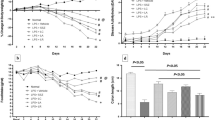
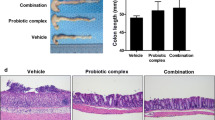
Abstract
Probiotics and their metabolites appear to be a promising approach that targets both the intestinal inflammation and dysbiosis in bowel diseases. In this context, the emergence of the probiotic cell-free supernatant (CFS) has attracted more attention as a safe and targeted alternative therapy with reduced side effects. The use of nonsteroidal anti-inflammatory drugs (NSAIDs) can cause significant intestinal alterations and inflammation, leading to experimental enterocolopathy resembling Crohn disease. Therefore, we investigated the effect of CFS supplementation on the inflammation and the mucosal intestinal alterations induced by NSAIDs, indomethacin. In the current study, a murine model of intestinal inflammation was generated by the oral gavage (o.g) of indomethacin (10 mg/kg) to BALB/C mice. A group of mice treated with indomethacin was concomitantly treated orally by CFS for 5 days. The Body Health Condition index was monitored, and histological scores were evaluated. Moreover, oxidative and pro-inflammatory markers were assessed. Interestingly, we observed that CFS treatment attenuated the severity of the intestinal inflammation in our enterocolopathy model and resulted in the improvement of the clinical symptoms and the histopathological features. Notably, nitric oxide, tumor necrosis factor alpha, malondialdehyde, and myeloperoxidase levels were down-modulated by CFS supplementation. Concomitantly, an attenuation of NF-κB p65, iNOS, COX2 expression in the ileum and the colon was reported. Collectively, our data suggest that CFS treatment has a beneficial effect in experimental enterocolopathy model and could constitute a good therapeutic candidate for alleviating inflammatory responses and to maintain mucosal homeostasis during chronic and severe conditions of intestinal inflammation.







Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Abdelouhab K, Rafa H, Toumi R, Bouaziz S, Medjeber O, Touil-Boukoffa C (2012) Mucosal intestinal alteration in experimental colitis correlates with nitric oxide production by peritoneal macrophages: effect of probiotics and prebiotics. Immunopharmacol Immunotoxicol 34:590–597. https://doi.org/10.3109/08923973.2011.641971
Abenavoli L, Scarpellini E, Colica C, Boccuto L, Salehi B, Sharifi-Rad J, Aiello V, Romano B, De Lorenzo A, Izzo AA, Capasso R (2019) Gut microbiota and obesity: a role for probiotics. Nutrients 11:2690. https://doi.org/10.3390/nu11112690
Abimosleh SM, Tran CD, Howarth GS (2013) Emu oil reduces small intestinal inflammation in the absence of clinical improvement in a rat model of indomethacin-induced enteropathy. Evid Based Complement Altern Med 2013:429706. https://doi.org/10.1155/2013/429706
Anbazhagan AN, Priyamvada S, Alrefai WA, Dudeja PK (2018) Pathophysiology of IBD associated diarrhea. Tissue Barriers 6:e1463897–e1463897. https://doi.org/10.1080/21688370.2018.1463897
Bassaganya-Riera J, Hontecillas R, Horne WT, Sandridge M, Herfarth HH, Bloomfeld R, Isaacs KL (2012) Conjugated linoleic acid modulates immune responses in patients with mild to moderately active Crohn’s disease. Clin Nutr 31:721–727. https://doi.org/10.1016/j.clnu.2012.03.002
Bermudez-Brito M, Muñoz-Quezada S, Gomez-Llorente C, Matencio E, Bernal MJ, Romero F, Gil A (2013) Cell-free culture supernatant of Bifidobacterium breve CNCM I-4035 decreases pro-inflammatory cytokines in human dendritic cells challenged with Salmonella typhi through TLR activation. PLoS ONE 8:e59370–e59370. https://doi.org/10.1371/journal.pone.0059370
Britto SL, Krishna M, Kellermayer R (2019) Weight loss is a sufficient and economical single outcome measure of murine dextran sulfate sodium colitis. FASEB Bioadv 1:493–497. https://doi.org/10.1096/fba.2019-00035
Bronte V, Pittet MJ (2013) The spleen in local and systemic regulation of immunity. Immunity 39:806–818. https://doi.org/10.1016/j.immuni.2013.10.010
Cervantes-Garcia D, Bahena-Delgado A, Jiménez M, Córdova L, Palacios V, Sánchez Alemán E, Martínez-Saldaña M, Salinas E (2020) Glycomacropeptide ameliorates indomethacin-induced enteropathy in rats by modifying intestinal inflammation and oxidative stress. Molecules 25:2351. https://doi.org/10.3390/molecules25102351
Chen Q, Chen T, Li W, Zhang W, Zhu J, Li Y, Huang Y, Shen Y, Yu C (2012) Mycoepoxydiene inhibits lipopolysaccharide-induced inflammatory responses through the of TRAF6 polyubiquitination. PLoS ONE 7:e44890. https://doi.org/10.1371/journal.pone.0044890
Cheng F-S, Pan D, Chang B, Jiang M, Sang L-X (2020) Probiotic mixture VSL#3: an overview of basic and clinical studies in chronic diseases. World J Clin Cases 8:1361–1384. https://doi.org/10.12998/wjcc.v8.i8.1361
Clarke JM, Young GP, Topping DL, Bird AR, Cobiac L, Scherer BL, Winkler JG, Lockett TJ (2011) Butyrate delivered by butyrylated starch increases distal colonic epithelial apoptosis in carcinogen-treated rats. Carcinogenesis 33:197–202. https://doi.org/10.1093/carcin/bgr254
Comalada M, Bailón E, De Haro O, Lara-Villoslada F, Xaus J, Zarzuelo A, Gálvez J (2006) The effects of short-chain fatty acids on colon epithelial proliferation and survival depend on the cellular phenotype. J Cancer Res Clin Oncol 132:487–497. https://doi.org/10.1007/s00432-006-0092-x
Conaghan PG (2012) A turbulent decade for NSAIDs: update on current concepts of classification, epidemiology, comparative efficacy, and toxicity. Rheumatol Int 32:1491–1502. https://doi.org/10.1007/s00296-011-2263-6
Crittenden S, Goepp M, Pollock J, Robb CT, Smyth DJ, Zhou Y, Andrews R, Tyrrell V, Gkikas K, Adima A, O’connor RA, Davies L, Li X-F, Yao HX, Ho G-T, Zheng X, Mair A, Vermeren S, Qian B-Z, Mole DJ, Gerasimidis K, Schwarze JKJ, Breyer RM, Arends MJ, O’donnell VB, Iredale JP, Anderton SM, Narumiya S, Maizels RM, Rossi AG, Howie SE, Yao C (2021) Prostaglandin E(2) promotes intestinal inflammation via inhibiting microbiota-dependent regulatory T cells. Sci Adv 7:eabd7954. https://doi.org/10.1101/2020.07.12.199513
Durazzo A, Nazhand A, Lucarini M, Atanasov AG, Souto EB, Novellino E, Capasso R, Santini A (2020) An updated overview on nanonutraceuticals: focus on nanoprebiotics and nanoprobiotics. Int J Mol Sci 21:2285. https://doi.org/10.3390/ijms21072285
Ewaschuk JB, Diaz H, Meddings L, Diederichs B, Dmytrash A, Backer J, Looijer-Van Langen M, Madsen KL (2008) Secreted bioactive factors from Bifidobacterium infantis enhance epithelial cell barrier function. Am J Physiol Gastrointest Liver Physiol 295:G1025–G1034. https://doi.org/10.1152/ajpgi.90227.2008
Filaretova LP, Bagaeva T, Morozova OY, Zelena D (2011) The healing of NSAID-induced gastric lesions may be followed by small intestinal and cardiovascular side effects. J Physiol Pharmacol 62:619. https://doi.org/10.3390/nu13103499
Ganji-Arjenaki M, Rafieian-Kopaei M (2018) Probiotics are a good choice in remission of inflammatory bowel diseases: a meta analysis and systematic review. J Cell Physiol 233:2091–2103. https://doi.org/10.1002/jcp.25911
Ghouri YA, Richards DM, Rahimi EF, Krill JT, Jelinek KA, Dupont AW (2014) Systematic review of randomized controlled trials of probiotics, prebiotics, and synbiotics in inflammatory bowel disease. Clin Exp Gastroenterol 7:473–487. https://doi.org/10.2147/ceg.s27530
Goldstein JL, Cryer B (2015) Gastrointestinal injury associated with NSAID use: a case study and review of risk factors and preventative strategies. Drug Healthc Patient Saf 7:31–41. https://doi.org/10.2147/DHPS.S71976
Gunaydin C, Bilge SS (2018) Effects of nonsteroidal anti-inflammatory drugs at the molecular level. Euras J Med 50:116–121. https://doi.org/10.5152/eurasianjmed.2018.0010
Gupta P, Andrew H, Kirschner BS, Guandalini S (2000) Is Lactobacillus GG helpful in children with Crohn’s disease? Results of a preliminary, open-label study. J Pediatr Gastroenterol Nutr 31:453–457. https://doi.org/10.1097/00005176-200010000-00024
Hernandez Y, Sotolongo J, Breglio K, Conduah D, Chen A, Xu R, Hsu D, Ungaro R, Hayes LA, Pastorini C, Abreu MT, Fukata M (2010) The role of prostaglandin E2 (PGE 2) in toll-like receptor 4 (TLR4)-mediated colitis-associated neoplasia. BMC Gastroenterol 10:82. https://doi.org/10.1186/1471-230X-10-82
Higashimori A, Watanabe T, Nadatani Y, Takeda S, Otani K, Tanigawa T, Yamagami H, Shiba M, Tominaga K, Fujiwara Y, Arakawa T (2016) Mechanisms of NLRP3 inflammasome activation and its role in NSAID-induced enteropathy. Mucosal Immunol 9:659–668. https://doi.org/10.1038/mi.2015.89
Hulit J, Wang C, Li Z, Albanese C, Rao M, Di Vizio D, Shah S, Byers SW, Mahmood R, Augenlicht LH, Russell R, Pestell RG (2004) Cyclin D1 genetic heterozygosity regulates colonic epithelial cell differentiation and tumor number in ApcMin mice. Mol Cell Biol 24:7598–7611. https://doi.org/10.1128/MCB.24.17.7598-7611.2004
Imaoka H, Ishihara S, Kazumori H, Kadowaki Y, Aziz MM, Rahman FB, Ose T, Fukuhara H, Takasawa S, Kinoshita Y (2010) Exacerbation of indomethacin-induced small intestinal injuries in Reg I-knockout mice. Am J Physiol Gastrointest Liver Physiol 299:311–319. https://doi.org/10.1152/ajpgi.00469.2009
Inan MS, Rasoulpour RJ, Yin L, Hubbard AK, Rosenberg DW, Giardina C (2000) The luminal short-chain fatty acid butyrate modulates NF-kappaB activity in a human colonic epithelial cell line. Gastroenterology 118:724–734. https://doi.org/10.1016/S0016-5085(00)70142-9
Ji J, Shu D, Zheng M, Wang J, Luo C, Wang Y, Guo F, Zou X, Lv X, Li Y, Liu T, Qu H (2016) Microbial metabolite butyrate facilitates M2 macrophage polarization and function. Sci Rep 6:24838. https://doi.org/10.1038/srep24838
Khairy H, Saleh H, Badr AM, Marie MS (2018) Therapeutic efficacy of osthole against dinitrobenzene sulphonic acid induced-colitis in rats. Biomed Pharmacother 100:42–51. https://doi.org/10.1016/j.biopha.2018.01.104
Kim YG, Ohta T, Takahashi T, Kushiro A, Nomoto K, Yokokura T, Okada N, Danbara H (2006) Probiotic Lactobacillus casei activates innate immunity via NF-kappaB and p38 MAP kinase signaling pathways. Microbes Infect 8:994–1005. https://doi.org/10.1016/j.micinf.2005.10.019
Kim MW, Kang J-H, Shin E, Shim K-S, Kim M, Lee C-K, Yoon Y, Oh S (2019) Processed Aloe vera gel attenuates non-steroidal anti-inflammatory drug (NSAID)-induced small intestinal injury by enhancing mucin expression. Food Funct. https://doi.org/10.1039/C9FO01307E
Klampfer L, Huang J, Sasazuki T, Shirasawa S, Augenlicht L (2003) Inhibition of interferon gamma signaling by the short chain fatty acid butyrate. Mol Cancer Res 1:855–862
Kumar M, Nagpal R, Verma V, Kumar A, Kaur N, Hemalatha R, Gautam SK, Singh B (2013) Probiotic metabolites as epigenetic targets in the prevention of colon cancer. Nutr Rev 71:23–34. https://doi.org/10.1111/j.1753-4887.2012.00542.x
Lázár B, László SB, Hutka B, Tóth AS, Mohammadzadeh A, Berekméri E, Ágg B, Balogh M, Sajtos V, Király K, Al-Khrasani M, Földes A, Varga G, Makra N, Ostorházi E, Szabó D, Ligeti B, Kemény Á, Helyes Z, Ferdinandy P, Gyires K, Zádori ZS (2021) A comprehensive time course and correlation analysis of indomethacin-induced inflammation, bile acid alterations and dysbiosis in the rat small intestine. Biochem Pharmacol 190:114590. https://doi.org/10.1016/j.bcp.2021.114590
Luo W, Shen Z, Deng M, Li X, Tan B, Xiao M, Wu S, Yang Z, Zhu C, Tian L, Wu X, Meng X, Quan Y, Wang X (2019) Roseburia intestinalis supernatant ameliorates colitis induced in mice by regulating the immune response. Mol Med Rep. https://doi.org/10.3892/mmr.2019.10327
Martins D, English AM (2014) Catalase activity is stimulated by H(2)O(2) in rich culture medium and is required for H(2)O(2) resistance and adaptation in yeast. Redox Biol 2:308–313. https://doi.org/10.1016/j.redox.2013.12.019
Miura N, Fukutake M, Yamamoto M, Ohtake N, Iizuka S, Imamura S, Tsuchiya N, Ishimatsu M, Nakamura Y, Ishige A, Watanabe K, Kase Y, Takeda S (2007) An herbal medicine orengedokuto prevents indomethacin-induced enteropathy. Biol Pharm Bull 30:495–501. https://doi.org/10.1248/bpb.30.495
Monteros MJM, Galdeano CM, Balcells MF, Weill R, De Paula JA, Perdigón G, Cazorla SI (2021) Probiotic lactobacilli as a promising strategy to ameliorate disorders associated with intestinal inflammation induced by a non-steroidal anti-inflammatory drug. Sci Rep 11:571. https://doi.org/10.1038/s41598-020-80482-z
Moriya K, Miura NN, Adachi Y, Ohno N (2002) Systemic inflammatory response associated with augmentation and activation of leukocytes in Candida/indomethacin administered mice. Biol Pharm Bull 25:816–822. https://doi.org/10.1248/bpb.25.816
Murthy SN, Cooper HS, Shim H, Shah RS, Ibrahim SA, Sedergran DJ (1993) Treatment of dextran sulfate sodium-induced murine colitis by intracolonic cyclosporin. Dig Dis Sci 38:1722–1734. https://doi.org/10.1007/BF01303184
Nandi J, Saud B, Zinkievich JM, Palma DT, Levine RA (2008) 5-Aminosalicylic acid improves indomethacin-induced enteropathy by inhibiting iNOS transcription in rats. Dig Dis Sci 53:123–132. https://doi.org/10.1007/s10620-007-9832-2
Nandi J, Saud B, Zinkievich M, Yang Z-J, Levine R (2009) TNF-α modulates INOS expression in an experimental rat model of indomethacin-induced jejunoileitis. Mol Cell Biochem 336:17–24. https://doi.org/10.1007/s11010-009-0259-2
Natarajan K, Abraham P, Kota R, Isaac B (2018) NF-κB-iNOS-COX2-TNF α inflammatory signaling pathway plays an important role in methotrexate induced small intestinal injury in rats. Food Chem Toxicol 118:766–783. https://doi.org/10.1016/j.fct.2018.06.040
Nikhil K, Keshari R, Bogra J, Kohli M, Abbas H, Malik A, Dikshit M, Barthwal M (2010) Increased myeloperoxidase enzyme activity in plasma is a indicator of inflammation and onset of sepsis. J Crit Care 26:435.e1–7. https://doi.org/10.1016/j.jcrc.2010.09.001
Park JW, Kim HY, Kim MG, Jeong S, Yun CH, Han SH (2019) Short-chain fatty acids inhibit staphylococcal lipoprotein-induced nitric oxide production in murine macrophages. Immune Netw 19:e9. https://doi.org/10.4110/in.2019.19.e9
Peng L, Li ZR, Green RS, Holzman IR, Lin J (2009) Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J Nutr 139:1619–1625. https://doi.org/10.3945/jn.109.104638
Polese B, Thurairajah B, Zhang H, Soo CL, Mcmahon CA, Fontes G, Hussain SNA, Abadie V, King IL (2021) Prostaglandin E2 amplifies IL-17 production by δ T cells during barrier inflammation. Cell Rep. https://doi.org/10.1016/j.celrep.2021.109456
Prisciandaro L, Geier M, Butler R, Cummins A, Howarth G (2009) Probiotics and their derivatives as treatments for inflammatory bowel disease. Inflamm Bowel Dis 15:1906–1914. https://doi.org/10.1002/ibd.20938
Rafa H, Benkhelifa S, Aityounes S, Saoula H, Belhadef S, Belkhelfa M, Boukercha A, Toumi R, Soufli I, Moralès O, De Launoit Y, Mahfouf H, Nakmouche MH, Delhem N, Touil-Boukoffa C (2017) All-Trans retinoic acid modulates TLR4/NF-iκiB signaling pathway targeting TNF-iαi and nitric oxide synthase 2 expression in colonic mucosa during ulcerative colitis and colitis associated cancer. Mediat Inflamm 2017:7353252. https://doi.org/10.1089/jir.2012.0063
Reiff C, Delday M, Rucklidge G, Reid M, Duncan G, Wohlgemuth S, Hörmannsperger G, Loh G, Blaut M, Collie-Duguid E (2009) Balancing inflammatory, lipid, and xenobiotic signaling pathways by VSL# 3, a biotherapeutic agent, in the treatment of inflammatory bowel disease. Inflamm Bowel Dis 15:1721–1736. https://doi.org/10.1002/ibd.20999
Ryma T, Samer A, Soufli I, Rafa H, Touil-Boukoffa C (2021) Role of probiotics and their metabolites in inflammatory bowel diseases (IBDs). Gastroenterol Insights 12:56–66. https://doi.org/10.3390/gastroent12010006
Saez-Lara MJ, Gomez-Llorente C, Plaza-Diaz J, Gil A (2015) The role of probiotic lactic acid bacteria and bifidobacteria in the prevention and treatment of inflammatory bowel disease and other related diseases: a systematic review of randomized human clinical trials. Biomed Res Int 2015:505878. https://doi.org/10.1155/2015/505878
Saitta KS, Zhang C, Lee KK, Fujimoto K, Redinbo MR, Boelsterli UA (2014) Bacterial β-glucuronidase inhibition protects mice against enteropathy induced by indomethacin, ketoprofen or diclofenac: mode of action and pharmacokinetics. Xenobiotica 44:28–35. https://doi.org/10.1016/j.chembiol.2015.08.005
Salvi PS, Cowles RA (2021) Butyrate and the intestinal epithelium: modulation of proliferation and inflammation in homeostasis and disease. Cells 10:1775. https://doi.org/10.3390/cells10071775
Santiago-López L, Hernández-Mendoza A, Vallejo-Cordoba B, Mata-Haro V, Wall-Medrano A, González-Córdova AF (2019) Milk fermented with lactobacillus fermentum ameliorates indomethacin-induced intestinal inflammation: an exploratory study. Nutrients 11:1610. https://doi.org/10.3390/nu11071610
Sartor RB (2005) Probiotic therapy of intestinal inflammation and infections. Curr Opin Gastroenterol 21:44–50
Satoh K (1978) Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta 90:37–43. https://doi.org/10.1016/0009-8981(78)90081-5
Saud B, Nandi J, Ong G, Finocchiaro S, Levine RA (2005) Inhibition of TNF-α improves indomethacin-induced enteropathy in rats by modulating iNOS expression. Dig Dis Sci 50:1677–1683. https://doi.org/10.1007/s10620-005-2914-0
Scarpignato C (2008) NSAID-induced intestinal damage: are luminal bacteria the therapeutic target? Gut 57:145–148. https://doi.org/10.1136/gut.2007.134502
Schwab M, Reynders V, Loitsch S, Steinhilber D, Stein J, Schröder O (2007) Involvement of different nuclear hormone receptors in butyrate-mediated inhibition of inducible NF kappa B signalling. Mol Immunol 44:3625–3632. https://doi.org/10.1016/j.molimm.2007.04.010
Servin AL (2004) Antagonistic activities of lactobacilli and bifidobacteria against microbial pathogens. FEMS Microbiol Rev 28:405–440. https://doi.org/10.1016/j.femsre.2004.01.003
Shu R, Wang C, Meng Q, Liu Z, Wu J, Sun P, Sun H, Ma X, Huo X, Liu K (2019) Resveratrol enhances the protective effects of JBP485 against indomethacin-induced rat intestinal damage in vivo and vitro through up-regulating oligopeptide transporter 1 (Pept1). Biomed Pharmacother 111:251–261. https://doi.org/10.1016/j.biopha.2018.12.084
Smale S, Tibble J, Sigthorsson G, Bjarnason I (2001) Epidemiology and differential diagnosis of NSAID-induced injury to the mucosa of the small intestine. Best Pract Res Clin Gastroenterol 15:723–738. https://doi.org/10.1053/bega.2001.0231
Soufli I, Toumi R, Rafa H, Touil-Boukoffa C (2016) Overview of cytokines and nitric oxide involvement in immuno-pathogenesis of inflammatory bowel diseases. World J Gastrointest Pharmacol Ther 7:353–360. https://doi.org/10.4292/wjgpt.v7.i3.353
Takeuchi K, Amagase K (2018) Roles of cyclooxygenase, prostaglandin E2 and EP receptors in mucosal protection and ulcer healing in the gastrointestinal tract. Curr Pharm Des 24:2002–2011. https://doi.org/10.2174/1381612824666180629111227
Terán-Ventura E, Aguilera M, Vergara P, Martínez V (2014) Specific changes of gut commensal microbiota and TLRs during indomethacin-induced acute intestinal inflammation in rats. J Crohns Colitis 8:1043–1054. https://doi.org/10.1016/j.crohns.2014.02.001
Thong-Ngam D, Choochuai S, Patumraj S, Chayanupatkul M, Klaikeaw N (2012) Curcumin prevents indomethacin-induced gastropathy in rats. World J Gastroenterol 18:1479–1484. https://doi.org/10.3748/wjg.v18.i13.1479
Tian T, Wang Z, Zhang J (2017) Pathomechanisms of oxidative stress in inflammatory bowel disease and potential antioxidant therapies. Oxid Med Cell Longev 2017:4535194. https://doi.org/10.1155/2017/4535194
Tigari P, Janadri S, Madhu K, Taj N (2021) Evaluation of anti-inflammatory effect of wedelolactone on indomethacin induced colitis in rats: involvement of IL-6/STAT3 pathway. Biointerface Res Appl Chem 12:2813–2825. https://doi.org/10.33263/BRIAC123.28132825
Touil-Boukoffa C, Bauvois B, Sancéau J, Hamrioui B, Wietzerbin J (1998) Production of nitric oxide (NO) in human hydatidosis: relationship between nitrite production and interferon-gamma levels. Biochimie 80:739–744. https://doi.org/10.1016/S0300-9084(99)80027-3
Toumi R, Abdelouhab K, Rafa H, Soufli I, Raissi-Kerboua D, Djeraba Z, Touil-Boukoffa C (2013) Beneficial role of the probiotic mixture Ultrabiotique on maintaining the integrity of intestinal mucosal barrier in DSS-induced experimental colitis. Immunopharmacol Immunotoxicol 35:403–409. https://doi.org/10.3109/08923973.2013.790413
Toumi R, Soufli I, Rafa H, Belkhelfa M, Biad A, Touil-Boukoffa C (2014) Probiotic bacteria lactobacillus and bifidobacterium attenuate inflammation in dextran sulfate sodium-induced experimental colitis in mice. Int J Immunopathol Pharmacol 27:615–627. https://doi.org/10.1177/039463201402700418
Vizoso Pinto MG, Rodriguez Gómez M, Seifert S, Watzl B, Holzapfel WH, Franz CM (2009) Lactobacilli stimulate the innate immune response and modulate the TLR expression of HT29 intestinal epithelial cells in vitro. Int J Food Microbiol 133:86–93. https://doi.org/10.1016/j.ijfoodmicro.2009.05.013
Voltan S, Castagliuolo I, Elli M, Longo S, Brun P, D’incà R, Porzionato A, Macchi V, Palù G, Sturniolo GC, Morelli L, Martines D (2007) Aggregating phenotype in Lactobacillus crispatus determines intestinal colonization and TLR2 and TLR4 modulation in murine colonic mucosa. Clin Vaccine Immunol 14:1138–1148. https://doi.org/10.1128/CVI.00079-07
Wu BN, Chen CW, Liou SF, Yeh JL, Chung HH, Chen IJ (2006) Inhibition of proinflammatory tumor necrosis factor-{alpha}-induced inducible nitric-oxide synthase by xanthine-based 7-[2-[4-(2-chlorobenzene)piperazinyl]ethyl]-1,3-dimethylxanthine (KMUP-1) and 7-[2-[4-(4-nitrobenzene)piperazinyl]ethyl]-1, 3-dimethylxanthine (KMUP-3) in rat trachea: The involvement of soluble guanylate cyclase and protein kinase G. Mol Pharmacol 70:977–985. https://doi.org/10.1124/mol.106.024919
Xiao X, Nakatsu G, Jin Y, Wong S, Yu J, Lau JYW (2017) Gut microbiota mediates protection against enteropathy induced by indomethacin. Sci Rep 7:40317. https://doi.org/10.1038/srep40317
Yamada T, Deitch E, Specian RD, Perry MA, Sartor RB, Grisham MB (1993) Mechanisms of acute and chronic intestinal inflammation induced by indomethacin. Inflammation 17:641–662. https://doi.org/10.1007/BF00920471
Yamamoto A, Itoh T, Nasu R, Nishida R (2014) Sodium alginate ameliorates indomethacin-induced gastrointestinal mucosal injury via inhibiting translocation in rats. World J Gastroenterol 20(10):2641–2652. https://doi.org/10.3748/wjg.v20.i10.2641
Yasutake Y, Tomita K, Higashiyama M, Furuhashi H, Shirakabe K, Takajo T, Maruta K, Sato H, Narimatsu K, Yoshikawa K, Okada Y, Kurihara C, Watanabe C, Komoto S, Nagao S, Matsuo H, Miura S, Hokari R (2017) Uric acid ameliorates indomethacin-induced enteropathy in mice through its antioxidant activity. J Gastroenterol Hepatol. https://doi.org/10.1111/jgh.13785
Yeşilyurt N, Yilmaz B, Ağagündüz D, Capasso R (2021) Involvement of probiotics and postbiotics in the immune system modulation. Biologics 1:89–110. https://doi.org/10.3390/biologics1020006
Zakostelska Z, Kverka M, Klimesova K, Rossmann P, Mrazek J, Kopecny J, Hornova M, Srutkova D, Hudcovic T, Ridl J, Tlaskalova-Hogenova H (2011) Lysate of probiotic Lactobacillus casei DN-114 001 ameliorates colitis by strengthening the gut barrier function and changing the gut microenvironment. PLoS ONE 6:e27961. https://doi.org/10.1371/journal.pone.0027961
Zhou L, Zhang M, Wang Y, Dorfman RG, Liu H, Yu T, Chen X, Tang D, Xu L, Yin Y, Pan Y, Zhou Q, Zhou Y, Yu C (2018) Faecalibacterium prausnitzii produces butyrate to maintain Th17/Treg balance and to ameliorate colorectal colitis by inhibiting histone deacetylase 1. Inflamm Bowel Dis 24:1926–1940. https://doi.org/10.1093/ibd/izy182
Acknowledgements
The authors acknowledge the technical staff of Anatomic Pathology service (Beni messous hospital, Algiers, Algeria). They greatly thank the national agency of research development in health (ATRSS) which supported their project. We would like to thank Oussama Medjeber, Sarra Benkhelifa, Asma Belkadi, Kahina Kious and Mourad Hamdaoui for their help and their excellent technical assistance (USTHB, Algiers, Algeria).
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare the absence of competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Samer, A., Toumi, R., Soufli, I. et al. Cell-free probiotic supernatant (CFS) treatment alleviates indomethacin-induced enterocolopathy in BALB/c mice by down-modulating inflammatory response and oxidative stress: potential alternative targeted treatment. Inflammopharmacol 30, 1685–1703 (2022). https://doi.org/10.1007/s10787-022-00996-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-022-00996-y