Abstract
Purpose
The aim was to study the association between embryonal mitochondrial DNA (mtDNA) content and embryo quality and implantation outcomes.
Methods
A retrospective chart review was performed with data collected from a private IVF center database. The study population included female infertility patients with ages ranging from 31 to 38 years old, and the main outcome measures were embryo quality and transfer outcomes.
Results
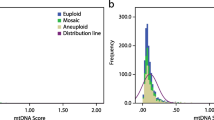
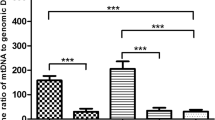
From a total of 1510 blastocyst biopsies, the majority of embryos consisted of grade 1 (High), followed by grade 2 (mid), and grade 3 (poor). Embryos with higher mtDNA content were found to be of poorer quality (grade 3) relative to grades 1 and 2 (P = 0.003). Using a logistic model, mtDNA best predicted lowest and highest grades, but not mid-grade embryos. There was no correlation between mtDNA content and the subjects’ age (R2 = 0.0018). In an analysis of only euploid embryos (N = 717), there was no longer an association between mtDNA content and embryo quality (P = 0.834). There was no difference in mtDNA content between groups of embryos that did and did not implant (P = 0.53). There was also no association noted between mtDNA content and ongoing pregnancy. Compared to day 6, day 5 blastocysts contain significantly higher amounts of mtDNA (P = 0.0005), lower rates of aneuploidy (P < 0.001), and were more likely to be high-quality blastocysts (grade 1) (P < 0.001).
Conclusion
Although the mtDNA content shows some association to the morphologic grade of an embryo, this association does not persist in an analysis of only euploid embryos. Mitochondrial DNA content also does not appear to be associated with implantation or ongoing pregnancy. Day 5 blastocysts have significantly higher mtDNA content compared to day 6 blastocysts.
Similar content being viewed by others
References:
Thoma ME, McLain AC, Louis JF, King RB, Trumble AC, Sundaram R, et al. Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil Steril. 2013;99(5):1324–31.
Menken J, Trussell J, Larsen U. Age and infertility. Science. 1986;233:1389–9415.
Gleicher N, Weghofer A & Barah DH 2011. Defining ovarian reserve to better understand ovarian aging. Reproductive Biology and Endocrinology.
Babayev E, et al. Reproductive aging is associated with changes in oocyte mitochondrial dynamics, function, and mtDNA quantity. Maturitas. 2016;14:121–30.
Cree LM, Hammond ER, Shelling AN, Berg MC, Peek JC, Green MP. Maternal age and ovarian stimulation independently affect oocyte mtDNA copy number and cumulus cell gene expression in bovine clones. Hum Reprod. 2015;30(6):1410–20. https://doi.org/10.1093/humrep/dev066.
Anderson S, Bankier AT, Barrell BG, de Bruijn MH, Coulson AR, Drouin J, et al. Sequence and organization of the human mitochondrial genome. Nature. 1981;290:457–65.
Leese HJ. Metabolism of the preimplantation embryo: 40 years on. Reproduction. 2012;143:417–27.
Cao L, Shitara H, Horii T, Nagao Y, Imai H, Abe K, et al. The mitochondrial bottleneck occurs without reduction of mtDNA content in female mouse germ cells. Nat Genet. 2007;39:386–90.
Cree LM, Samuels DC, de Sousa Lopes SC, Rajasimha HK, Wonnapinij P, Mann JR, et al. A reduction of mitochondrial DNA molecules during embryogenesis explains the rapid segregation of genotypes. Nat Genet. 2008;40:249–54.
Chan CC, Liu VW, Lau EY, Yeung WS, Ng EH, Ho PC. Mitochondrial DNA content and 4977 bp deletion in unfertilized oocytes. Mol Hum Reprod. 2005;11:843–6.
Seifer DB, DeJesus V, Hubbard K. Mitochondrial deletions in luteinized granulosa cells as a function of age in women under- going in vitro fertilization. Fertil Steril. 2002;78(5):1046–8.
Keefe DL, Niven-Fairchild T, Powell S, Buradagunta S. Mitochondrial deoxyribonucleic acid deletions in oocytes and reproductive aging in women. Fertil Steril. 1995;64(3):577–83.
Desquiret-Dumas V, Clement A, Seegers V, et al. The mitochondrial DNA content of cumulus granulosa cells in linked to embryo quality. Hum Reprod. 2017;32(3):607–14.
Santos TA, El Shourbagy S, St John JC. Mitochondrial content reflects oocyte variability and fertilization outcome. Fertil Steril. 2006;85:584–91.
Reynier P, May-Panloup P, Chrétien MF, Morgan CJ, Jean M, Savagner F, et al. Mitochondrial DNA content affects the fertilizability of human oocytes. Mol Hum Reprod. 2001;7(5):425–9.
Murakoshi Y, Sueoka K, Takahashi K, Sato S, Sakurai T, Tajima H, et al. Embryo developmental capability and pregnancy outcome are related to the mitochondrial DNA copy number and ooplasmic volume. J Assist Reprod Genet. 2013;30:1367–75.
Fragouli E, Spath K, Alfarawati S, Kaper F, Craig A, Michel CE, et al. Altered levels of mitochondrial DNA are associated with female age, aneuploidy, and provide an independent measure of embryonic implantation potential. PLoS Genet. 2015;11:e1005241.
Diez-Juan A, Rubio C, Marin C, Martinez S, Al-Asmar N, Riboldi M, et al. Mitochondrial DNA content as a viability score in human euploid embryos: less is better. Fertil Steril. 2015;104:534–41. e531
Victor AR, Brake AJ, Tyndall JC, Griffin DK, Zouves CG, Barnes FL, et al. Accurate quantitation of mitochondrial DNA reveals uniform levels in human blastocysts irrespective of ploidy, age, or implantation potential. Fertil Steril. 2016;107:34–42.
Treff NR, Zhan Y, Tao X, OlchaM HM, Rajchel J, et al. Levels of trophectoderm mitochondrial DNA do not predict the reproductive potential of sibling embryos. Hum Reprod. 2017;32(4):954–62.
Bancsi LF, Broekmans FJ, Eijkemans MJ, de Jong FH, Habbema JD, te Velde ER. Predictors of poor ovarian response in in vitro fertilization: a prospective study comparing basal markers of ovarian reserve. Fertil Steril. 2002;77:328–36.
Müller-Höcker J, Schäfer S, Weis S, Münscher C, Strowitzki T. Morphological-cytochemical and molecular genetic analyses of mitochondria in isolated human oocytes in the reproductive age. Mol Hum Reprod. 1996, 2(12):951–8.
Kushnir VA, Ludaway T, Russ RB, Fields EJ, Koczor C, Lewis W. Reproductive aging is associated with decreased mitochondrial abundance and altered structure in murine oocytes. J Assist Reprod Genet. 2012;29(7):637–42.
Boucret L, Chao de la Barca JM, Moriniere C, Desquiret V, Ferre-L’Hotellier V, Descamps P, et al. Relationship between diminished ovarian reserve and mitochondrial biogenesis in cumulus cells. Hum Reprod. 2015;30:1653–64.
Duran HE, Simsek-Duran F, Oehninger SC, Jones HW Jr, Castora FJ. The association of reproductive senescence with mitochondrial quantity, function, and DNA integrity in human oocytes at different stages of maturation. Fertil Steril. 2011;96:384–8.
Alexeyev M, Shokolenko I, Wilson G, et al. The maintenance of mitochondrail DNA integrity- critical analysis and update. Cold Spring Harb Perspect Biol. 2013;5:a012641.
Piko L, Taylor KD. Amounts of mitochondrial DNA and abundance of some mitochondrial gene transcripts in early mouse embryos. Dev Biol. 1987;123:364–74.
Piko L, Matsumoto L. Number of mitochondria and some properties of mitochondrial DNA in the mouse egg. Dev Biol. 1976;49:1–10.
Ebert KM, Liem H, Hecht NB. Mitochondrial DNA in the mouse preimplantation embryo. J Reprod Fertil. 1988;82:145–9.
Larsson J, Want H, Wilhemsson A, Oldfors P, Rustin M, et al. Mitochondrial transcription factor A is necessary for mtDNA maintenance and embryogenesis in mice. Nat Genet. 1998;18:231–6.
Barnett DK, Kimura J, Bavister BD. Translocation of active mitochondria during hamster preimplantation embryo development studied by confocal laser scanning microscopy. Dev Dyn. 1996;205(1):64–72.
Van Blerkom J. Mitochondria as regulatory forces in oocytes, preimplantation embryos and stemcells. Reprod BioMed Online. 2008;16(4):553–69.
DiMauro S, Schon EA. Mitochondrial respiratory-chain diseases. N Engl J Med. 2003;348:2656–68.
Monnot S, Samuels DC, Hesters L, Frydman N, Gigarel N, Burlet P, et al. Mutation dependance of the mitochondrial DNA copy number in the first stages of human embryogenesis. Hum Mol Genet. 2013;22:1867–72.
Sobenin IA, Mitrofanov KY, Zhelankin AV, et al. Quantitative assessment of heteroplasmy of mitochondrial genome: perspectives in diagnostics and methodological pitfalls. Biomed Res Int. 2014;2014(292017):9. https://doi.org/10.1155/2014/292017.
van den Ouweland JMW, Lemkes HHPJ, Ruitenbeek W, et al. Mutation in mitochondrial tRMALeu(UUR) gene in a large pedigree with maternally transmitted type II diabetes mellitus and deafness. Nat Genet. 1992;1(5):368–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
For this type of study, formal consent is not required.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Accepted as poster presentations at the ASRM 2017 Meeting in San Antonio, Texas, and PCRS 2018 Meeting in Indian Wells, California.
Rights and permissions
About this article
Cite this article
Klimczak, A.M., Pacheco, L.E., Lewis, K.E. et al. Embryonal mitochondrial DNA: relationship to embryo quality and transfer outcomes. J Assist Reprod Genet 35, 871–877 (2018). https://doi.org/10.1007/s10815-018-1147-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-018-1147-z