Abstract
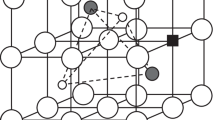
We present an analytical model for the ionic conductivity of a strongly acceptor doped, fluorite-type oxygen ion conductor, (A1 − x B B x B )O2 − x B/2, i.e. a concentrated solution of AO2 and B2O3, which can be applied, e.g., to yttria doped zirconia (YSZ). The model considers nearest neighbor interactions between oxygen vacancies and dopant cations, which may be negligible, attractive or repulsive. The vacancies are distributed to the tetrahedra formed by the cations using quasi-chemical reactions for the exchange between the different sites. The resulting vacancy distribution is used in a simplified model for the oxygen ion conductivity which considers jump rates between different oxygen sites that depend on their local neighborhood and the nature of the cation-cation edge which has to be crossed during a jump between edge-sharing tetrahedra. Among the various possibilities, only attractive dopant-vacancy interaction together with reduced jump rates through A-B and B-B edges (compared to A-A edges) can explain satisfactorily the experimental findings, i.e. the maximum of the conductivity at dopant fractions x B ≈ 0.15, the slight decrease of the activation energy with increasing temperature and the increase of activation energy with dopant fraction.
Similar content being viewed by others
References
A. Hammou and J. Guindet, in The CRC Handbook of Solid State Electrochemistry, edited by P.J. Gellings and H.J.M. Bouwmeester (CRC Press, Boca Raton, 1996), p. 407.
S.P.S. Badwal, Solid State Ionics, 52, 23 (1992).
P.S. Manning, J.D. Sirman, R.A. De Souza, and J.A. Kilner, Solid State Ionics, 100, 107 (1997).
D.W. Stickler and W.G. Carlson, J. Am. Ceram. Soc., 47, 122 (1964).
H. Inaba and H. Takawa, Solid State Ionics, 83, 1 (1996).
H. Schmalzried, Z. Phys. Chem. NF, 105, 47 (1977).
M.S. Khan, M.S. Islam, and D.R. Bates, J. Mater. Chem., 8, 2229 (1998).
M.O. Zacate, L. Minervini, D.J. Bradfield, R.W. Grimes, and K.E. Sickafus, Solid State Ionics, 128, 243 (2000).
F. Shimojo, T. Okabe, F. Tachibana, M. Kobayashi, and H. Okazaki, J. Phys. Soc. Japan, 61, 2842 (1992).
F. Shimojo and H. Okazaki, J. Phys. Soc. Japan, 61, 4106 (1992).
M. Meyer and N. Nicoloso, Ber. Bunsenges. Phys. Chem., 101, 1393 (1997).
A.D. Murray, G.E. Murch, and C.R.A. Catlow, Solid State Ionics, 19, 196 (1986).
R. Krishnamurthy, Y.-G. Yoon, D.J. Srolovitz, and R. Carr, J. Am. Cer. Soc., 87, 1821 (2004).
S.P.S. Badwal, F.T. Ciacchi, S. Rajendran, and J. Drennan, Solid State Ionics, 109, 167 (1998).
R.E.W. Casselton, Phys. Status Solidi A, 2, 571 (1970).
M. Martin, Z. Phys. Chem., 219, 105 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martin, M. On the ionic conductivity of strongly acceptor doped, fluorite-type oxygen ion conductors. J Electroceram 17, 765–773 (2006). https://doi.org/10.1007/s10832-006-6007-z
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10832-006-6007-z