Abstract
Purpose
To investigate the changes in the choroidal vascularity index (CVI) with age and to compare the effect of the binarised area on CVI in healthy eyes using spectral-domain optical coherence tomography (SD-OCT).
Materials and methods
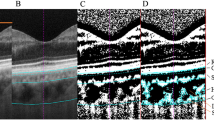
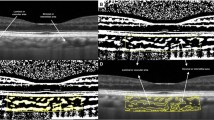
Two hundred and twenty-four eyes of 224 healthy subjects were included in this prospective cross-sectional study. The eyes were divided into different age groups to analyse the possible age-related choroidal structural changes. Subfoveal choroidal thickness (SFCT), CVI, total choroidal area (TCA), stromal area (SA), luminal area (LA), and CVI within the central 1500 µm of the macula were analysed using enhanced depth imaging SD-OCT. The CVI was defined as the proportion of the LA to the TCA, and its values for the two binarised areas were compared (CVItotal vs. CVI1500).
Results
The mean age was 34.77 ± 20.97 (range: 5–70) years. The mean CVItotal was statistically lower (66.71 ± 2.58%) than the mean CVI1500 (67.54 ± 3.13%, p = 0.008) among all the healthy participants. TCA, LA, CVItotal, and CVI1500 were statistically higher in the ≤ 18-year-old group compared to the > 18-year-old group (p < 0.05), but SA was not significantly different between the groups (p = 0.327). Similarly, TCA, LA, CVItotal, and CVI1500 between the five studied age groups were statistically different (p < 0.001), showing larger figures in the 0–10-year-old group. However, this was not true for the stromal region (p = 0.139). CVItotal exhibited a very strong positive correlation with CVI1500. No significant gender-related difference was observed in CVI.
Conclusion
Decreased LA, TCA, and CVI were observed in healthy eyes with increasing age. CVI1500 was higher than CVItotal in a single B scan OCT. This result may provide valuable information about the choroid under different conditions, such as its physiological changes and disease pathophysiology.



Similar content being viewed by others
References
Tan KA, Gupta P, Agarwal A, Chhablani J, Cheng CY, Keane PA, Agrawal R (2016) State of science: choroidal thickness and systemic health. Surv Ophthalmol 61(5):566–581. https://doi.org/10.1016/j.survophthal.2016.02.007
Chung SE, Kang SW, Lee JH, Kim YT (2011) Choroidal thickness in polypoidal choroidal vasculopathy and exudative age-related macular degeneration. Ophthalmology 118(5):840–845. https://doi.org/10.1016/j.ophtha.2010.09.012
Koizumi H, Yamagishi T, Yamazaki T, Kawasaki R, Kinoshita S (2011) Subfoveal choroidal thickness in typical age-related macular degeneration and polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol 249(8):1123–1128. https://doi.org/10.1007/s00417-011-1620-1
Wood A, Binns A, Margrain T, Drexler W, Považay B, Esmaeelpour M, Sheen N (2011) Retinal and choroidal thickness in early age-related macular degeneration. Am J Ophthalmol 152(6):1030-1038.e1032. https://doi.org/10.1016/j.ajo.2011.05.021
Imamura Y, Fujiwara T, Margolis R, Spaide RF (2009) Enhanced depth imaging optical coherence tomography of the choroid in central serous chorioretinopathy. Retina 29(10):1469–1473. https://doi.org/10.1097/IAE.0b013e3181be0a83
Maruko I, Iida T, Sugano Y, Ojima A, Sekiryu T (2011) Subfoveal choroidal thickness in fellow eyes of patients with central serous chorioretinopathy. Retina 31(8):1603–1608. https://doi.org/10.1097/IAE.0b013e31820f4b39
Ueta T, Obata R, Inoue Y, Iriyama A, Takahashi H, Yamaguchi T, Tamaki Y, Yanagi Y (2009) Background comparison of typical age-related macular degeneration and polypoidal choroidal vasculopathy in Japanese patients. Ophthalmology 116(12):2400–2406. https://doi.org/10.1016/j.ophtha.2009.06.013
Stanga PE, Lim JI, Hamilton P (2003) Indocyanine green angiography in chorioretinal diseases indications and interpretation an evidence-based update. Ophthalmology 110(1):15–21. https://doi.org/10.1016/s0161-6420(02)01563-4
Fujiwara T, Imamura Y, Margolis R, Slakter JS, Spaide RF (2009) Enhanced depth imaging optical coherence tomography of the choroid in highly myopic eyes. Am J Ophthalmol 148(3):445–450. https://doi.org/10.1016/j.ajo.2009.04.029
Ikuno Y, Tano Y (2009) Retinal and choroidal biometry in highly myopic eyes with spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci 50(8):3876–3880. https://doi.org/10.1167/iovs.08-3325
Spaide RF (2009) Age-related choroidal atrophy. Am J Ophthalmol 147(5):801–810. https://doi.org/10.1016/j.ajo.2008.12.010
Maruko I, Iida T, Sugano Y, Ojima A, Ogasawara M, Spaide RF (2010) Subfoveal choroidal thickness after treatment of central serous chorioretinopathy. Ophthalmology 117(9):1792–1799. https://doi.org/10.1016/j.ophtha.2010.01.023
Agrawal R, Gupta P, Tan KA, Cheung CM, Wong TY, Cheng CY (2016) Choroidal vascularity index as a measure of vascular status of the choroid: Measurements in healthy eyes from a population-based study. Sci Rep 6:21090. https://doi.org/10.1038/srep21090
Sonoda S, Sakamoto T, Yamashita T, Uchino E, Kawano H, Yoshihara N, Terasaki H, Shirasawa M, Tomita M, Ishibashi T (2015) Luminal and stromal areas of choroid determined by binarization method of optical coherence tomographic images. Am J Ophthalmol 159(6):1123-1131.e1121. https://doi.org/10.1016/j.ajo.2015.03.005
Agrawal R, Chhablani J, Tan KA, Shah S, Sarvaiya C, Banker A (2016) Choroidal vascularity index in central serous chorioretinopathy. Retina 36(9):1646–1651. https://doi.org/10.1097/iae.0000000000001040
Ağın A, Kadayıfçılar S, Sönmez HE, Baytaroğlu A, Demir S, Sağ E, Özen S, Eldem B (2019) Evaluation of choroidal thickness, choroidal vascularity index and peripapillary retinal nerve fiber layer in patients with juvenile systemic lupus erythematosus. Lupus 28(1):44–50. https://doi.org/10.1177/0961203318814196
Wei X, Kumar S, Ding J, Khandelwal N, Agarwal M, Agrawal R (2019) Choroidal structural changes in smokers measured using choroidal vascularity index. Invest Ophthalmol Vis Sci 60(5):1316–1320. https://doi.org/10.1167/iovs.18-25764
Ruiz-Medrano J, Ruiz-Moreno JM, Goud A, Vupparaboina KK, Jana S, Chhablani J (2018) Age-related changes in choroidal vascular density of healthy subjects based on image binarization of swept-source optical coherence tomography. Retina 38(3):508–515
Spaide RF, Koizumi H, Pozzoni MC (2008) Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 146(4):496–500. https://doi.org/10.1016/j.ajo.2008.05.032
Fleiss JL, Cohen J (1973) The equivalence of weighted kappa and the intraclass correlation coefficient as measures of reliability. Educ Psychol Measur 33(3):613–619
Agrawal R, Li LKH, Nakhate V, Khandelwal N, Mahendradas P (2016) Choroidal vascularity index in Vogt-Koyanagi-Harada disease: an EDI-OCT derived tool for monitoring disease progression. Transl vision Sci Technol 5(4):7–7
Agrawal R, Salman M, Tan K-A, Karampelas M, Sim DA, Keane PA, Pavesio C (2016) Choroidal vascularity index (CVI)-a novel optical coherence tomography parameter for monitoring patients with panuveitis? PLoS ONE 11(1):e0146344
Branchini LA, Adhi M, Regatieri CV, Nandakumar N, Liu JJ, Laver N, Fujimoto JG, Duker JS (2013) Analysis of choroidal morphologic features and vasculature in healthy eyes using spectral-domain optical coherence tomography. Ophthalmology 120(9):1901–1908
Sonoda S, Sakamoto T, Yamashita T, Shirasawa M, Uchino E, Terasaki H, Tomita M (2014) Choroidal structure in normal eyes and after photodynamic therapy determined by binarization of optical coherence tomographic images. Invest Ophthalmol Vis Sci 55(6):3893–3899
van der Schaft TL, Mooy CM, de Bruijn WC, Oron FG, Mulder PG, de Jong PT (1992) Histologic features of the early stages of age-related macular degeneration: a statistical analysis. Ophthalmology 99(2):278–286
Sobin SS, Bernick S, Ballard KW (1992) Histochemical characterization of the aging microvasculature in the human and other mammalian and non-mammalian vertebrates by the periodic acid-Schiff reaction. Mech Ageing Dev 63(2):183–192
Toda N (2012) Age-related changes in endothelial function and blood flow regulation. Pharmacol Ther 133(2):159–176
Stenmark KR, Yeager ME, El Kasmi KC, Nozik-Grayck E, Gerasimovskaya EV, Li M, Riddle SR, Frid MG (2013) The adventitia: essential regulator of vascular wall structure and function. Annu Rev Physiol 75:23–47
Friedman E, SMITH TR, KUWABARA T, (1963) Senile choroidal vascular patterns and drusen. Arch Ophthalmol 69(2):220–230
Sohn EH, Khanna A, Tucker BA, Abramoff MD, Stone EM, Mullins RF (2014) Structural and biochemical analyses of choroidal thickness in human donor eyes. Invest Ophthalmol Vis Sci 55(3):1352–1360
Kumar A, Zhao L, Fariss RN, McMenamin PG, Wong WT (2014) Vascular associations and dynamic process motility in perivascular myeloid cells of the mouse choroid: implications for function and senescent change. Invest Ophthalmol Vis Sci 55(3):1787–1796
Singh SR, Invernizzi A, Rasheed MA, Cagini C, Goud A, Vupparaboina KK, Cozzi M, Lupidi M, Chhablani J (2018) Wide-field choroidal vascularity in healthy eyes. Am J Ophthalmol 193:100–105
Agrawal R, Wei X, Goud A, Vupparaboina KK, Jana S, Chhablani J (2017) Influence of scanning area on choroidal vascularity index measurement using optical coherence tomography. Acta Ophthalmol 95(8):e770–e775. https://doi.org/10.1111/aos.13442
Hayreh SS (1990) In vivo choroidal circulation and its watershed zones. Eye 4(2):273–289
Kakiuchi N, Terasaki H, Sonoda S, Shiihara H, Yamashita T, Tomita M, Shinohara Y, Sakoguchi T, Iwata K, Sakamoto T (2019) Regional differences of choroidal structure determined by wide-field optical coherence tomography. Invest Ophthalmol Vis Sci 60(7):2614–2622
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Koçak, N., Subaşı, M. & Yeter, V. Effects of age and binarising area on choroidal vascularity index in healthy eyes: an optical coherence tomography study. Int Ophthalmol 41, 825–834 (2021). https://doi.org/10.1007/s10792-020-01636-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-020-01636-6