Abstract
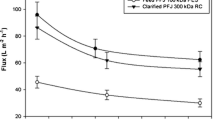
A two-stage ultrafiltration process was applied to the aqueous phase of Tetraselmis suecica after breaking its cell wall by high-pressure homogenization. Microscopic observation revealed that the cells were completely disrupted from 600 bar and cell fragmentation of the cells was also noticeable after 800 bar. In addition, the highest concentration of all the molecules of interest in the aqueous phase was observed at 1,000 bar and a temperature of 46 °C while preserving the integrity of the molecules of interest in the downstream process. After centrifugation, the aqueous phase was submitted to ultrafiltration through two consecutive membranes of different molecular weight cutoffs. Complete retention of starch was possible with a 100-kDa membrane and separation of sugars from proteins with a 10-kDa membrane on the remaining mixture. After testing the process with model solutions, the transmembrane pressure selected was 2.07 bar, which succeeded in retaining starch and pigments during the first part of the process, and proteins during the second part. A linear correlation between the permeate flux rate and the pressure was observed in both parts of the process.






Similar content being viewed by others
References
Araujo GS, Matos LJ, Fernandes JO, Cartaxo SJ, Goncalves LR, Fernandes FA, Farias WR (2013) Extraction of lipids from microalgae by ultrasound application: prospection of the optimal extraction method. Ultrason Sonochem 20:95–98
Barbarino E, Lourenço SO (2005) An evaluation of methods for extraction and quantification of protein from marine macro- and microalgae. J Appl Phycol 17:447–460
Brown MR (1991) The amino-acid and sugar composition of 16 species of microalgae used in mariculture. J Exp Mar Biol Ecol 145:79–99
Converti A, Casazza AA, Ortiz EY, Perego P, Del Borghi M (2009) Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem Eng Process 48:1146–1151
D'Souza FML, Kelly GJ (2000) Effects of a diet of a nitrogen-limited alga (Tetraselmis suecica) on growth, survival and biochemical composition of tiger prawn (Penaeus semisulcatus) larvae. Aquaculture 181:311–329
Dunstan GA, Volkman JK, Jeffrey SW, Barrett SM (1992) Biochemical composition of microalgae from the green algal classes Chlorophyceae and Prasinophyceae. 2. Lipid classes and fatty acids. J Exp Mar Biol Ecol 161:115–134
Eteshola E, Gottlieb M, Arad S (1996) Dilute solution viscosity of red microalga exopolysaccharide. Chem Eng Sci 51:1487–1494
Fabregas J, Herrero C, Cabezas B, Abalde J (1985) Mass culture and biochemical variability of the marine microalga Tetraselmis suecica Kylin (Butch) with high nutrient concentrations. Aquaculture 49:231–244
Frappart M, Massé A, Jaffrin MY, Pruvost J, Jaouen P (2011) Influence of hydrodynamics in tangential and dynamic ultrafiltration systems for microalgae separation. Desalination 265:279–283
Gloaguen V, Ruiz G, Morvan H, Mouradi-Givernaud A, Maes E, Krausz P, Strecker G (2004) The extracellular polysaccharide of Porphyridium sp.: an NMR study of lithium-resistant oligosaccharidic fragments. Carbohydr Res 339:97–103
Halim R, Danquah MK, Webley PA (2012) Extraction of oil from microalgae for biodiesel production: a review. Biotechnol Adv 30:709–732
Kim J-D, Lee W-S, Kim B, Lee C-G (2006) Proteomic analysis of proteins expression patterns associated with astaxanthin accumulation by green alga Haematococcus pluvialis (Chlorophyceae) under high light stress. J Microbiol Biotechnol 16:1222–1228
Li Y, Horsman M, Wu N, Lan CQ, Dubois-Calero N (2008) Biofuels from microalgae. Biotechnol Prog 24:815–820
Lourenço SO, Barbarino E, Marquez UML, Aidar E (1998) Distribution of intracellular nitrogen in marine microalgae: basis for the calculation of specific nitrogen-to-proteins conversion factors. J Phycol 34:798–811
Morineau-Thomas O, Jaouen P, Legentilhomme P (2002) The role of exopolysaccharides in fouling phenomenon during ultrafiltration of microalgae (Chlorella sp. and Porphyridium purpureum): advantage of a swirling decaying permeate flux. Bioprocess Biosyst Eng 25:35–42
Morris HJ, Carrillo OV, Almarales Á, Bermúdez RC, Alonso ME, Borges L, Quintana MM, Fontaine R, Llauradó G, Hernández M (2009) Proteins hydrolysates from the alga Chlorella vulgaris 87/1 with potentialities in immunonutrition. Biotechnol Appl 26:162–165
Olmstead IL, Hill DR, Dias DA, Jayasinghe NS, Callahan DL, Kentish SE, Scales PJ, Martin GJ (2013) A quantitative analysis of microalgal lipids for optimization of biodiesel and omega-3 production. Biotechnol Bioeng 110:2096–2104
Patel AK, Laroche C, Marcati A, Ursu AV, Jubeau S, Marchal L, Petit E, Djelveh G, Michaud P (2013) Separation and fractionation of exopolysaccharides from Porphyridium cruentum. Bioresour Technol 145:345–350
Pugh N, Ross SA, ElSohly HN, ElSohly MA, Pasco DS (2001) Isolation of three high molecular weight polysaccharide preparations with potent immunostimulatory activity from Spirulina platensis, Aphanizomenon flos-aquae and Chlorella pyrenoidosa. Planta Med 67:737–742
Renaud SM, Thinh L-V, Parry DL (1999) The gross chemical composition and fatty acid composition of 18 species of tropical Australian microalgae for possible use in mariculture. Aquaculture 170:147–159
Ritchie RJ (2006) Consistent sets of spectrophotometric chlorophyll equations for acetone, methanol and ethanol solvents. Photosynth Res 89:27–41
Safi C, Charton M, Pignolet O, Silvestre F, Vaca-Garcia C, Pontalier P-Y (2013) Influence of microalgae cell wall characteristics on proteins extractability and determination of nitrogen-to-proteins conversion factors. J Appl Phycol 25:523–529
Schwenzfeier A, Wierenga PA, Gruppen H (2011) Isolation and characterization of soluble proteins from the green microalgae Tetraselmis sp. Bioresour Technol 102:9121–9127
Shi Y, Sheng J, Yang F, Hu Q (2007) Purification and identification of polysaccharide derived from Chlorella pyrenoidosa. Food Chem 103:101–105
Singh A, Olsen SI (2011) A critical review of biochemical conversion, sustainability and life cycle assessment of algal biofuels. Appl Energy 88:3548–3555
Susanto H, Arafat H, Janssen EML, Ulbricht M (2008) Ultrafiltration of polysaccharide–proteins mixtures: elucidation of fouling mechanisms and fouling control by membrane surface modification. Sep Purif Technol 63:558–565
Volkman JK, Jeffrey SW, Nichols PD, Rogers GI, Garland CD (1989) Fatty acid and lipid composition of 10 species of microalgae used in mariculture. J Exp Mar Biol Ecol 128:219–240
Wang X, Kolattukudy PE (1996) Isolation of a proteins containing covalently linked large and small subunits of ribulose-1,5-bisphosphate carboxylase/oxygenase from Botryococcus braunii. Plant Physiol 111:441–445
Whyte JNC (1987) Biochemical composition and energy content of six species of phytoplankton used in mariculture of bivalves. Aquaculture 60:231–241
Widjaja A, Chien C-C, Ju Y-H (2009) Study of increasing lipid production from fresh water microalgae Chlorella vulgaris. J Taiwan Inst Chem Eng 40:13–20
Wijffels RH, Barbosa MJ (2010) An outlook on microalgal biofuels. Science 329(5993):796–799
Williams PJB, Laurens LML (2010) Microalgae as biodiesel & biomass feedstocks: review & analysis of the biochemistry, energetics & economics. Energy Environ Sci 3:554–590
Yang J, Xu M, Zhang X, Hu Q, Sommerfeld M, Chen Y (2011) Life-cycle analysis on biodiesel production from microalgae: water footprint and nutrients balance. Bioresour Technol 102:159–165
Yao C, Ai J, Cao X, Xue S, Zhang W (2012) Enhancing starch production of a marine green microalga Tetraselmis subcordiformis through nutrient limitation. Bioresour Technol 118:438–444
Zhang X, Hu Q, Sommerfeld M, Puruhito E, Chen Y (2010) Harvesting algal biomass for biofuels using ultrafiltration membranes. Bioresour Technol 101:5297–5304
Acknowledgments
This work was supported by the French national research agency (ANR) within the framework of “the Algoraffinerie” project. The authors are grateful to the “Direction de la Recherche de l’INP” for its additional financial support SMI-2013 and are also sincerely thankful to the Department of Chemical and Biomolecular Engineering of The University of Melbourne for providing the facilities and all the necessary tools to conduct this work and produce this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Safi, C., Liu, D.Z., Yap, B.H.J. et al. A two-stage ultrafiltration process for separating multiple components of Tetraselmis suecica after cell disruption. J Appl Phycol 26, 2379–2387 (2014). https://doi.org/10.1007/s10811-014-0271-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-014-0271-0