Abstract
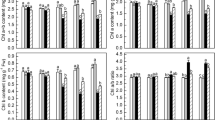
As a precursor of chlorophyll, 5-aminolevulinic acid (ALA) acts as a growth-promoting factor in plants, but little is known of molecular basis of plant growth action. The exogenous supply of an optimal concentration of 75 µM ALA without photooxidative risks, as indicated by a negligible change in conductivity, increased dry shoot biomass of rice up to 16 % after 10 days of ALA treatment, compared to the untreated control. Although rice plants treated with ALA gradually increased levels of ALA-synthesizing capacity during 30 h of the treatment, a noticeable increase in chlorophyll content was not observed. Levels of protoporphyrin IX and Mg–porphyrins were not significantly changed 6 h after ALA treatment and then decreased 30 h after the treatment, while transcript levels of most biosynthetic genes in tetrapyrrole pathway almost kept constant or slightly decreased. In microarray analysis of ALA-treated rice plants, categories of significantly up-regulated transcripts highlight particular biological processes involved in cell cycle, transcription factors, posttranscriptional regulation, and metabolisms of macromolecules. Our results demonstrate that the regulation of tetrapyrrole biosynthesis did not contribute significantly to growth enhancement in ALA-treated rice. We propose that the elevated status of ALA in plant cells alters the transcriptional control of genes involved in physiological processes vital for plant growth, thereby contributing to enhancement of plant biomass.






Similar content being viewed by others
Abbreviations
- ALA:
-
5-Aminolevulinic acid
- Mg–Proto IX:
-
Mg–protoporphyrin IX
- Mg–Proto IX ME:
-
Mg–protoporphyrin IX methyl ester
- Pchlide:
-
Protochlorophyllide
- Proto IX:
-
Protoporphyrin IX
References
Ambawat S, Sharma P, Yadav NR, Yadav RC (2013) MYB transcription factor genes as regulators for plant responses: an overview. Physiol Mol Biol Plants 19:307–321
Ankele E, Kindgren P, Pesquet E, Strand A (2007) In vivo visualization of Mg–protoporphyrin IX, a coordinator of photosynthetic gene expression in the nucleus and the chloroplast. Plant Cell 19:1964–1979
Beale SI (1999) Enzymes of chlorophyll biosynthesis. Photosynth Res 60:43–73
Beale SI, Weinstein JD (1990) Tetrapyrrole metabolism in photosynthetic organisms. In: Daily HA (ed) Biosynthesis of heme and chlorophyll. McGraw-Hill, New York, pp 287–391
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Foyer CH (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875
Franceschini A, Szklarczyk D, Frankild S, Kuhn M, Simonovic M, Roth A, Lin J, Minguez P, Bork P, von Mering C, Jensen LJ (2013) STRING v9.1: protein–protein interaction networks, with increased coverage and integration. Nucleic Acids Res 41:D808–D815
Hara M, Takahashi I, Yamori M, Tanaka T, Funada S, Watanabe K (2011) Effects of 5-aminolevulinic acid on growth and amylase activity in the radish taproot. Plant Growth Regul 64:287–291
Hotta Y, Tanaka T, Takaoka H, Takeuchi Y, Konnai M (1997a) New physiological effects of 5-aminolevulinic acid in plants: the increase of photosynthesis, chlorophyll content, and plant growth. Biosci Biotechnol Biochem 61:2025–2028
Hotta Y, Tanaka T, Takaoka H, Takeuchi Y, Konnai M (1997b) Promotive effects of 5-aminolevulinic acid on the yield of several crops. Plant Growth Regul 22:109–114
Jung S, Yang K, Lee D-E, Back K (2004) Expression of Bradyrhizobium japonicum 5-aminolevulinic acid synthase induces severe photodynamic damage in transgenic rice. Plant Sci 167:789–795
Kim JG, Back K, Lee HY, Lee HJ, Phung TH, Grimm B, Jung S (2014) Increased expression of Fe-chelatase leads to increased metabolic flux into heme and confers protection against photodynamically induced oxidative stress. Plant Mol Biol 86:271–287
Kleine T, Voigt C, Leister D (2009) Plastid signalling to the nucleus: messengers still lost in the mists? Trends Genet 25:185–192
Kobayashi Y, Kanesaki Y, Tanaka A, Kuroiwa H, Kuroiwa T, Tanaka K (2009) Tetrapyrrole signal as a cell-cycle coordinator from organelle to nuclear DNA replication in plant cells. Proc Natl Acad Sci USA 106:803–807
Kotera E, Tasaka M, Shikanai T (2005) A pentatricopeptide repeat protein is essential for RNA editing in chloroplasts. Nature 433:326–330
Koussevitzky S, Nott A, Mockler TC, Hong F, Sachetto-Martins G, Surpin M, Lim J, Mittler R, Chory J (2007) Signals from chloroplasts converge to regulate nuclear gene expression. Science 316:715–719
Lermontova I, Grimm B (2000) Overexpression of plastidic protoporphyrinogen IX oxidase leads to resistance to the diphenyl-ether herbicide acifluorfen. Plant Physiol 122:75–83
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Liu L, Nguyen NT, Ueda A, Saneoka H (2014) Effects of 5-aminolevulinic acid on Swiss chard (Beta vulgaris L. subsp. cicla) seedling growth under saline conditions. Plant Growth Regul 74:219–228
Memon SA, Hou X, Wang L, Li Y (2009) Promotive effect of 5-aminolevulinic acid on chlorophyll, antioxidative enzymes and photosynthesis of Pakchoi (Brassica campestris ssp. chinensis var. communis Tsen et Lee). Acta Physiol Plant 31:51–57
Naeem MS, Jin ZL, Wan GL, Liu D, Liu HB, Yoneyama K, Zhou WJ (2010) 5-Aminolevulinic acid improves photosynthetic gas exchange capacity and ion uptake under salinity stress in oilseed rape (Brassica napus L.). Plant Soil 332:405–415
Papenbrock J, Grimm B (2001) Regulatory network of tetrapyrrole biosynthesis—studies of intracellular signaling involved in metabolic and developmental control of plastids. Planta 213:667–681
Papenbrock J, Mock HP, Kruse E, Grimm B (1999) Expression studies in tetrapyrrole biosynthesis: inverse maxima of magnesium chelatase and ferrochelatase activity during cyclic photoperiods. Planta 208:264–273
Pfannschmidt T (2010) Plastidial retrograde signalling—a true “plastid factor” or just metabolite signatures? Trends Plant Sci 15:427–435
Phung TH, Jung S (2014) Perturbed porphyrin biosynthesis contributes to differential herbicidal symptoms in photodynamically stressed rice (Oryza sativa) treated with 5-aminolevulinic acid and oxyfluorfen. Pestic Biochem Physiol 116:103–110
Phung TH, Jung HI, Park JH, Kim JG, Back K, Jung S (2011) Porphyrin biosynthesis control under water stress: sustained porphyrin status correlates with drought tolerance in transgenic rice. Plant Physiol 157:1746–1764
Ramel F, Birtic S, Ginies C, Soubigou-Taconnat L, Triantaphylidés C, Havaux M (2012) Carotenoid oxidation products are stress signals that mediate gene responses to singlet oxygen in plants. Proc Natl Acad Sci USA 109:5535–5540
Rebeiz CA, Montazer-Zouhool A, Hopen H, Wu SM (1984) Photodynamic herbicides: 1. Concepts and phenomenology. Enzyme Microb Technol 6:390–401
Saeed AI, Sharov V, White J, Li J, Liang W, Bhagabati N, Braisted J, Klapa M, Currier T, Thiagarajan M, Sturn A, Snuffin M, Rezantsev A, Popov D, Ryltsov A, Kostukovich E, Borisovsky I, Liu Z, Vinsavich A, Trush V, Quackenbush J (2003) TM4: a free, open-source system for microarray data management and analysis. Biotechniques 34:374–378
Sasaki K, Marquez F, Nishio N, Nagai S (1995) Promotive effects of 5-aminolevulinic acid on the growth and photosynthesis of Spirulina platensis. J Ferment Bioeng 79:453–457
Schmitz-Linneweber C, Small I (2008) Pentatricopeptide repeat proteins: a socket set for organelle gene expression. Trends Plant Sci 13:663–670
Stirnimann CU, Petsalaki E, Russell RB, Müller CW (2010) WD40 proteins propel cellular networks. Trends Biochem Sci 35:565–574
Strand A, Asami T, Alonso J, Ecker JR, Chory J (2003) Chloroplast to nucleus communication triggered by accumulation of Mg–protoporphyrin IX. Nature 421:79–83
Tanaka R, Tanaka A (2007) Tetrapyrrole biosynthesis in higher plants. Annu Rev Plant Biol 58:321–346
Tanaka Y, Tanaka A, Tsuji H (1993) Effects of 5-aminolevulinic acid on the accumulation of chlorophyll b and apoproteins of the light-harvesting chlorophyll a/b-protein complex of photosystem II. Plant Cell Physiol 34:465–472
Tatusov RL, Galperin MY, Natale DA, Koonin EV (2000) The COG database: a tool for genome-scale analysis of protein functions and evolution. Nucleic Acids Res 28:33–36
Tripathy BC, Chakraborty N (1991) 5-aminolevulinic acid induced photodynamic damage of the photosynthetic electron transport chain of cucumber (Cucumis sativus L.) cotyledons. Plant Physiol 96:761–767
Watanabe K, Tanaka T, Kuramochi H, Takeuchi Y (2000) Improving salt tolerance of cotton seedling with 5-aminolevulinic acid. Plant Growth Regul 32:97–101
Wolf S, Höfte H (2014) Growth control: a saga of cell walls, ROS, and peptide receptors. Plant Cell 26:1848–1856
Workman C, Jensen LJ, Jarmer H, Berka R, Gautier L, Nielser HB, Saxild HH, Nielsen C, Brunak S, Knudsen S (2002) A new non-linear normalization method for reducing variability in DNA microarray experiments. Genome Biol 3:1–16
Xie SS, Wu HJ, Zang HY, Wu LM, Zhu QQ, Gao XW (2014) Plant growth promotion by spermidine-producing Bacillus subtilis OKB105. Mol Plant Microbe Interact 27:655–663
Zhang ZJ, Li HZ, Zhou WJ, Takeuchi Y, Yoneyama K (2006) Effect of 5-aminolevulinic acid on development and salt tolerance of potato (Solanum tuberosum L.) microtubers in vitro. Plant Growth Regul 49:27–34
Acknowledgments
This work was supported by National Research Foundation of Korea Grant funded by the Korean Government (Ministry of Education, Science and Technology) (NRF-2009-0076123 and NRF-2010-0005635).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nguyen, H., Kim, HS. & Jung, S. Altered tetrapyrrole metabolism and transcriptome during growth-promoting actions in rice plants treated with 5-aminolevulinic acid. Plant Growth Regul 78, 133–144 (2016). https://doi.org/10.1007/s10725-015-0080-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-015-0080-8