Abstract
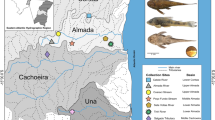
The pattern of morphological and mtDNA cytochrome b diversity of three calanoid copepod species belonging to the diaptomid genus Hemidiaptomus has been investigated with the aim of checking the reliability of the morphological characters currently used for species identification, and the possible presence of cryptic taxa. A sharply different molecular structuring has been observed in the studied species: while Hemidiaptomus amblyodon exhibits a remarkable constancy throughout the European range of its distribution area (maximum inter-populations cytochrome b divergence of 3%), observed distances between presumed conspecific lineages of Hemidiaptomus gurneyi (maximum divergence of 21.5%) and Hemidiaptomus ingens (maximum 19.1%) suggest that under these binomens are in fact included complexes of cryptic, or currently just unrecognized, independent evolutionary lineages. The application of the “4x rule” shows that the two lineages singled out within H. ingens are in fact independent evolutionary units, while the complex molecular structure observed in H. gurneyi s.l. could not be resolved based on the currently available data. Applying standard crustacean mtDNA evolutionary rates to the observed divergence values, the separation of the main lineages within both H. ingens and H. gurneyi might dates back to the Miocene; however, it has also to be considered that the rate of mtDNA evolution might be accelerated in copepods, as already observed in other arthropod taxa. Present results gives further evidences of the high potential for copepod speciation with no or little morphological changes, and stress the need of a revision of the most controversial Palaearctic diaptomid genera.




Similar content being viewed by others
References
Adamowicz, S. J., S. Menu-Marque, P. D. N. Hebert & A. Purvis, 2007. Molecular systematics and patterns of morphological evolution in the Centropagidae (Copepoda: Calanoida) of Argentina. Biological Journal of the Linnean Society 90: 279–292.
Alonso, M., 1998. Las lagunas de la Espaňa peninsular. Limnetica 15: 1–176.
Bennike, O., 1998. Fossil egg sacs of Diaptomus (Crustacea: Copepoda) in Late Quaternary lake sediments. Journal of Paleolimnology 19: 77–79.
Birky, C. W. Jr. & T. Barraclough, 2010. Asexual Speciation. In Schon, I., K. Martens, & P. van Dijk (eds), Lost Sex. Springer, New York: 201–216.
Birky, C. W. Jr., C. Wolf, H. Maughan, L. Herbertson & E. Henry, 2005. Speciation and selection without sex. Hydrobiologia 546: 29–45.
Birky, C. W. Jr., J. Adams, M. Gemmel & J. Perry, 2010. Using Population genetic theory and DNA sequences for species detection and identification in asexual organisms. PLoS One. (in press).
Bohonak, A. J., M. D. Holland, B. Santer, M. Zeller, C. M. Kearns & N. G. Hairston, 2006. The population genetic consequences of diapause in Eudiaptomus copepods. Archiv für Hydrobiologie 167: 183–202.
Boileau, M. G., 1991. A genetic determination of cryptic species (Copepoda: Calanoida) and their postglacial biogeography in North America. Zoological Journal of the Linnean Society 102: 375–396.
Borutzky, E. B., L. A. Stepanova & M. S. Koss, 1991. Opredelitel’ Calanoida presnykh vod SSSR. Nauka, St. Petersburg.
Bucklin, A. & B. W. Frost, 2009. Morphological and molecular phylogenetic analysis of evolutionary lineages within Clausocalanus (Copepoda: Calanoida). Journal of Crustacean Biology 29: 111–120.
Cáceres, C. E. & D. A. Soluk, 2002. Blowing in the wind: a field test of overland dispersal and colonization by aquatic invertebrates. Oecologia 131: 402–408.
Costa, F. O., J. R. de Waard, J. Boutillier, S. Ratnasingham, R. T. Dooh, M. Hajibabaei & P. D. N. Hebert, 2007. Biological identifications through DNA barcodes: the case of the Crustacea. Canadian Journal of Fisheries and Aquatic Sciences 64: 272–295.
De Queiroz, K., 2005. Ernst Mayr and the modern concept of species. Proceedings of the National Academy of Science 102: 6600–6607.
De Queiroz, K., 2007. Species concepts and species delimitation. Systematic Biology 56: 879–886.
De Stasio, B. T., 1989. The seed bank of a freshwater crustacean: copepodology for the plant ecologist. Ecology 70: 1377–1389.
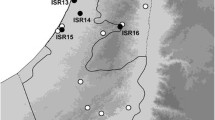
Dimentman, C. & F. D. Por, 1985. Diaptomidae (Copepoda) of Israel and Northern Sinai. Hydrobiologia 127: 89–95.
Dussart, B. & D. Defaye, 2001. Introduction to the Copepoda, 2nd edn. Backhuys Publishers, Leiden.
Dussart, B. & D. Defaye, 2002. World Directory of Crustacea Copepoda of Inland Waters, I—Calaniformes. Backhuys Publishers, Leiden.
Einsle, U., 1993. Crustacea Copepoda. Calanoida und Cyclopoida. Süsswasserfauna Mitteleuropas 8/4-1, Gustav Fisher Verlag.
Elias-Gutierrez, M., F. M., Jeronimo, N. V., Ivanova, M. Valdez-Moreno & P. D. N. Hebert, 2008. DNA barcodes for Cladocera and Copepoda from Mexico and Guatemala, highlights and new discoveries. Zootaxa 1839: 1–42.
Figuerola, J. & A. J. Green, 2002. Dispersal of aquatic organisms by waterbirds: a review of past research and priorities for future studies. Freshwater Biology 47: 483–494.
Gauthier, H., 1928. Recherches sur la faune des eaux continentales de l’Algérie et de la Tunisie. Imprimerie Minerva, Alger.
Graham, T. B. & D. Wirth, 2008. Dispersal of large branchiopod cysts: potential movement by wind from potholes. Hydrobiologia 600: 17–27.
Grant, W. S. & B. W. Bowen, 1998. Shallow population histories in deep evolutionary lineages of marine fishes: insights from sardines and anchovies and lessons for conservation. The Journal of Heredity 89: 415–426.
Grishanin, A. K., E. M. Rasch, S. I. Dodson & G. A. Wyngaard, 2006. Genetic architecture of the cryptic species complex of Acanthocyclops vernalis (Crustacea: Copepoda). II. Crossbreeding experiments, cytogenetics, and a model of chromosomal evolution. Evolution 60: 247–256.
Guindon, S. & O. Gascuel, 2003. A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Systematic Biology 52: 696–704.
Hey, J., 2006. On the failure of modern species concepts. Trends in Ecology and Evolution 21: 447–450.
Hewitt, G., 2004. Genetic consequences of climatic oscillations in the Quaternary. Philosophical Transactions of the Royal Society B 359: 183–195.
Huys, R. & G. A. Boxshall, 1991. Copepod Evolution. The Ray Society, London.
Kiefer, F., 1954. Freilebende Ruderfusskrebse (Crustacea Copepoda) aus Binnengewaessern Marokkos. Bulletin de la Société des Sciences Naturelles et Physiques du Maroc 34: 317–336.
Kiefer, F., 1973. Zur Kenntnis der roubaui-Gruppe der Gattung Hemidiaptomus G. O. Sars (Copepoda). Crustaceana 25: 281–291.
Kiefer, F., 1978. Das Zooplankton der Binnengewaesser. Freilebende Copepoda. Die Binnengewaesser, 26/2. E. Schweizerbart’sche Verlagbuchhandlung, Stuttgart.
Lee, C. E., 2000. Global phylogeography of a cryptic copepod species complex and reproductive isolation between genetically proximate ‘populations’. Evolution 54: 2014–2027.
Machida, R. J., M. U. Miya, M. Nishida & S. Nishida, 2004. Large-scale gene rearrangements in the mitochondrial genomes of two calanoid copepods Eucalanus bungii and Neocalanus cristatus (Crustacea), with notes on new versatile primers for the srRNA and COI genes. Gene 332: 71–78.
Marrone, F. & L. Naselli-Flores, 2004. First record and morphological features of Hemidiaptomus (Occidodiaptomus) ingens (Gurney, 1909) (Copepoda Calanoida) in Italy. Journal of Limnology 63: 250–255.
Marszalek, M. A., S. Dayanandan & E. J. Maly, 2009. Phylogeny of the genus Hesperodiaptomus Copepoda based on nucleotide sequence data of the nuclear ribosomal gene. Hydrobiologia 624: 61–69.
Merrit, T. J. S., L. Shi, M. C. Chase, M. A. Rex, R. J. Etter & J. M. Quattro, 1998. Universal cytochrome b primers facilitate intraspecific studies in molluscan taxa. Molecular Marine Biology and Biotechnology 7: 7–11.
Papadopoulos, L. N., K. T. C. A. Peijnenburg & P. C. Luttikhuizen, 2005. Phylogeography of the calanoid copepods Calanus helgolandicus and C. euxinus suggests Pleistocene divergences between Atlantic, Mediterranean, and Black Sea populations. Marine Biology 147: 1353–1365.
Posada, D., 2008. jModelTest: phylogenetic model averaging. Molecular Biology and Evolution 25: 1253–1256.
Roy, J. & H. Gauthier, 1927. Sur les copépodes d’Algérie et Tunisie (eaux douces et eaux saumâtres). Bulletin de la Société Zoologique de France 52: 558–575.
Schmitt, T., 2007. Molecular biogeography of Europe Pleistocene cycles and postglacial trends. Frontiers in Zoology 4: 11. doi:10.1186/1742-9994-4-11
Stella, E., 1984. Copepoda: Calanoida. Fauna d’Italia, vol 21, Ed. Calderini, Bologna.
Stepanova, L. A., 2005. Composition of the genus Hemidiaptomus: identification of Gigantodiaptomus and Occidodiaptomus (Crustacea, Diaptomidae) as independent genera. Zoologicheskii Zhurnal 84: 754–760.
Steward, J. R. & A. M. Lister, 2001. Cryptic northern refugia and the origins of the modern biota. Trends in Ecology and Evolution 16: 608–613.
Swofford, D. L., 1998. PAUP*. Phylogenetic Analysis Using Parsimony (*and Other Methods). Version 4. Sinauer Associates, Sunderland, Massachusetts.
Thompson, J. D., T. J. Gibson, F. Plewniak, F. Jeanmougin & D. G. Higgins, 1997. The ClustalX windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research 24: 4876–4882.
Thum, R. A., 2004. Using 18S rDNA to resolve diaptomid copepod (Copepoda: Calanoida: Diaptomidae) phylogeny: an example with the North American genera. Hydrobiologia 519: 135–141.
Thum, R. A. & A. M. Derry, 2008. Taxonomic implications for diaptomid copepods based on contrasting patterns of mitochondrial DNA sequence divergences in four morphospecies. Hydrobiologia 614: 197–207.
Thum, R. A. & R. G. Harrison, 2009. Deep genetic divergences among morphologically similar and parapatric Skistodiaptomus (Copepoda: Calanoida: Diaptomidae) challenge the hypothesis of Pleistocene speciation. Biological Journal of the Linnean Society 96: 150–165.
Zeller, M., T. B. H. Reusch & W. Lampert, 2006. A comparative population genetic study on calanoid freshwater copepods: investigation of isolation-by-distance in two Eudiaptomus species with a different potential for dispersal. Limnology and Oceanography 51: 117–124.
Acknowledgments
Bill Birky (University of Arizona, Tucson) provided a much appreciated support with the implementation of the “4x rule” and sensibly improved a former draft of the manuscript with his constructive criticism. The friends and colleagues listed in Table 1 generously provided the Hemidiaptomus samples included in this study, and they are gratefully acknowledged for their support. Research supported by “Fondi di Ateneo - Università di Palermo”. This research has been carried out as a part of the PhD of FM at the Department of Animal Biology of the University of Palermo.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: K. Martens
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Marrone, F., Brutto, S.L. & Arculeo, M. Molecular evidence for the presence of cryptic evolutionary lineages in the freshwater copepod genus Hemidiaptomus G.O. Sars, 1903 (Calanoida, Diaptomidae). Hydrobiologia 644, 115–125 (2010). https://doi.org/10.1007/s10750-010-0101-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-010-0101-6