Abstract
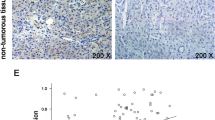
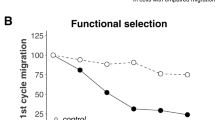
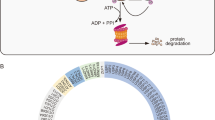
Breast cancer is the second leading cause of cancer-related death in women. Previously, evidence suggested that ubiquitin-specific protease 14 (USP14) was associated with various signal transduction pathways and tumourigenesis. In this study, we demonstrate that USP14 is a novel therapeutic target in breast cancer. A Western blot analysis of USP14 was performed using seven breast cancer tissues and paired adjacent normal tissues and showed that the expression of USP14 was increased in the breast cancer tissues. Immunohistochemistry was conducted on formalin-fixed paraffin-embedded sections of breast cancer samples from 100 cases. Using Pearson’s χ2 test, it was demonstrated that USP14 expression was associated with the histological grade, lymph node status and Ki-67 expression in the tumour. The Kaplan–Meier analysis revealed that increased USP14 expression in patients with breast cancer was associated with a poorer prognosis. In in vitro experiments, the highly migratory MDA-MB-231 cells that were treated with USP14-shRNA (shUSP14) exhibited decreased motility using Transwell migration assays. Next, we employed a starvation and re-feeding assay, and the CCK-8 assay demonstrated that USP14 regulated breast cancer cell proliferation. Furthermore, we used flow cytometry to analyse cellular apoptosis following USP14 knockdown. Taken together, our results suggested that USP14 was involved in the progression of breast cancer.






Similar content being viewed by others
References
Acloque H, Adams MS, Fishwick K, Bronner-Fraser M, Nieto MA (2009) Epithelial-mesenchymal transitions: the importance of changing cell state in development and disease. J Clin Invest 119(6):1438–1449. doi:10.1172/JCI38019
Adamczyk A, Niemiec JA, Ambicka A, Mucha-Malecka A, Mitus J, Rys J (2014) CD44/CD24 as potential prognostic markers in node-positive invasive ductal breast cancer patients treated with adjuvant chemotherapy. J Mol Histol 45(1):35–45. doi:10.1007/s10735-013-9523-6
Adhikari A, Xu M, Chen ZJ (2007) Ubiquitin-mediated activation of TAK1 and IKK. Oncogene 26(22):3214–3226. doi:10.1038/sj.onc.1210413
Anderson C, Crimmins S, Wilson JA, Korbel GA, Ploegh HL, Wilson SM (2005) Loss of Usp14 results in reduced levels of ubiquitin in ataxia mice. J Neurochem 95(3):724–731. doi:10.1111/j.1471-4159.2005.03409.x
Bayraktar S, Gutierrez Barrera AM, Liu D et al (2013) USP-11 as a predictive and prognostic factor following neoadjuvant therapy in women with breast cancer. Cancer J 19(1):10–17. doi:10.1097/PPO.0b013e3182801b3a
Brnjic S, Mazurkiewicz M, Fryknas M et al (2014) Induction of tumor cell apoptosis by a proteasome deubiquitinase inhibitor is associated with oxidative stress. Antioxid Redox Signal 21(17):2271–2285. doi:10.1089/ars.2013.5322
Chitta K, Paulus A, Akhtar S et al (2015) Targeted inhibition of the deubiquitinating enzymes, USP14 and UCHL5, induces proteotoxic stress and apoptosis in Waldenstrom macroglobulinaemia tumour cells. Br J Haematol. doi:10.1111/bjh.13304
Chuensumran U, Saelee P, Punyarit P et al (2011) Ubiquitin-specific protease 14 expression associated with intrahepatic cholangiocarcinoma cell differentiation. Asian Pac J Cancer Prev: APJCP 12(3):775–779
Ciechanover A, Schwartz AL (2004) The ubiquitin system: pathogenesis of human diseases and drug targeting. Biochim Biophys Acta 1695(1–3):3–17. doi:10.1016/j.bbamcr.2004.09.018
Cui X, Liu Y, Wan C et al (2014) Decreased expression of SERPINB1 correlates with tumor invasion and poor prognosis in hepatocellular carcinoma. J Mol Histol 45(1):59–68. doi:10.1007/s10735-013-9529-0
D’Arcy P, Linder S (2012) Proteasome deubiquitinases as novel targets for cancer therapy. Int J Biochem Cell Biol 44(11):1729–1738. doi:10.1016/j.biocel.2012.07.011
D’Arcy P, Brnjic S, Olofsson MH et al (2011) Inhibition of proteasome deubiquitinating activity as a new cancer therapy. Nat Med 17(12):1636–1640. doi:10.1038/nm.2536
Daviet L, Colland F (2008) Targeting ubiquitin specific proteases for drug discovery. Biochimie 90(2):270–283. doi:10.1016/j.biochi.2007.09.013
Dou X, Wei J, Sun A et al (2015) PBK/TOPK mediates geranylgeranylation signaling for breast cancer cell proliferation. Cancer Cell Int 15(1):27. doi:10.1186/s12935-015-0178-0
Foroni C, Broggini M, Generali D, Damia G (2012) Epithelial-mesenchymal transition and breast cancer: role, molecular mechanisms and clinical impact. Cancer Treat Rev 38(6):689–697. doi:10.1016/j.ctrv.2011.11.001
Frisan T, Coppotelli G, Dryselius R, Masucci MG (2012) Ubiquitin C-terminal hydrolase-L1 interacts with adhesion complexes and promotes cell migration, survival, and anchorage independent growth. FASEB J Off Publ Fed Am Soc Exp Biol 26(12):5060–5070. doi:10.1096/fj.12-211946
Hallengren J, Chen PC, Wilson SM (2013) Neuronal ubiquitin homeostasis. Cell Biochem Biophys 67(1):67–73. doi:10.1007/s12013-013-9634-4
Hartmann WH, Ozzello L, Sobin LH, Stalsberg H (1981) Histological typing of breast tumours. In: International classification of tumours. World Health Organization, Geneva, pp 15–25
Hay ED (1995) An overview of epithelio-mesenchymal transformation. Acta Anat 154(1):8–20
Huang Z, Wen P, Kong R et al (2015) USP33 mediates Slit–Robo signaling in inhibiting colorectal cancer cell migration. Int J Cancer J Int Du Cancer 136(8):1792–1802. doi:10.1002/ijc.29226
Ji L, Li H, Gao P et al (2013) Nrf2 pathway regulates multidrug-resistance-associated protein 1 in small cell lung cancer. PLoS One 8(5):e63404. doi:10.1371/journal.pone.0063404
Jin C, Yang YA, Anver MR, Morris N, Wang X, Zhang YE (2009) Smad ubiquitination regulatory factor 2 promotes metastasis of breast cancer cells by enhancing migration and invasiveness. Cancer Res 69(3):735–740. doi:10.1158/0008-5472.CAN-08-1463
Jung H, Kim BG, Han WH et al (2013) Deubiquitination of Dishevelled by Usp14 is required for Wnt signaling. Oncogenesis 2:e64. doi:10.1038/oncsis.2013.28
Kapuria V, Peterson LF, Fang D, Bornmann WG, Talpaz M, Donato NJ (2010) Deubiquitinase inhibition by small-molecule WP1130 triggers aggresome formation and tumor cell apoptosis. Cancer Res 70(22):9265–9276. doi:10.1158/0008-5472.CAN-10-1530
Koulich E, Li X, DeMartino GN (2008) Relative structural and functional roles of multiple deubiquitylating proteins associated with mammalian 26S proteasome. Mol Biol Cell 19(3):1072–1082. doi:10.1091/mbc.E07-10-1040
Liu Y, Chen Y, Lu X et al (2012) SCYL1BP1 modulates neurite outgrowth and regeneration by regulating the Mdm2/p53 pathway. Mol Biol Cell 23(23):4506–4514. doi:10.1091/mbc.E12-05-0362
Liu X, Ni Q, Xu J et al (2014) Expression and prognostic role of SKIP in human breast carcinoma. J Mol Histol 45(2):169–180. doi:10.1007/s10735-013-9546-z
Lopez-Castejon G, Luheshi NM, Compan V et al (2013) Deubiquitinases regulate the activity of caspase-1 and interleukin-1beta secretion via assembly of the inflammasome. J Biol Chem 288(4):2721–2733. doi:10.1074/jbc.M112.422238
Lyons JG, Lobo E, Martorana AM, Myerscough MR (2008) Clonal diversity in carcinomas: its implications for tumour progression and the contribution made to it by epithelial-mesenchymal transitions. Clin Exp metastasis 25(6):665–677. doi:10.1007/s10585-007-9134-2
Mialki RK, Zhao J, Wei J, Mallampalli DF, Zhao Y (2013) Overexpression of USP14 protease reduces I-kappaB protein levels and increases cytokine release in lung epithelial cells. J Biol Chem 288(22):15437–15441. doi:10.1074/jbc.C112.446682
Mines MA, Goodwin JS, Limbird LE, Cui FF, Fan GH (2009) Deubiquitination of CXCR4 by USP14 is critical for both CXCL12-induced CXCR4 degradation and chemotaxis but not ERK ativation. J Biol Chem 284(9):5742–5752. doi:10.1074/jbc.M808507200
Phillips AH, Zhang Y, Cunningham CN et al (2013) Conformational dynamics control ubiquitin–deubiquitinase interactions and influence in vivo signaling. Proc Natl Academy Sci USA 110(28):11379–11384. doi:10.1073/pnas.1302407110
Runowicz CD, Leach CR, Henry NL et al (2015) American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline. J Clin Oncol off J Am Soc Clin Oncol. doi:10.1200/JCO.2015.64.3809
Shi M, Cao M, Song J et al (2015a) PinX1 inhibits the invasion and metastasis of human breast cancer via suppressing NF-kappaB/MMP-9 signaling pathway. Mol Cancer 14(1):66. doi:10.1186/s12943-015-0332-2
Shi Y, Liu X, Sun Y et al (2015b) Decreased expression and prognostic role of EHD2 in human breast carcinoma: correlation with E-cadherin. J Mol Histol 46(2):221–231. doi:10.1007/s10735-015-9614-7
Shinji S, Naito Z, Ishiwata S et al (2006) Ubiquitin-specific protease 14 expression in colorectal cancer is associated with liver and lymph node metastases. Oncol Rep 15(3):539–543
Tao T, Cheng C, Ji Y et al (2012) Numbl inhibits glioma cell migration and invasion by suppressing TRAF5-mediated NF-kappaB activation. Mol Biol Cell 23(14):2635–2644. doi:10.1091/mbc.E11-09-0805
Thiery JP (2002) Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer 2(6):442–454. doi:10.1038/nrc822
Thiery JP, Acloque H, Huang RY, Nieto MA (2009) Epithelial-mesenchymal transitions in development and disease. Cell 139(5):871–890. doi:10.1016/j.cell.2009.11.007
Tian Z, D’Arcy P, Wang X et al (2014) A novel small molecule inhibitor of deubiquitylating enzyme USP14 and UCHL5 induces apoptosis in multiple myeloma and overcomes bortezomib resistance. Blood 123(5):706–716. doi:10.1182/blood-2013-05-500033
Voulgari A, Pintzas A (2009) Epithelial-mesenchymal transition in cancer metastasis: mechanisms, markers and strategies to overcome drug resistance in the clinic. Biochim Biophys Acta 1796(2):75–90. doi:10.1016/j.bbcan.2009.03.002
Wang Y, Yang S, Ni Q et al (2012) Overexpression of forkhead box J2 can decrease the migration of breast cancer cells. J Cell Biochem 113(8):2729–2737. doi:10.1002/jcb.24146
Wang X, Stafford W, Mazurkiewicz M et al (2014) The 19S Deubiquitinase inhibitor b-AP15 is enriched in cells and elicits rapid commitment to cell death. Mol Pharmacol 85(6):932–945. doi:10.1124/mol.113.091322
Wang Y, Wang J, Zhong J et al (2015) Ubiquitin-specific protease 14 (USP14) regulates cellular proliferation and apoptosis in epithelial ovarian cancer. Med Oncol 32(1):379. doi:10.1007/s12032-014-0379-8
Wu N, Liu C, Bai C, Han YP, Cho WC, Li Q (2013) Over-Expression of deubiquitinating enzyme USP14 in lung adenocarcinoma promotes proliferation through the accumulation of beta-catenin. Int J Mol Sci 14(6):10749–10760. doi:10.3390/ijms140610749
Zhang D, Zaugg K, Mak TW, Elledge SJ (2006) A role for the deubiquitinating enzyme USP28 in control of the DNA-damage response. Cell 126(3):529–542. doi:10.1016/j.cell.2006.06.039
Zhu T, Ji Z, Xu C et al (2014) Expression and prognostic role of SGTA in human breast carcinoma correlates with tumor cell proliferation. J Mol Histol 45(6):665–677. doi:10.1007/s10735-014-9586-z
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81302285) and the Natural Scientific Foundation of Nantong University Grant (No. 12Z009).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Lianxin Zhu and Shuyun Yang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhu, L., Yang, S., He, S. et al. Downregulation of ubiquitin-specific protease 14 (USP14) inhibits breast cancer cell proliferation and metastasis, but promotes apoptosis. J Mol Hist 47, 69–80 (2016). https://doi.org/10.1007/s10735-015-9650-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-015-9650-3