Abstract
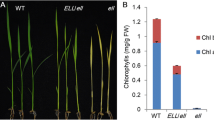
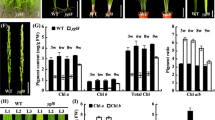
Mg-protoporphyrin IX methyltransferase (CHLM) plays a crucial role in chlorophyll biosynthesis and converts Mg-protoporphyrin IX to Mg-protoporphyrin IX monomethyl ester. Previous research has indicated that down-expression of CHLM causes a pale green phenotype and Mg-protoporphyrin IX accumulation in Arabidopsis thaliana plants. Here, we report that a novel missense mutation (G to A transition) in the CHLM gene, results in the Gly59 → Glu amino acid substitution in the corresponding protein of Arabidopsis chlm-4 mutant plants. In silico modelling studies suggested that this substitution may disrupt membrane association of CHLM. Functional studies showed that the mutant form of CHLM leads to the excessive accumulation of superoxide anion radicals (O2−), although elimination of O2− could alleviate the yellow phenotype observed in chlm-4. In addition, qRT-PCR and western blotting analysis demonstrated that the expression of O2− scavengers, FSD1 and CSD2, were repressed in chlm-4 plants. The accumulation of photosystem II proteins was also decreased due to the CHLM mutation. Furthermore, we found that chlm-4 plants exhibited hypersensitive salt-stress during seed germination and down-expression of the salt stress-responsive CBFs-family genes. These results suggest CHLM positively regulates the expression of multiple photosynthetic-related and stress-responsive nuclear genes.






Similar content being viewed by others
References
Alawady AE, Grimm B (2005) Tobacco Mg protoporphyrin IX methyltransferase is involved in inverse activation of Mg porphyrin and protoheme synthesis. Plant J 41:282–290
Andres-Colas N, Zhu Q, Takenaka M, De Rybel B, Weijers D, Van Der Straeten D (2017) Multiple PPR protein interactions are involved in the RNA editing system in Arabidopsis mitochondria and plastids. Proc Natl Acad Sci USA 114:8883–8888
Ankele E, Kindgren P, Pesquet E, Strand A (2007) In vivo visualization of Mg-protoporphyrin IX, a coordinator of photosynthetic gene expression in the nucleus and the chloroplast. Plant Cell 19:1964–1979
Brzezowski P, Richter AS, Grimm B (2015) Regulation and function of tetrapyrrole biosynthesis in plants and algae. Biochim Biophys Acta 1847:968–985
Cao ZL, Lin GN, Ding WQ, Yang ZN, Cui YL (2010) Mapping and analysis of a yellow mutant chlm-4 of Arabidopsis thaliana (Cruciferae). Acta Bot Yunnanica 32:134–140
Chan Z, Bigelow PJ, Loescher W, Grumet R (2012) Comparison of salt stress resistance genes in transgenic Arabidopsis thaliana indicates that extent of transcriptomic change may not predict secondary phenotypic or fitness effects. Plant Biotechnol J 10:284–300
Chen X, Bao H, Guo J, Jia W, Tai F, Nie L, Jiang P, Feng J, Lv S, Li Y (2014a) Na(+)/H(+) exchanger 1 participates in tobacco disease defence against Phytophthora parasitica var. nicotianae by affecting vacuolar pH and priming the antioxidative system. J Exp Bot 65:6107–6122
Chen X, Wang X, Feng J, Chen Y, Fang Y, Zhao S, Zhao A, Zhang M, Liu L (2014b) Structural insights into the catalytic mechanism of Synechocystis magnesium protoporphyrin IX O-methyltransferase (ChlM). J Biol Chem 289:25690–25698
Chi W, Sun X, Zhang L (2013) Intracellular signaling from plastid to nucleus. Annu Rev Plant Biol 64:559–582
Chu CC, Lee WC, Guo WY, Pan SM, Chen LJ, Li HM, Jinn TL (2005) A copper chaperone for superoxide dismutase that confers three types of copper/zinc superoxide dismutase activity in Arabidopsis. Plant Physiol 139:425–436
Cornah JE, Terry MJ, Smith AG (2003) Green or red: what stops the traffic in the tetrapyrrole pathway? Trends Plant Sci 8:224–230
Davison PA, Schubert HL, Reid JD, Iorg CD, Heroux A, Hill CP, Hunter CN (2005) Structural and biochemical characterization of Gun4 suggests a mechanism for its role in chlorophyll biosynthesis. Biochemistry 44:7603–7612
Dockter C, Volkov A, Bauer C, Polyhach Y, Joly-Lopez Z, Jeschke G, Paulsen H (2009) Refolding of the integral membrane protein light-harvesting complex II monitored by pulse EPR. Proc Natl Acad Sci USA 106:18485–18490
Ferro S, B, Salvi D, Seigneurin-Berny D, Court MML, Ramus C, Miras S, Mellal M, Le Gall S, Kieffer-Jaquinod S, Bruley C, Garin J, Joyard J, Masselon C, Rolland N (2010) AT_CHLORO, a comprehensive chloroplast proteome database with subplastidial localization and curated information on envelope proteins. Mol Cell Proteomics 9:1063–1084
Froehlich JE, Wilkerson CG, Ray W, McAndrew RS OK, Gage DA, Phinney BS (2003) Proteomic study of the Arabidopsis thaliana chloroplastic envelope membrane utilizing alternatives to traditional two-dimensional electrophoresis. J Proteome Res 2:413–425
Gao S, Yang L, Zeng HQ, Zhou ZS, Yang ZM, Li H, Sun D, Xie F, Zhang B (2016) A cotton miRNA is involved in regulation of plant response to salt stress. Sci Rep 6:19736
Halliwell B (2006) Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 141:312–322
Hsieh D, Davis A, Nanda V (2012) A knowledge-based potential highlights unique features of membrane alpha-helical and beta-barrel protein insertion and folding. Protein Sci 21:50–62
Huang C, Yu QB, Yuan XB, Li ZR, Wang J, Ye LS, Xu L, Yang ZN (2015) Rubisco accumulation is important for the greening of the fln2-4 mutant in Arabidopsis. Plant Sci 236:185–194
Kleffmann T, Russenberger D, von Zychlinski A, Christopher W, Sjolander K, Gruissem W, Baginsky S (2004) The Arabidopsis thaliana chloroplast proteome reveals pathway abundance and novel protein functions. Curr Biol 14:354–362
Kuo WY, Huang CH, Liu AC, Cheng CP, Li SH, Chang WC, Weiss C, Azem A, Jinn TL (2013) CHAPERONIN 20 mediates iron superoxide dismutase (FeSOD) activity independent of its co-chaperonin role in Arabidopsis chloroplasts. New Phytol 197:99–110
Li J, Mu J, Bai J, Fu F, Zou T, An F, Zhang J, Jing H, Wang Q, Li Z, Yang S, Zuo J (2013) Paraquat Resistant1, a Golgi-localized putative transporter protein, is involved in intracellular transport of paraquat. Plant Physiol 162:470–483
Mehterov N, Balazadeh S, Hille J, Toneva V, Mueller-Roeber B, Gechev T (2012) Oxidative stress provokes distinct transcriptional responses in the stress-tolerant atr7 and stress-sensitive loh2 Arabidopsis thaliana mutants as revealed by multi-parallel quantitative real-time PCR analysis of ROS marker and antioxidant genes. Plant Physiol Biochem 59:20–29
Moulin M, Smith A (2005) Coenzymology: biochemistry of vitamin biogenesis and cofactor-containing enzymes. Biochem Soc Transact 33:737–742
Myouga F, Hosoda C, Umezawa T, Iizumi H, Kuromori T, Motohashi R, Shono Y, Nagata N, Ikeuchi M, Shinozaki K (2008) A heterocomplex of iron superoxide dismutases defends chloroplast nucleoids against oxidative stress and is essential for chloroplast development in Arabidopsis. Plant Cell 20:3148–3162
Nakashima H, Yoshihara A, Kitamura K (2013) Favorable and unfavorable amino acid residues in water-soluble and transmembrane proteins. J Biomed Sci Eng 06:36–44
Nott A, Jung HS, Koussevitzky S, Chory J (2006) Plastid-to-nucleus retrograde signaling. Annu Rev Plant Biol 57:739
Peltier JB (2002) Central functions of the lumenal and peripheral thylakoid proteome of Arabidopsis determined by experimentation and genome-wide prediction. Plant Cell Online 14:211–236
Pfalz J, Bayraktar OA, Prikryl J, Barkan A (2009) Site-specific binding of a PPR protein defines and stabilizes 5’ and 3’ mRNA termini in chloroplasts. EMBO J 28:2042–2052
Pilon M, Ravet K, Tapken W (2011) The biogenesis and physiological function of chloroplast superoxide dismutases. Biochim Biophys Acta 1807:989–998
Pontier D, Albrieux C, Joyard J, Lagrange T, Block MA (2007) Knock-out of the magnesium protoporphyrin IX methyltransferase gene in Arabidopsis. Effects on chloroplast development and on chloroplast-to-nucleus signaling. J Biol Chem 282:2297–2304
Richter AS, Grimm B (2013) Thiol-based redox control of enzymes involved in the tetrapyrrole biosynthesis pathway in plants. Front Plant Sci 4:371
Richter AS, Wang P, Grimm B (2016) Arabidopsis Mg-protoporphyrin IX methyltransferase activity and redox regulation depend on conserved cysteines. Plant Cell Physiol 57:519–527
Rodriguez-Serrano M, Romero-Puertas MC, Zabalza ANA, Corpas FJ, Gomez M, Del Rio LA, Sandalio LM (2006) Cadmium effect on oxidative metabolism of pea (Pisum sativum L.) roots. Imaging of reactive oxygen species and nitric oxide accumulation in vivo. Plant Cell Environ 29:1532–1544
Sakuraba Y, Balazadeh S, Tanaka R, Mueller-Roeber B, Tanaka A (2012) Overproduction of chl B retards senescence through transcriptional reprogramming in Arabidopsis. Plant Cell Physiol 53:505–517
Singh R, Singh S, Parihar P, Mishra RK, Tripathi DK, Singh VP, Chauhan DK, Prasad SM (2016) Reactive oxygen species (ROS): beneficial companions of plants’ developmental processes. Front Plant Sci 7:1299
Strand Å, Asami T, Alonso J (2003) Chloroplast to nucleus communication triggered by accumulation of Mg-protoporphyrinIX. Nature 421:79–83
Sunkar R, Kapoor A, Zhu JK (2006) Posttranscriptional induction of two Cu/Zn superoxide dismutase genes in Arabidopsis is mediated by downregulation of miR398 and important for oxidative stress tolerance. Plant Cell 18:2051–2065
Tomiyama M, Inoue S, Tsuzuki T, Soda M, Morimoto S, Okigaki Y, Ohishi T, Mochizuki N, Takahashi K, Kinoshita T (2014) Mg-chelatase I subunit 1 and Mg-protoporphyrin IX methyltransferase affect the stomatal aperture in Arabidopsis thaliana. J Plant Res 127:553–563
Xu XF, Wang B, Lou Y, Han WJ, Lu JY, Li DD, Li LG, Zhu J, Yang ZN (2015) Magnesium transporter 5 plays an important role in Mg transport for male gametophyte development in Arabidopsis. Plant J 84:925–936
Yakushevska AE, Keegstra W, Boekema EJ, Dekker JP, Andersson J, Jansson S, Ruban AV, Horton P (2003) The structure of photosystem II in Arabidopsis: localization of the CP26 and CP29 antenna complexes. Biochemistry 42:608–613
Yruela I, Pueyo JJ, Alonso PJ, Picorel R (1996) Photoinhibition of photosystem II from higher plants. J Biol Chem 271:27408–27415
Yu QB, Zhao TT, Ye LS, Cheng L, Wu YQ, Huang C, Yang ZN (2018) pTAC10, an S1-domain-containing component of the transcriptionally active chromosome complex, is essential for plastid gene expression in Arabidopsis thaliana and is phosphorylated by chloroplast-targeted casein kinase II. Photosynth Res 137:69–83
Zhang F, Tang W, Hedtke B, Zhong L, Liu L, Peng L, Lu C, Grimm B, Lin R (2014) Tetrapyrrole biosynthetic enzyme protoporphyrinogen IX oxidase 1 is required for plastid RNA editing. Proc Natl Acad Sci USA 111:2023–2028
Zhang ZW, Zhang GC, Zhu F, Zhang DW, Yuan S (2015) The roles of tetrapyrroles in plastid retrograde signaling and tolerance to environmental stresses. Planta 242:1263–1276
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31900387), China Postdoctoral Science Foundation (2019M652767) and the Youth Fund of Hunan Agricultural University (18QN15).
Author information
Authors and Affiliations
Contributions
CH and LTX drafted the manuscript; MPD provided the bioinformatics analysis; CH, ZLC participated in the experimental design. All authors read and approved the manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, C., David, M.P., Cao, ZL. et al. Mutation of chloroplast CHLM contributes to down-regulation of multiple stress response genes in Arabidopsis. Plant Growth Regul 91, 209–219 (2020). https://doi.org/10.1007/s10725-020-00600-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00600-9