Abstract
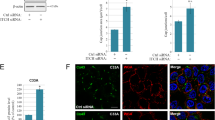
The human E-cadherin is a single transmembrane domain protein involved in Ca2+-dependent cell–cell adhesion. In a previous study, we demonstrated that all of four potential N-glycosylation sites in E-cadherin are occupied by N-glycans in human breast carcinoma cells in vivo and the elimination of N-glycan at Asn-633 dramatically affected E-cadherin expression and made it degraded. In this study we investigated the molecular mechanism of E-cadherin, which lacks N-glycosylation at Asn-633 (M4), degradation and the role of the N-glycan at Asn-633 in E-cadherin folding. We treated cells stably expressed M4 E-cadherin with MG123, DMM, respectively. Either MG132 or DMM could efficiently block degradation of M4 E-cadherin. M4 E-cadherin was recognized as the substrate of ERAD and was retro-translocated from ER lumen to cytoplasm by p97. It was observed that the ration of M4 E-cadherin binding to calnexin was significantly increased compared with that of other variants, suggesting that it was a misfolded protein, though cytoplasmic domain of M4 E-cadherin could associate with β-catenin. Furthermore, we found that N-glycans of M4 E-cadherin were modified in immature high mannose type, suggesting that it could not depart to Golgi apparatus. In conclusion, this study revealed that N-glycosylation at Asn-633 is essential for E-cadherin expression, folding and trafficking.









Similar content being viewed by others
Abbreviations
- EC:
-
extracellular domain
- AJs:
-
adherence junction complexes
- ER:
-
endoplasmic reticulum
- ERAD:
-
endoplasmic reticulum-associated degradation
- WT:
-
wild type E-cadherin
- M1:
-
E-cadherin lacking N-glycan at Asn-554 site
- M2:
-
E-cadherin lacking N-glycan at Asn-566 site
- M3:
-
E-cadherin lacking N-glycan at Asn-618 site
- M4:
-
E-cadherin lacking N-glycan at Asn-633 site
- M123:
-
E-cadherin has a single N-glycan at Asn-633 site
- Endo H:
-
endoglycosidase
- PNGase F:
-
peptide N-glycosidase F
- PBS:
-
phosphate-buffered saline
References
Takeichi, M.: Cadherin cell adhesion receptors as a morphogenetic regulator. Science 251, 1451–1455 (1991)
Kemler, R.: From cadherins to catenins: cytoplasmic protein interactions and regulation of cell adhesion. Trends Genet. 9, 317–321 (1993)
Pokutta, S., Herrenknecht, K., Kemler, R., Engel, J.: Conformational changes of the recombinant extracellular domain of E-cadherin upon calcium binding. Eur. J. Biochem. 223, 1019–1026 (1994)
Ozawa, M., Engel, J., Kemler, R.: Single amino acid substitutions in one Ca2+ binding site of uvomorulin abolish the adhesive function. Cell 63, 1033–1038 (1990)
Kemler, R., Ozawa, M.: Uvomorulin–catenin complex: cytoplasmic anchorage of a calcium-dependent cell adhesion molecule. Bioessays 11, 88–91 (1989)
Ozawa, M.R., Ringwald, M., Kemler, R.: Uvomorulin–catenin complex formation is regulated by a specific domain in the cytoplasmic region of the cell adhesion molecule. Proc. Natl. Acad. Sci. U. S. A. 87, 4246–4250 (1990)
Reynolds, A.B., Daniel, J., McCrea, P.D., Wheelock, M.J., Wu, J., Zhang, Z.: Identification of a new catenin: the tyrosine kinase substrate p120cas associates with E-cadherin complexes. Mol. Cell. Biol. 14, 8333–8342 (1994)
Hinck, L., Näthke, I.S., Papkoff, J., Nelson, W.J.: Dynamics of cadherin/catenin complex formation novel: protein interactions and pathways of complex assembly. J. Cell Biol. 125, 1327–1340 (1994)
Wheelock, M.J., Johnson, K.R.: Cadherin-mediated cellular signaling. Curr. Opin. Cell Biol. 15, 509–514 (2003)
Thoreson, M.A., Anastasiadis, P.Z., Daniel, J.M., Ireton, R.C., Wheelock, M.J., Johnson, K.R., Hummingbird, D.K., Reynolds, A.B.: Selective uncoupling of p120ctn from E-cadherin disrupts strong adhesion. J. Cell Biol. 148, 189–201 (2000)
Ozawa, M., Ohkubo, T.: Tyrosine phosphorylation of p120(ctn) in v-Src transfected L cells depends on its association with E-cadherin and reduces adhesion activity. J. Cell Sci. 114, (3), 503–512 (2001)
Nose, A., Tsuji, K., Takeichi, M.: Localization of specificity determining sites in cadherin cell adhesion molecules. Cell 61, (1), 147–155 (1990)
Nagar, B., Overduin, M., Ikura, M., Rini, J.M.: Structural basis of calcium-induced E-cadherin rigidification and dimerization. Nature 380, 360–364 (1996)
Ozawa, M.: Lateral dimerization of the E-cadherin extracellular domain is necessary but not sufficient for adhesive activity. J. Biol. Chem. 227, 19600–19608 (2002)
Yoshimura, M., Ihara, Y., Masuzawa, Y., Taniguchi, N.: Aberrant glycosylation of E-cadherin enhances cell–cell binding to suppress metastasis. J. Biol. Chem. 271, 13811–13815 (1996)
Kitada, T., Miyoshi, E., Noda, K., Higashiyama, S., Ihara, H., Matsuura, N., Hayashi, N., Kawata, S., Matsuzawa, Y., Taniguchi, N.: The addition of bisecting N-acetylglucosamine residues to E-cadherin down-regulates the tyrosine phosphorylation of b-catenin. J. Biol. Chem. 276, 475–480 (2000)
Liwosz, A., Lei, T., Kukuruzinska, M.A.: N-glycosylation affects the molecular organization and stability of E-cadherin junctions. J. Biol. Chem. 281, 23138–23149 (2006)
Parodi, A.J.: Protenin glucosylation and its role in protein folding. Annu. Rev. Biochem. 69, 69–93 (2000)
Tokunaga, F., Brostrom, C., Koide, T., Arvan, P.: Endoplasmic reticulum (ER)-associated degradation of misfolded N-linked glycoproteins is suppressed upon inhibitor of ER mannosidase I. J. Biol. Chem. 275, 40757–40764 (2000)
Hirsh, C., Jarosch, E., Sommer, T., Wolf, D.H.: Endoplasmic reticulum-associated protein degradation—one model fits all? Biochim. Biophys. Acta 1695, 208–216 (2004)
Ruddock, L.W., Molinari, M.: N-glycan processing in ER quality control. J. Cell Sci. 119, 4373–4380 (2006)
Kleizen, B., Braakman, L.: Protein folding and quality control in the endoplasmic reticulum. Curr. Opin. Cell Biol. 16, 343–349 (2004)
Nishkawa, S., Brodsky, J.L., Nakatsukasa, K.: Roles of molecular chaperones in endoplasmic reticulum (ER) quality control and ER-associated degradation(ERAD). J. Biochem. 137, 551–555 (2005)
Lee, M.C., Miller, E.A., Goldberg, J., Orci, L., Schekman, R.: Bi-directional protein transport between the ER and Golgi. Annu. Rev. Cell Dev. Biol. 20, 87–123 (2004)
Watanabe, R., Riezman, H.: Differential ER exit in yeast and mammalian cells. Curr. Opin. Cell Biol. 16, 350–355 (2004)
Ritter, C., Helenius, A.: Recognition of local glycoprotein misfolding by the ER folding sensor UDP-glucose:glycoprotein glucosyltransferase. Nat. Struct. Biol. 7, 278–280 (2000)
Taylor, S.C., Ferguson, A.D., Bergeron, J.J.M., Thomas, D.Y.: The ER protein folding sensor UDP-glucose glycoprotein-glucosyltransferase modifies substrates distant to local changes in glycoprotein conformation. Nat. Struct. Mol. Biol. 11, 128–134 (2004)
Hosokawa, N., Tremblay, L.O., You, Z., Herscovics, A., Wada, I., Nagata, K.: Enhancement of endoplasmic reticulum (ER) degradation of misfolded Null Hong Kong a1-antitrypsin by human ER mannosidase I. J. Biol. Chem. 278, 26287–26294 (2003)
Frenkel, Z., Gregory, W., Kornfeld, S., Lederkremer, G.Z.: Endoplasmic reticulum-associated degradation of mammalian glycoproteins involves sugar chain trimming to Man6-5GlcNAc2. J. Biol. Chem. 278, 34119–34124 (2003)
Hitt, R., Wolf, D.H.: DER7, encoding a-glucosidase I is essential for degradation of malfolded glycoproteins of the endoplasmic reticulum. FEMS Yeast Research 4, 815–820 (2004)
Mast, S.W., Diekman, K., Karaveg, K., Davis, A., Sifers, R.N., Moremen, K.W.: Human EDEM2, a novel homolog of family 47 glycosidases, is involved in ER-associated degradation of glycoproteins. Glycobiology 15, 421–436 (2005)
Hirao, K., Natsuka, Y., Tamura, T., Wada, I., Morito, D., Natsuka, S., Romero, P., Sleno, B., Tremblay, L.O.: EDEM3, a soluble EDEM homolog, enhances glycoprotein ERAD and mannose trimming. J. Biol. Chem. 281, 9650–9658 (2006)
Spiro, R.G.: Role of N-linked polymannose oligosaccharides in targeting glycoproteins for endoplasmic reticulum-associated degradation. Cell. Mol. Life Sci. 61, 1025–1041 (2004)
Lederkremer, G.Z., Glickman, M.H.: A window of opportunity: timing protein degradation by trimming of sugars and ubiquitins. Trends Biochem. Sci. 30, 297–303 (2005)
Rabinovich, E., Kerem, A., Fröhlich, K.-U., Diamant, N., Bar-Nun, S.: AAA-ATPase p97/Cdc48p, a cytosolic chaperone required for endoplasmic reticulum-associated protein degradation. Mol. Cell. Biol. 22, 626–634 (2002)
Ye, Y., Shibata, Y., Yun, C., Ron, D., Rapoport, T.A.: A membrane protein complex mediates retro-translocation from the ER lumen into the cytosol. Nature 429, 841–847 (2004)
Richly, H., Rape, M., Braun, S., Rumpf, S., Hoege, C., Jentsch, S.: A series of ubiquitin binding factors connects CDC48/p97 to substrate multiubiquitylation and proteasomal targeting. Cell 120, 73–84 (2005)
Zhao, H.B., Liang, Y.L., Xu, Z.B., Wang, L.Y., Zhou, F., Li, Z.X., Jin, J.W., Yang, Y., Fang, Z.Y., Hu, Y.L., Zhang, L.N., Su, J.M., Zha, X.L.: N-glycosylation affects the adhesive function of E-cadherin through modifying the composition of adherens junctions (AJs) in human breast carcinoma cell line MDA-MB-435. J. Cell. Biochem. 104(1), 162–175 (2008)
Wang, L., Liang, Y., Li, X., Cai, X., Zhang, W., Wu, G., Jin, J., Fang, Z., Zha, V.: Increase in beta1-6 GlcNAc branching caused by N-acetylglucosaminyltransferase V directs integrin beta1 stability in human hepatocellular carcinoma cell line SMMC-7721. J. Cell. Biochem. 100, 230–241 (2007)
Yoshida, H.: ER stress and diseases. FEBS 274, 630–658 (2007)
Wójcik, C., Rowicka, M., Kudlicki, A., Nowis, D., McConnel, E., Kujawa, M., DeMartino, G.N.: VCP (valosin-containing protein, p97) is a regulator of ER stress and of the degradation of N-end rule and UFD (ubiquitin-fusion degradation) pathway substrates in mammalian cells. Mol. Cell. Biol. 17, 4606–4618 (2006)
Chen, Y.-T., Stewart, D.B., Nelson, W.J.: Coupling assembly of the E-cadherin/b-catenin complex to efficient endoplasmic reticulum exit and basal–lateral membrane targeting of E-cadherin in polarized MDCK cells. J. Cell Biol. 144, 687–699 (1999)
Kyte, J., Doolittle, R.F.: A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132 (1982)
Marambaud, P., Shioi, J., Serban, G., Georgakopoulos, A., Sarner, S., Nagy, V., Baki, L., Wen, P., Efthimiopoulos, S., Shao, Z., Wisniewski, T., Robakis, N.K.: A presenilin-1/g-secretase cleavage releases the E-cadherin intracellular domain and regulates disassembly of adherens junctions. EMBO. J. 21, 1948–1956 (2001)
Kim, D.Y., Ingano, M.L.A., Kovacs, D.M.: Nectin-1a, an immunoglobulin-like receptor involved in the formation of synapses, is a substrate for presenilin/g-secretase-like cleavage. J. Biol. Chem. 277, 49976–49981 (2002)
Häussinger, D., Ahrens, T., Aberle, T., Engel, J., Stetefeld, J., Grzesiek, S.: Proteolytic E-cadherin activation followed by solution NMR and X-ray crystallography. EMBO. J. 23, 1699–1708 (2004)
Le, T.L., Yap, A.S., Stow, J.L.: Recycling of E-cadherin: a potential mechanism for regulating cadherin dynamics. J. Cell Biol. 146, 219–232 (1999)
Fujita, Y., Krause, G., Scheffner, M., Zechnner, D., Leddy, H.E., Behrens, J., Sommer, T., Birchmeier, W.: Hakai, a c-CbI-like protein, ubiquitinates and induces endocytosis of the E-cadherin complex. Nature Cell Biol. 4, 222–231 (2002)
Ioannou, Y.A., Zeidner, K.M., Grace, M.E., Desnick, R.J.: Human a-galactosidase A: glycosylation site 3 is essential for enzyme solubility. Biochem. J. 332, 789–797 (1998)
Isaji, T., Sato, Y., Zhao, Y., Miyoshi, E., Wada, Y., Taniguchi, N., Gu, J.: N-glycosylation of the b-propeller domain of the integrin a5 subunit is essential for a5b1 heterodimerization, expression on the cell surface and its biological function. J. Biol. Chem. 281, 33258–33267 (2006)
Kane, S.: Mouse procathepsin L lacking a functional glycosylation site is properly folded, stable, and secreted by NIH 3T3 cells. J. Biol. Chem. 268, (15), 11456–11462 (1993)
Rohl, C.A., Strauss, C.E.M., Chivian, D., Baker, D.: Modeling structurally variable regions in homologous proteins with Rosetta. Proteins 55, 656–677 (2004)
Wang, X., Matteson, J., An, Y., Moyer, B., Yoo, J.-S., Bannykh, S., Wilson, L.A., Riordan, J.R., Balch, W.E.: COPII-dependent export of cystic fibrosis transmembrane conductance regulator from the ER uses a di-acidic exit code. J. Cell Biol. 167, 65–74 (2004)
Le, A., Graham, K.S., Sifers, R.N.: Intracellular degradation of the transport-impaired human PiZ alpha 1-antitrypsin variant. Biochemical mapping of the degradative event among compartments of the secretory pathway. J. Biol. Chem. 265, 14001–14007 (1990)
Caldwell, S.R., Hill, K.J., Cooper, A.A.: Degradation of endoplasmic reticulum (ER) quality control substrates requires transport between the ER and Golgi. J. Biol. Chem. 276, 23296–23303 (2001)
Vashist, S., Ng, D.T.W.: Misfolded proteins are sorted by a sequential checkpoint mechanism of ER quality control. J. Cell Biol. 165, 41–52 (2004)
Kincaid, M.M., Copper, A.A.: Misfolded proteins traffic from the endoplasmic reticulum (ER) due to ER export signals. Mol. Biol. Cell 18, 455–463 (2007)
Acknowledgements
This work was supported by Shanghai Leading Academic Disciplines Project, Project Number: B110, National Nature Science Foundation of China, No 30670468 and the Grant of Shanghai Health Bureau, No. 044083.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, F., Su, J., Fu, L. et al. Unglycosylation at Asn-633 made extracellular domain of E-cadherin folded incorrectly and arrested in endoplasmic reticulum, then sequentially degraded by ERAD. Glycoconj J 25, 727–740 (2008). https://doi.org/10.1007/s10719-008-9133-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-008-9133-9