Abstract
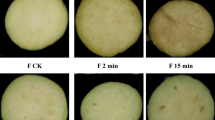
Lotus root (Nelumbo nucifera) is an edible rhizome that the consumption/production has continuously increased as more consumers demand convenient and ready-to-eat foods. However, the processing, storage, and transportation of fresh-cut fruits and vegetables promotes physiological deterioration, such as browning, which leads to a reduction in the value of the product. This study aimed to reduce the browning of lotus root pericarps during long-term storage and long-distance transport, and to clarify the functions of unigenes and browning-associated metabolic pathways using RNA-sequencing techniques. Intact lotus root cv. ‘Kanasumi No.34’ browning decreased after the roots were packed along with an anti-browning solution. Over 200 million short single-end reads were mapped onto the N. nucifera consensus coding sequence set. The significantly differentially expressed genes (DEGs) were identified. Based on the Uniprot, GO, and KEGG databases, secondary metabolism, lipid metabolism, and redox state genes were significantly upregulated in the un-packed and packed with water treatments compared to after harvest sample. Additionally, 16 expected DEGs (e.g., PPO, PAL, POD, CHS, PDCR, and SOD), which are affected by browning development, were differentially regulated in lotus root pericarp. The gene expression data presented in this study will help elucidate the molecular mechanism underlying browning development in intact lotus root during long-term storage. The results may also inform future research on improving the post-harvest shelf life of lotus roots’.








Adapted from Grotheer et al., 2005)
Similar content being viewed by others
References
Alegria C, Gonçalves EM, Moldão-Martins M, Cisneros-Zevallos L, Abreu M (2016) Peel removal improves quality without antioxidant loss, through wound-induced phenolic biosynthesis in shredded carrot. Postharvest Biol Technol 120:232–239. https://doi.org/10.1016/j.postharvbio.2016.07.004
Amin M, Kapadnis BP (2005) Heat stable antimicrobial activity of Allium ascalonicum against bacteria and fungi. Indian J Exp Biol 43:751–754
And OL, Watson MA (2001) Effects of ascorbic acid on peroxidase and polyphenoloxidase activities in fresh-cut cantaloupe melon. J Food Sci 66(9):1283–1286. https://doi.org/10.1111/j.1365-2621.2001.tb15202.x
Aquino-Bolaños EN, Mercado-Silva E (2004) Effects of polyphenol oxidase and peroxidase activity, phenolics and lignin content on the browning of cut jicama. Postharvest Biol Technol 33(3):275–283. https://doi.org/10.1016/j.postharvbio.2004.03.009
Bairoch A, Apweiler R (2000) The SWISS-PROT protein sequence database and its supplement TrEMBL in 2000. Nucleic Acids Res 28(1):45–48. https://doi.org/10.1093/nar/28.1.45
Bairoch A, Boeckmann B (1991) The SWISS-PROT protein sequence data bank. Nucleic Acids Res 19(Suppl):2247. https://doi.org/10.1093/nar/19.suppl.2247
Banerjee A, Suprasanna P, Variyar PS, Sharma A (2015) Gamma irradiation inhibits wound induced browning in shredded cabbage. Food Chem 173:38–44. https://doi.org/10.1016/j.foodchem.2014.09.166
Bernaś E, Jaworska G (2015) Use of onion extract to prevent enzymatic browning of frozen Agaricus bisporus mushrooms. Int J Refrig 57:257–264. https://doi.org/10.1016/j.ijrefrig.2015.04.022
Cantos E, Tudela JA, Gil MI, Espín JC (2002) Phenolic compounds and related enzymes are not rate-limiting in browning development of fresh-cut potatoes. J Agric Food Chem 50(10):3015–3023. https://doi.org/10.1021/jf0116350
Chang MS, An SJ, Jeong MC, Kim DM, Kim GH (2011) Effects of antioxidative activities and antibrowning of extracts from onion, apple and mandarin orange peel as natural antibrowning agents. Korean J Food Nutr 24(3):406–413. https://doi.org/10.9799/ksfan.2011.24.3.406
Cheng Y, Liu L, Zhao G, Shen C, Yan H, Guan J, Yang K (2015) The effects of modified atmosphere packaging on core browning and the expression patterns of PPO and PAL genes in ‘Yali’ pears during cold storage. LWT Food Sci Technol 60(2):1243–1248. https://doi.org/10.1016/j.lwt.2014.09.005
Chi M, Bhagwat B, Lane WD, Tang G, Su Y, Sun R, Oomah BD, Wiersma PA, Xiang Y (2014) Reduced polyphenol oxidase gene expression and enzymatic browning in potato (Solanum tuberosum L.) with artificial microRNAs. BMC Plant Biol 14(1):62. https://doi.org/10.1186/1471-2229-14-62
Chisari M, Barbagallo RN, Spagna G (2008) Characterization and role of polyphenol oxidase and peroxidase in browning of fresh-cut melon. J Agric Food Chem 56(1):132–138. https://doi.org/10.1021/jf0721491
Degl’Innocenti E, Pardossi A, Tognoni F, Guidi L (2007) Physiological basis of sensitivity to enzymatic browning in ‘lettuce’, ‘escarole’ and ‘rocket salad’ when stored as fresh-cut products. Food Chem 104(1):209–215. https://doi.org/10.1016/j.foodchem.2006.11.026
Ding CK, Chachin K, Ueda Y, Wang CY (2002) Inhibition of loquat enzymatic browning by sulfhydryl compounds. Food Chem 76(2):213–218. https://doi.org/10.1016/S0308-8146(01)00270-9
Dokhanieh AY, Aghdam MS, Sarcheshmeh MA (2016) Impact of postharvest hot salicylic acid treatment on aril browning and nutritional quality in fresh-cut pomegranate. Hortic Environ Biotechnol 57(4):378–384. https://doi.org/10.1007/s13580-016-0087-8
Droillard MJ, Paulin A, Massot JC (1987) Free radical production, catalase and superoxide dismutase activities and membrane integrity during senescence of petals of cut carnations (Dianthus caryophyllus). Physiol Plant 71(2):197–202. https://doi.org/10.1111/j.1399-3054.1987.tb02867.x
Duan X, Liu T, Zhang D, Su X, Lin H, Jiang Y (2011) Effect of pure oxygen atmosphere on antioxidant enzyme and antioxidant activity of harvested litchi fruit during storage. Food Res Int 44(7):1905–1911. https://doi.org/10.1016/j.foodres.2010.10.027
Eissa HA, Fadel HHM, Ibrahim GE, Hassan IM, Elrashid AA (2006) Thiol containing compounds as controlling agents of enzymatic browning in some apple products. Food Res Int 39(8):855–863. https://doi.org/10.1016/j.foodres.2006.04.004
Fan M, Li W, Hu X, Sun Yn YuG, Zhang X (2016) Effect of micro-vacuum storage on active oxygen metabolism, internal browning and related enzyme activities in Laiyang pear (Pyrus bretschneideri Reld). LWT Food Sci Technol 72:467–474. https://doi.org/10.1016/j.lwt.2016.05.015
Gao H, Chai H, Cheng N, Cao W (2017) Effects of 24-epibrassinolide on enzymatic browning and antioxidant activity of fresh-cut lotus root slices. Food Chem 217:45–51. https://doi.org/10.1016/j.foodchem.2016.08.063
Griffiths G, Trueman L, Crowther T, Thomas B, Smith B (2002) Onions e a global benefit to health. Phytother Res 16:603e615
Hisaminato H, Murata M, Homma S (2014) Relationship between the enzymatic browning and phenylalanine ammonia-lyase activity of cut lettuce, and the prevention of browning by inhibitors of polyphenol biosynthesis. Biosci Biotechnol Biochem 65(5):1016–1021. https://doi.org/10.1271/bbb.65.1016
Ioannou I, Ghoul M (2013) Prevention of enzymatic browning in fruit and vegetables. Eur Sci J 9:1857–7881. https://doi.org/10.1021/bk-1995-0600.ch004
Jiang J, Jiang L, Luo H, Yu Z (2014) Establishment of a statistical model for browning of fresh-cut lotus root during storage. Postharvest Biol Technol 92:164–171. https://doi.org/10.1016/j.postharvbio.2014.01.025
Kanehisa M, Sato Y, Morishima K (2016) BlastKOALA and GhostKOALA: KEGG tools for functional characterization of genome and metagenome sequences. J Mol Biol 428(4):726–731. https://doi.org/10.1016/j.jmb.2015.11.006
Kim M-J, Kim CY, Park I (2005) Prevention of enzymatic browning of pear by onion extract. Food Chem 89(2):181–184. https://doi.org/10.1016/j.foodchem.2004.02.018
Lante A, Tinello F, Nicoletto M (2016) UV-A light treatment for controlling enzymatic browning of fresh-cut fruits. Innov Food Sci Emerg Technol 34:141–147. https://doi.org/10.1016/j.ifset.2015.12.029
Lee M-K (2007) Inhibitory effect of banana polyphenol oxidase during ripening of banana by onion extract and Maillard reaction products. Food Chem 102(1):146–149. https://doi.org/10.1016/j.foodchem.2006.05.012
Liao Z, Chen R, Chen M, Yang Y, Fu Y, Zhang Q, Lan X (2006) Molecular cloning and characterization of the polyphenol oxidase gene from sweetpotato. Mol Biol 40(6):907–913. https://doi.org/10.1134/s0026893306060094
Lindley MG (1998) The impact of food processing on antioxidants in vegetable oils, fruits and vegetables. Trends Food Sci Technol 9(8–9):336–340. https://doi.org/10.1016/S0924-2244(98)00050-8
Liu X, Lu Y, Yang Q, Yang H, Li Y, Zhou B, Li T, Gao Y, Qiao L (2018) Cod peptides inhibit browning in fresh-cut potato slices: a potential anti-browning agent of random peptides for regulating food properties. Postharvest Biol Technol 146:36–42. https://doi.org/10.1016/j.postharvbio.2018.08.001
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. Embnet Journal 17(1):10–12. https://doi.org/10.14806/ej.17.1.200
Martinez MV, Whitaker JR (1995) The biochemistry and control of enzymatic browning. Trends Food Sci Technol 6(6):195–200. https://doi.org/10.1016/S0924-2244(00)89054-8
McEvily AJ, Iyengar R, Otwell WS (1992) Inhibition of enzymatic browning in foods and beverages. Crit Rev Food Sci Nutr 32(3):253–273. https://doi.org/10.1080/10408399209527599
Mellidou I, Buts K, Hatoum D, Ho QT, Johnston JW, Watkins CB, Schaffer RJ, Gapper NE, Giovannoni JJ, Rudell DR, Hertog ML (2014) Transcriptomic events associated with internal browning of apple during postharvest storage. BMC Plant Biol 14(1):328. https://doi.org/10.1186/s12870-014-0328-x
Murata M, Nishimura M, Murai N, Haruta M, Homma S, Itoh Y (2001) A transgenic apple callus showing reduced polyphenol oxidase activity and lower browning potential. Biosci Biotechnol Biochem 65(2):383–388. https://doi.org/10.1271/bbb.65.383
Negishi O, Ozawa T (2000) Inhibition of enzymatic browning and protection of sulfhydryl enzymes by thiol compounds. Phytochemistry 54(5):481–487. https://doi.org/10.1016/S0031-9422(00)00125-4
Osuga DT, Whitaker JR (1995) Chapter 7. In: Lee CY, Whitaker JR (eds) Enzymatic browning and its prevention; ACS symposium series 600. American Chemical Society, Washington, DC
Park M, Kim G-H (2013) The antioxidative and antibrowning effects of citrus peel extracts on fresh-cut apples. Korean J Food Sci Technol 45(5):598–604. https://doi.org/10.9721/kjfst.2013.45.5.598
Persic M, Mikulic-Petkovsek M, Halbwirth H, Solar A, Veberic R, Slatnar A (2018) Red walnut: characterization of the phenolic profiles, activities and gene expression of selected enzymes related to the phenylpropanoid pathway in pellicle during walnut development. J Agric Food Chem 66(11):2742–2748. https://doi.org/10.1021/acs.jafc.7b05603
Queiroz C, Mendes Lopes ML, Fialho E, Valente-Mesquita VL (2008) Polyphenol oxidase: characteristics and mechanisms of browning control. Food Rev Int 24(4):361–375
Rojas-Graü MA, Sobrino-López A, Soledad Tapia M, Martín-Belloso O (2006) Browning inhibition in fresh-cut ‘Fuji’apple slices by natural antibrowning agents. J Food Sci 71(1):S59-65. https://doi.org/10.1111/j.1365-2621.2006.tb12407.x
Saltveit ME (2004) Effect of 1-methylcyclopropene on phenylpropanoid metabolism, the accumulation of phenolic compounds, and browning of whole and fresh-cut ‘iceberg’ lettuce. Postharvest Biol Technol 34(1):75–80. https://doi.org/10.1016/j.postharvbio.2004.05.001
Saquet AA, Streif J, Bangerth F (2003) Energy metabolism and membrane lipid alterations in relation to brown heart development in ‘Conference’ pears during delayed controlled atmosphere storage. Postharvest Biol Technol 30(2):123–132. https://doi.org/10.1016/s0925-5214(03)00099-1
Suehiro Y, Mochida K, Itamura H, Esumi T (2014) Skin browning and expression of PPO, STS, and CHS genes in the grape berries of ‘Shine Muscat.’ J Jpn Soc Hortic Sci 83(2):122–132. https://doi.org/10.2503/jjshs1.CH-095
Sun D, Liang G, Xie J, Lei X, Mo Y (2010) Improved preservation effects of litchi fruit by combining chitosan coating with ascorbic acid treatment during postharvest storage. Afr J Biotech 9(22):3272–3279
Sun Y, Zhang W, Zeng T, Nie Q, Zhang F, Zhu L (2015) Hydrogen sulfide inhibits enzymatic browning of fresh-cut lotus root slices by regulating phenolic metabolism. Food Chem 177:376–381. https://doi.org/10.1016/j.foodchem.2015.01.065
Tattelman E (2005) Health effects of garlic. Am Fam Phys 72:103–106
Tinello F, Lante A (2018) Recent advances in controlling polyphenol oxidase activity of fruit and vegetable products. Innov Food Sci Emerg Technol 50:73–83. https://doi.org/10.1016/j.ifset.2018.10.008
Tortoe C, Orchard J, Beezer A (2007) Prevention of enzymatic browning of apple cylinders using different solutions. Int J Food Sci Technol 42(12):1475–1481. https://doi.org/10.1111/j.1365-2621.2006.01367.x
Untergasser A, Nijveen H, Rao X, Bisseling T, Geurts R, Leunissen JAM (2007) Primer3Plus, an enhanced web interface to Primer3. Nucleic Acids Res 35(Web Server):W71–W74. https://doi.org/10.1093/nar/gkm306
Wan CY, Wilkins TA (1994) A modified hot borate method significantly enhances the yield of high-quality RNA from cotton (Gossypium hirsutum L.). Anal Biochem 223(1):7–12. https://doi.org/10.1006/abio.1994.1538
Wessels B, Schulze-Kaysers N, Damm S, Kunz B (2014) Effect of selected plant extracts on the inhibition of enzymatic browning in fresh-cut apple. J Appl Bot Food Qual 87:16–23. https://doi.org/10.5073/JABFQ.2014.087.003
Wu Y, Liu F, Yang D-G, Li W, Zhou X-J, Pei X-Y, Liu Y-G, He K-L, Zhang W-S, Ren Z-Y, Zhou K-H, Ma X-F, Li Z-H (2018) Comparative chloroplast genomics of Gossypium species: insights into repeat sequence variations and phylogeny. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00376
Yan S, Li L, He L, Liang L, Li X (2013) Maturity and cooling rate affects browning, polyphenol oxidase activity and gene expression of ‘Yali’ pears during storage. Postharvest Biol Technol 85:39–44. https://doi.org/10.1016/j.postharvbio.2013.04.016
Yang M, Zhu L, Pan C, Xu L, Liu Y, Ke W, Yang P (2015) Transcriptomic analysis of the regulation of rhizome formation in temperate and tropical lotus (Nelumbo nucifera). Sci Rep. https://doi.org/10.1038/srep13059
Zdobnov EM, Apweiler R (2001) InterProScan—an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17(9):847–848. https://doi.org/10.1093/bioinformatics/17.9.847
Zhang S, Yu Y, Xiao C, Wang X, Tian Y (2013) Effect of carbon monoxide on browning of fresh-cut lotus root slice in relation to phenolic metabolism. LWT Food Sci Technol 53(2):555–559. https://doi.org/10.1016/j.lwt.2013.04.001
Zhang S, Tian L, Zhang Y, Zhao H, Zhao J, Guo J, Zhu G-P (2019) De novo transcriptome assembly of the fresh-cut white husk of Juglans cathayensis Dode: Insights for enzymatic browning mechanism of fresh-cut husk of walnut. Sci Hortic. https://doi.org/10.1016/j.scienta.2019.108654
Zhou Y, Dahler JM, Underhill SJ, Wills RB (2003) Enzymes associated with blackheart development in pineapple fruit. Food Chem 80(4):565–572. https://doi.org/10.1016/S0308-8146(02)00375-8
Zhu H, Liu J, Wen Q, Chen M, Wang B, Zhang Q, Xue Z (2017) De novo sequencing and analysis of the transcriptome during the browning of fresh-cut Luffa cylindrica’Fusi-3’fruits. PLoS ONE 12(11):e0187117. https://doi.org/10.1371/journal.pone.0187117
Acknowledgements
The authors would like to thank Ibaraki University, and grants from the NARO Bio-oriented Technology Research Advancement Institution (the special scheme project on regional developing strategy) for supporting throughout this work. Thanks to Dr. Deden Derajat Matra for guidance about bioinformatic analysis. Thanks for Editage service for editing English language.
Funding
This research was supported by grants from the Project of the NARO Bio oriented Technology Research Advancement Institution (the special scheme project on regional developing strategy).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Xingfeng Shao.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Worarad, K., Suzuki, T., Norii, H. et al. Transcriptome profiling for pericarp browning during long-term storage of intact lotus root (Nelumbo nucifera). Plant Growth Regul 95, 207–221 (2021). https://doi.org/10.1007/s10725-021-00736-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00736-2