the basis of 6–311 + G(d) for both neutrals and charged ions. The total energies of these clusters are then used to study the evolution of their binding energy, relative stability, and electronic properties as a function of size. The geometries are found to undergo a structural change from two dimensional to three-dimensional when the cluster contains four atoms. The geometrical changes are companied by corresponding changes in the nearest-neighbor distances and coordination numbers. In the whole size range, both ionization potential and electron affinity have the tendency of decrease when the number of As units in the cluster increases. The stability of clusters exhibits strong even-odd alternations with several magic numbers. The neutral Asn clusters are found to be even-numbered with local maxima at n = 2 and 4, while the cationic and anionic clusters are preferentially odd-numbered with As +3 , As +5 , and As −5 being the most stable ions according to the calculated results of the both energy gain and electronic properties.






Similar content being viewed by others
References
Ballone P, Jones RO (1994) J Chem Phys 100:4941
Shen M, Schaefer III (1994) J Chem Phys 101:2261
Yoo RK, Ruscic B, Berkowitz J (1992) J Chem Phys 96:6696
Lippa TP, Xu SJ, Lyapustina SA (1998) J Chem Phys 109:10727
Chi X, Tian S, Xu K (2002) Chin J Chem Phys 15:22
Jones RO, Ganteor G, Hunsicker S, Pieperhoff P (1995) J Chem Phys 103:9549
Polak ML, Joe Ho, Gustav G, Lineberger WC (1992) J Chem Phys 97:8990
Zhang H, Balasubramanian K (1992) J Chem Phys 97:3437
Igel-Mann G, Stoll H, Preuss H (1993) Mol Phys 80:325
Haser M, Schneide U, Ahlrichs R (1992) J Am Chem Soc 114:9551
Scherer OJ (1990) Angew Chem Int Ed Engl 102:1137
Dimaio AJ, Rheingold AL (1990) Chem Rev 90:169
Voako SH, Wilk L, Nusair M (1980) Can J Phys 58:1200
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Beck AD (1993) J Chem Phys 98:5468
Knight WD, Clemenger K, Saunders WA, Chou MY, Cohen ML (1984) Phys Rev Lett 52:2141
Rao BK, Jena P (1985) Phys Rev B 32:2058
Bhasker ND, Frueholz RP, Klimeak CM, Cook RA (1987) Phys Rev B 36:4418
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghava-chari K, Foresman JB, Cioslowski J, Ortiz JV, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (1998) computer code GAUSSIAN98, revision A.6. Gaussian, Inc., Pittsburgh, PA
Zhao J, Zhou X, Chen X, Wang J, Jellinek J (2006) Phys Rev B 73:115418
Bosworth YM, Clark JH, Rippon DM (1973) J Mol Spectrosc 46:240
Huber KP, Herzberg G (1979) Molecular spectra and molecular structure IV constants of diatomic molecules. Van Nostrand Reinhold, New York
Yi J (2000) Chem Phys Lett 325:269
Lide DR (ed) (2000). In: Chemical rubber company handbook of chemistry and physics, 81st edn. CRC Press, Boca Raton, Florida, USA
Schiferl D, Barrett CS (1969) J Appl Crystallogr 2:30
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 20603021), Youth Foundation of Shanxi (2007021009) and the Youth Academic Leader of Shanxi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, L. Evolution of the electronic structure and properties of neutral and charged arsenic clusters. J Mater Sci 42, 9154–9162 (2007). https://doi.org/10.1007/s10853-007-1928-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-007-1928-8