Abstract
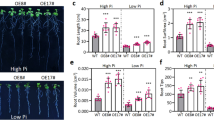
Transcription factors (TFs) fused to the SRDX (a modified repressor domain at the C-terminal region of Arabidopsis SUPERMAN (SUPRD) with the sequence of LDLDLELRLGFA) motif repression domain at their C terminus, generating a chimeric repressor, could act as a dominant suppressor and overcome problems related to functional redundancy between TFs. Here, we demonstrate that transgenic Arabidopsis containing a chimeric AtERF4-SRDX construct display a variety of changes in plant growth and development, such as short primary roots and root hairs at early developmental stages, an increased number of rosette leaves with decreased leaf area, elongation of leaf petioles and inflorescence stems, prolonged growth period, reduced stem and leaf angle and a modified seed coat mucilage structure. The short primary root and the elongation of petioles and stems were due to reduced and enhanced lengthwise cell expansion respectively, while the redistribution of seed mucilage appeared to be the result of altered pectin methylesterase activity. Expression of AtERF4-SRDX also affected abiotic stress tolerance of transgenic plants, inducing enhanced resistance to drought but reduced tolerance to salinity and heat, which could be linked to abscisic acid signaling. Altogether, our findings provide new insights into the possible functions of AtERF4 in Arabidopsis growth and development, and in mediating plant responses to abiotic stresses.






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Arsovski AA, Haughn GW, Western TL (2010) Seed coat mucilage cells of Arabidopsis thaliana as a model for plant cell wall research. Plant Signal Behav 5:796–801. https://doi.org/10.4161/psb.5.7.11773
Bai MY, Fan M, Oh E et al (2012) A triple helix-loop-helix/basic helix-loop-helix cascade controls cell elongation downstream of multiple hormonal and environmental signaling pathways in Arabidopsis. Plant Cell 24:4917–4929. https://doi.org/10.1105/tpc.112.105163
Barker R, Fernandez Garcia MN, Powers SJ et al (2021) Mapping sites of gibberellin biosynthesis in the Arabidopsis root tip. New Phytol 229:1521–1534. https://doi.org/10.1111/nph.16967
Chen JH, Jiang HW, Hsieh EJ et al (2012) Drought and salt stress tolerance of an Arabidopsis glutathione S-transferase U17 knockout mutant are attributed to the combined effect of glutathione and abscisic acid. Plant Physiol 158:340–351. https://doi.org/10.1104/pp.111.181875
Chen Z, Liu Y, Yin Y et al (2019) Expression of AtGA2ox1 enhances drought tolerance in maize. Plant Growth Regul 89:203–215. https://doi.org/10.1007/s10725-019-00526-x
Chung K, Nakano T, Fujiwara S et al (2016a) The ERF transcription factor EPI1 is a negative regulator of dark-induced and jasmonate-stimulated senescence in Arabidopsis. Plant Biotechnol 33:235–243. https://doi.org/10.5511/plantbiotechnology.16.0127a
Chung K, Sakamoto S, Mitsuda N et al (2016b) WUSCHEL-RELATED HOMEOBOX 2 is a transcriptional repressor involved in lateral organ formation and separation in Arabidopsis. Plant Biotechnol 33:245–253. https://doi.org/10.5511/plantbiotechnology.16.0202a
Chuong NN, Hoang XLT, Nghia DHT et al (2021) Ectopic expression of GmHP08 enhances resistance of transgenic Arabidopsis toward drought stress. Plant Cell Rep. https://doi.org/10.1007/s00299-021-02677-6
Denancé N, Sánchez-Vallet A, Goffner D et al (2013) Disease resistance or growth: the role of plant hormones in balancing immune responses and fitness costs. Front Plant Sci 4:155. https://doi.org/10.3389/fpls.2013.00155
Ding AM, Tang XF, Yang DH et al (2021) ERF4 and MYB52 transcription factors play antagonistic roles in regulating homogalacturonan de-methylesterification in Arabidopsis seed coat mucilage. Plant Cell 33:381–403. https://doi.org/10.1093/plcell/koaa031
Fujimoto SY, Ohta M, Usui A et al (2000) Arabidopsis ethylene-responsive element binding factors act as transcriptional activators or repressors of GCC box-mediated gene expression. Plant Cell 12:393–404. https://doi.org/10.1105/tpc123393
Gocal GF, Sheldon CC, Gubler F et al (2001) GAMYB-like genes, flowering, and gibberellin signaling in Arabidopsis. Plant Physiol 127:1682–1693. https://pubmed.ncbi.nlm.nih.gov/11743113/
Greene GH, Dong X (2018) To grow and to defend. Science 361:976–977. https://doi.org/10.1126/scienceaau9065
Guo S, Dai S, Singh PK et al (2018) A membrane-bound NAC-like transcription factor OsNTL5 represses the flowering in Oryza sativa. Front Plant Sci 9:555. https://doi.org/10.3389/fpls.2018.00555
Haughn GW, Western TL (2012) Arabidopsis seed coat mucilage is a specialized cell wall that can be used as a model for genetic analysis of plant cell wall structure and function. Front Plant Sci 3:64. https://doi.org/10.3389/fpls.2012.00064
Hiratsu K, Matsui K, Koyama T et al (2003) Dominant repression of target genes by chimeric repressors that include the EAR motif, a repression domain, in Arabidopsis. Plant J 34:733–739. https://doi.org/10.1046/j1365-313x200301759x
Kalve S, De Vos D, Beemster GT (2014a) Leaf development: a cellular perspective. Front Plant Sci 5:362. https://doi.org/10.3389/fpls201400362
Kalve S, Fotschki J, Beeckman T et al (2014b) Three-dimensional patterns of cell division and expansion throughout the development of Arabidopsis thaliana leaves. J Exp Bot 65:6385–6397. https://doi.org/10.1093/jxb/eru358
Koyama T, Sato F (2018) The function of ETHYLENE RESPONSE FACTOR genes in the light-induced anthocyanin production of Arabidopsis thaliana leaves. Plant Biotechnol 35:87–91. https://doi.org/10.5511/plantbiotechnology180122b
Koyama T, Nii H, Mitsuda N et al (2013) A regulatory cascade involving class II ETHYLENE RESPONSE FACTOR transcriptional repressors operates in the progression of leaf senescence. Plant Physiol 162:991–1005. https://doi.org/10.1104/pp113218115
Li Y, Cai H, Liu P et al (2017) Arabidopsis MAPKKK18 positively regulates drought stress resistance via downstream MAPKK3. Biochem Biophys Res Commun 484:292–297. https://doi.org/10.1016/j.bbrc.2017.01.104
Lyons R, Iwase A, Gänsewig T et al (2013) The RNA-binding protein FPA regulates flg22-triggered defense responses and transcription factor activity by alternative polyadenylation. Sci Rep 3:2866. https://doi.org/10.1038/srep02866
Matsui K, Hiratsu K, Koyama T et al (2005) A chimeric AtMYB23 repressor induces hairy roots, elongation of leaves and stems, and inhibition of the deposition of mucilage on seed coats in Arabidopsis. Plant Cell Physiol 46:147–155. https://doi.org/10.1093/pcp/pci005
McGrath KC, Dombrecht B, Manners JM et al (2005) Repressor- and activator-type ethylene response factors functioning in jasmonate signaling and disease resistance identified via a genome-wide screen of Arabidopsis transcription factor gene expression. Plant Physiol 139:949–959. https://doi.org/10.1104/pp105068544
Nakano T, Suzuki K, Fujimura T et al (2006) Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol 140:411–432. https://doi.org/10.1104/pp105073783
Nguyen KH, Mostofa MG, Li W et al (2018) The soybean transcription factor GmNAC085 enhances drought tolerance in Arabidopsis. Environ Exp Bot 151:12–20. https://doi.org/10.1016/j.envexpbot.2018.03.017
Ohta M, Matsui K, Hiratsu K et al (2001) Repression domains of class II ERF transcriptional repressors share an essential motif for active repression. Plant Cell 13:1959–1968. https://doi.org/10.1105/tpc010127
Riester L, Köster-Hofmann S, Doll J et al (2019) Impact of alternatively polyadenylated isoforms of ETHYLENE RESPONSE FACTOR4 with activator and repressor function on senescence in Arabidopsis thaliana L. Genes 10:91. https://doi.org/10.3390/genes10020091
Sakuma Y, Liu Q, Dubouzet JG et al (2002) DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREBs, transcription factors involved in dehydration- and cold-inducible gene expression. Biochem Biophys Res Commun 290:998–1009. https://doi.org/10.1006/bbrc20016299
Tiwari SB, Hagen G, Guilfoyle TJ (2004) Aux/IAA proteins contain a potent transcriptional repression domain. Plant Cell 16:533–543. https://doi.org/10.1105/tpc017384
Waśkiewicz A, Beszterda M, Goliński P (2013) ABA: Role in plant signaling under salt stress. In: Ahmad P, Azooz MM, Prasad MNV (eds) Salt Stress in Plants. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-6108-1_8
Wu J, Yu C, Hunag L et al (2020) Overexpression of MADS-box transcription factor OsMADS25 enhances salt stress tolerance in Rice and Arabidopsis. Plant Growth Regul 90:163–171. https://doi.org/10.1007/s10725-019-00539-6
Yang Z, Tian L, Latoszek-Green M et al (2005) Arabidopsis ERF4 is a transcriptional repressor capable of modulating ethylene and abscisic acid responses. Plant Mol Biol 58:585–596. https://doi.org/10.1007/s11103-005-7294-5
Ye J, Zhong T, Zhang D et al (2019) The auxin-regulated protein ZmAuxRP1 coordinates the balance between root growth and stalk rot disease resistance in Maize. Mol Plant 12:360–373. https://doi.org/10.1016/jmolp201810005
Zhang H, Zhao Y, Zhu JK (2020) Thriving under stress: how plants balance growth and the stress response. Dev Cell 55:529–543. https://doi.org/10.1016/j.devcel.2020.10.012
Zhang H, Liu P, Wang B et al (2021) The roles of trichome development genes in stress resistance. Plant Growth Regul 95:137–148. https://doi.org/10.1007/s10725-021-00733-5
Zhou X, Zhang ZL, Park J et al (2016) The ERF11 transcription factor promotes internode elongation by activating gibberellin biosynthesis and signaling. Plant Physiol 171:2760–2770. https://doi.org/10.1104/pp1600154
Zhu SY, Yu XC, Wang XJ et al (2007) Two calcium-dependent protein kinases, CPK4 and CPK11, regulate abscisic acid signal transduction in Arabidopsis. Plant Cell 19:3019–3036. https://doi.org/10.1105/tpc.107.050666
Funding
This work was supported by the National Natural Science Foundation of China (31600237), the Key Science and Technology Projects of Sichuan Tobacco Institute (SCYC202002), the Science and Technology Projects of Yunnan Academy of Tobacco Agricultural Sciences (2020530000241008), the Central Public-Interest Scientific Institution Basal Research Fund (16102320200002 and 1610232016017), and the Science Foundation for Young Scholars of Tobacco Research Institute of CAAS (2016B05).
Author information
Authors and Affiliations
Contributions
AD, ZC and YL conceived and designed research. AD, XY, XY, WW and YS conducted experiments. AD, ZC and YL analyzed the data and wrote the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Mohsin Tanveer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ding, A., Yang, X., Yu, X. et al. A chimeric AtERF4 repressor modulates pleiotropic aspects of plant growth and abiotic stress tolerance in transgenic Arabidopsis. Plant Growth Regul 96, 255–267 (2022). https://doi.org/10.1007/s10725-021-00773-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00773-x