Abstract
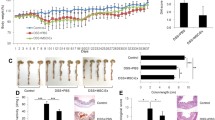
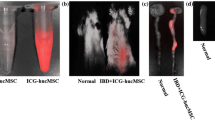
Inflammatory bowel disease (IBD) can be caused by a variety of factors, including hereditary and environmental influences, that lead to dysfunction of the intestinal immune system. Mesenchymal stem cells (MSCs) exhibit important regulatory roles in relieving inflammation and repairing damaged tissues. Although neutrophils are important participants in the development of inflammatory reactions, they are also essential for maintaining intestinal balance during the process of mitigation of IBD by MSCs. Here, we constructed a dextran sulfate sodium (DSS)-induced mouse IBD model and evaluated the effects of treatment with human umbilical cord MSCs. Mouse body weight, faecal traits, colon/spleen gross morphology, tissue histology and immunohistochemical staining, and inflammatory factors were analysed. Magnetic beads were used to sort infiltrating neutrophils from intestinal tissues, and their phenotypes were identified. The neutrophil inflammatory environment was also simulated in vitro, and signalling pathways involved in MSC regulation of neutrophil phenotype were analysed. Human umbilical cord MSCs effectively alleviated DSS-induced weight loss, colon shortening, and intestinal mucosal injury, and reduced clinical disease activity index. The number of neutrophils that infiltrated the intestines of mice treated with human umbilical cord MSCs were decreased and polarised toward the N2 phenotype; at the same time, ERK phosphorylation was inhibited. In vitro experiments showed that addition of the ERK phosphorylation inhibitor, PD98059, down-regulated the expression of N1 neutrophils, while up-regulating that of N2 neutrophils. The colon tissues from patients with IBD were infiltrated with neutrophils. Further, relative to healthy controls, the markers of N1 neutrophils (ICAM-1, FAS, and CCL3) were highly expressed in colon tissues from patients with IBD, whereas the markers of N2 neutrophils (VEGF, CCL2, and CXCR4) were almost undetectable. In conclusion, during alleviation of IBD, human umbilical cord MSCs polarise neutrophils toward the “N2” phenotype by inhibiting activation of ERK signalling.







Similar content being viewed by others
Abbreviations
- CD:
-
Crohn’s disease
- CXCR4:
-
CXC chemokine receptor 4
- DAI:
-
Disease activity index
- DSS:
-
Dextran sodium sulfate
- FAS:
-
Factor-associated suicide
- FCM:
-
Flow cytometry
- HE:
-
Haematoxylin–eosin
- IBD:
-
Inflammatory bowel disease
- ICAM-1:
-
Intercellular cell adhesion molecule-1
- IHC:
-
Immunohistochemistry analysis
- IL:
-
Interleukin
- LPS:
-
Lipopolysaccharide
- MACS:
-
Magnetic-activated cell sorting
- MCP1/CCL2:
-
Low expression of monocyte chemotactic protein 1
- MIP-1α/CCL3:
-
Giant phage cell inflammatory protein-1α
- MSC:
-
Mesenchymal stem cell
- QRT-PCR:
-
Quantitative real-time polymerase chain reaction
- TNF:
-
Tumour necrosis factor
- UC:
-
Ulcerative colitis
- VEGF:
-
Vascular endothelial growth factor
References
Akiyama K, Chen C, Wang D, Xu X, Qu C, Yamaza T, Cai T, Chen W, Sun L, Shi S (2012) Mesenchymal-stem-cell-induced immunoregulation involves FAS-ligand-/FAS-mediated T cell apoptosis. Cell Stem Cell 10(5):544–555
Andrews C, McLean MH, Durum SK (2016) Interleukin-27 as a novel therapy for inflammatory bowel disease: a critical review of the literature. Inflamm Bowel Dis 22(9):2255–2264
Axelrad JE, Lichtiger S, Yajnik V (2016) Inflammatory bowel disease and cancer: the role of inflammation, immunosuppression, and cancer treatment. World J Gastroenterol 22(20):4794–4801
Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, Risso M, Gualandi F, Mancardi GL, Pistoia V, Uccelli A (2006) Human mesenchymal stem cells modulate B-cell functions. Blood 107(1):367–372
Dayan V, Yannarelli G, Billia F, Filomeno P, Wang XH, Davies JE, Keating A (2011) Mesenchymal stromal cells mediate a switch to alternatively activated monocytes/macrophages after acute myocardial infarction. Basic Res Cardiol 106(6):1299–1310
Fridlender ZG, Sun J, Kim S, Kapoor V, Cheng G, Ling L, Worthen GS, Albelda SM (2009) Polarization of tumor-associated neutrophil phenotype by TGF-beta: “N1” versus “N2” TAN. Cancer Cell 16(3):183–194
Fu J, Zhang H, Zhuang Y, Liu H, Shi Q, Li D, Ju X (2014) The role of N-acetyltransferase 8 in mesenchymal stem cell-based therapy for liver ischemia/reperfusion injury in rats. PLoS ONE 9(7):e103355
He H, Zhao ZH, Han FS, Liu XH, Wang R, Zeng YJ (2016) Overexpression of protein kinase C varepsilon improves retention and survival of transplanted mesenchymal stem cells in rat acute myocardial infarction. Cell Death Dis 7:e2056
He R, Li Y, Han C, Lin R, Qian W, Hou X (2019) L-Fucose ameliorates DSS-induced acute colitis via inhibiting macrophage M1 polarization and inhibiting NLRP3 inflammasome and NF-kB activation. Int Immunopharmacol 73:379–388
Jiang D, Qi Y, Walker NG, Sindrilaru A, Hainzl A, Wlaschek M, MacNeil S, Scharffetter-Kochanek K (2013) The effect of adipose tissue derived MSCs delivered by a chemically defined carrier on full-thickness cutaneous wound healing. Biomaterials 34(10):2501–2515
Jiang D, Muschhammer J, Qi Y, Kugler A, de Vries JC, Saffarzadeh M, Sindrilaru A, Beken SV, Wlaschek M, Kluth MA, Ganss C, Frank NY, Frank MH, Preissner KT, Scharffetter-Kochanek K (2016) Suppression of neutrophil-mediated tissue damage—a novel skill of mesenchymal stem cells. Stem Cells 34(9):2393–2406
Kavanagh H, Mahon BP (2011) Allogeneic mesenchymal stem cells prevent allergic airway inflammation by inducing murine regulatory T cells. Allergy 66(4):523–531
Kim KW, Moon SJ, Park MJ, Kim BM, Kim EK, Lee SH, Lee EJ, Chung BH, Yang CW, Cho ML (2016) Erratum to: Optimization of adipose tissue-derived mesenchymal stem cells by rapamycin in a murine model of acute graft-versus-host disease. Stem Cell Res Ther 7(1):80
Lin Y, Lin L, Wang Q, Jin Y, Zhang Y, Cao Y, Zheng C (2015) Transplantation of human umbilical mesenchymal stem cells attenuates dextran sulfate sodium-induced colitis in mice. Clin Exp Pharmacol Physiol 42(1):76–86
Mao F, Xu WR, Qian H, Zhu W, Yan YM, Shao QX, Xu HX (2010) Immunosuppressive effects of mesenchymal stem cells in collagen-induced mouse arthritis. Inflamm Res 59(3):219–225
Mumy KL, McCormick BA (2009) The role of neutrophils in the event of intestinal inflammation. Curr Opin Pharmacol 9(6):697–701
Nauta AJ, Kruisselbrink AB, Lurvink E, Willemze R, Fibbe WE (2006) Mesenchymal stem cells inhibit generation and function of both CD34+-derived and monocyte-derived dendritic cells. J Immunol 177(4):2080–2087
Okamoto R, Watanabe M (2016) Investigating cell therapy for inflammatory bowel disease. Expert Opin Biol Ther 16(8):1015–1023
Owczarek D, Rodacki T, Domagala-Rodacka R, Cibor D, Mach T (2016) Diet and nutritional factors in inflammatory bowel diseases. World J Gastroenterol 22(3):895–905
Piccard H, Muschel RJ, Opdenakker G (2012) On the dual roles and polarized phenotypes of neutrophils in tumor development and progression. Crit Rev Oncol Hematol 82(3):296–309
Qi Y, Jiang D, Sindrilaru A, Stegemann A, Schatz S, Treiber N, Rojewski M, Schrezenmeier H, Vander Beken S, Wlaschek M, Bohm M, Seitz A, Scholz N, Durselen L, Brinckmann J, Ignatius A, Scharffetter-Kochanek K (2014) TSG-6 released from intradermally injected mesenchymal stem cells accelerates wound healing and reduces tissue fibrosis in murine full-thickness skin wounds. J Invest Dermatol 134(2):526–537
Qian H, Yang H, Xu W, Yan Y, Chen Q, Zhu W, Cao H, Yin Q, Zhou H, Mao F, Chen Y (2008) Bone marrow mesenchymal stem cells ameliorate rat acute renal failure by differentiation into renal tubular epithelial-like cells. Int J Mol Med 22(3):325–332
Qiao C, Xu W, Zhu W, Hu J, Qian H, Yin Q, Jiang R, Yan Y, Mao F, Yang H, Wang X, Chen Y (2008) Human mesenchymal stem cells isolated from the umbilical cord. Cell Biol Int 32(1):8–15
Qin F, Zhang H, Liu A, Wang Q, Sun Q, Lu S, Li Q, Guo H, Liu X, Lu Z (2019) Analgesic effect of zanthoxylum nitidum extract in inflammatory pain models through targeting of ERK and NF-kappaB signaling. Front Pharmacol 10:359
Ren G, Zhang L, Zhao X, Xu G, Zhang Y, Roberts AI, Zhao RC, Shi Y (2008) Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell 2(2):141–150
Selmani Z, Naji A, Zidi I, Favier B, Gaiffe E, Obert L, Borg C, Saas P, Tiberghien P, Rouas-Freiss N, Carosella ED, Deschaseaux F (2008) Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+regulatory T cells. Stem Cells 26(1):212–222
Sotiropoulou PA, Perez SA, Gritzapis AD, Baxevanis CN, Papamichail M (2006) Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells 24(1):74–85
Teimourian S, Moghanloo E (2016) Thwarting PTEN expression by siRNA augments HL-60 cell differentiation to neutrophil-like cells by DMSO and ATRA. DNA Cell Biol 35(10):591–598
Uranga JA, Lopez-Miranda V, Lombo F, Abalo R (2016) Food, nutrients and nutraceuticals affecting the course of inflammatory bowel disease. Pharmacol Rep 68(4):816–826
Wera O, Lancellotti P, Oury C (2016) The dual role of neutrophils in inflammatory bowel diseases. J Clin Med 5(12):118
Zhang X, Zhang W, Yuan X, Fu M, Qian H, Xu W (2016) Neutrophils in cancer development and progression: roles, mechanisms, and implications (review). Int J Oncol 49(3):857–867
Zhang X, Shi H, Yuan X, Jiang P, Qian H, Xu W (2018) Tumor-derived exosomes induce N2 polarization of neutrophils to promote gastric cancer cell migration. Mol Cancer 17(1):146
Zhou GX, Liu ZJ (2017) Potential roles of neutrophils in regulating intestinal mucosal inflammation of inflammatory bowel disease. J Dig Dis 18(9):495–503
Zhu Y, Yang Y, Li F, Fan S, Chen X, Lu Y, Wei Y, Chen Q, Xia L, Tang J, Huang Q, Zhu Q, Zheng J, Liu X (2019) Stimulation of the class—a scavenger receptor induces neutrophil extracellular traps (NETs) by ERK dependent NOX2 and ROMO1 activation. Biochem Biophys Res Commun 511(4):847–854
Acknowledgements
This study was funded by the National Natural Science Foundation of China (Grant Nos. 81670502, 81672416 and 81602883), Jiangsu Key Research and Development Project (Grant No. BE2016717), the Scientific Research Foundation of Jiangsu University (Grant No. FCJJ2015023), the opening project of the Key Laboratory of Embryo Molecular Biology, Ministry of Health of China, and Shanghai Key Laboratory of Embryo and Reproduction Engineering (Grant No. KF201601), Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions and Zhenjiang Key Laboratory of High Technology Research on Exosomes Foundation and Transformation Application (Grant No. SS2018003).
Funding
The National Natural Science Foundation of China (Grant No. 81670502).
Author information
Authors and Affiliations
Contributions
GW, conception and design, collection and/or assembly of data, data analysis and interpretation, and manuscript writing; MDMJ, collection and/or assembly of data and data analysis; JY, conception and design, collection of data, data analysis and interpretation; JW, collection and/or assembly of data and data analysis; XC, data analysis and interpretation; DKWO, collection and/or assembly of data and data analysis; YY, provision of study material and interpretation; HQ, data analysis and interpretation; XZ, collection and/or assembly of data; WX, data analysis and interpretation; FM, study design, data analysis and interpretation, manuscript writing, and final approval of manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, G., Joel, M.D.M., Yuan, J. et al. Human umbilical cord mesenchymal stem cells alleviate inflammatory bowel disease by inhibiting ERK phosphorylation in neutrophils. Inflammopharmacol 28, 603–616 (2020). https://doi.org/10.1007/s10787-019-00683-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-019-00683-5