Abstract
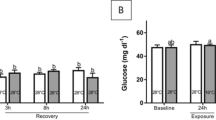
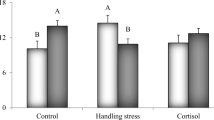
Stress is an energy-demanding process, as well as the responses of the innate immune system, that impose a metabolic overload on cellular energy production, which can affect the cellular redox balance, causing oxidative damage. We evaluated the role of stress in the modulation of innate immune and oxidative/antioxidant mechanisms in juvenile pacu exposed to acute and chronic stressors. The experimental period lasted 30 days, and fish (113.7 ± 35.1 g) were fed commercial feed. During this period, half of the fish were not manipulated (Condition A), and the other half were chased with a dip net for 5 min twice a day (Condition C). After the 30-day period, fish from both groups were sampled (baseline sampling), and the remainders (not sampled) were air exposed for 3 min (acute stressor), returned to the tanks, and were sampled again 30 min, 3 h, 6 h, and 24 h after air exposure. We evaluated biomarkers of stress (circulating cortisol and glucose), the innate immune system (respiratory burst activity/RBA, hemolytic activity of the complement system (HA-AP) and serum concentration of lysozyme), oxidative damage (lipid peroxidation/LPO), and antioxidant enzymes (superoxide dismutase, SOD; catalase, CAT; and glutathione peroxidase, GSH-Px). Our results showed that stress, acutely or chronically, caused a transient reduction of RAL and activated the HA-AP. Acutely, stress increased the lysozyme concentration. Furthermore, both conditions caused oxidative stress in the liver, and differently they modulated the antioxidant system, enhancing SOD activity and impairing CAT and GSH-Px activity.




Similar content being viewed by others
Data Availability
All relevant data are within the paper, and those are available at the corresponding author.
References
Aebi H (1984) Catalase in Vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Anderson DP, Siwicki AK (1995) Basic haematology and serology for fish health programs. In: Shariff M, Arthur JR, Subasinghe RP (eds) Diseases in Asian Aquaculture II. Manila: Fish Health Section, Asian Fish Soc, pp 185–202
Ardiansyah, Fotedar R (2016) Water quality, growth and stress responses of juvenile barramundi (Lates calcarifer Bloch), reared at four different densities in integrated recirculating aquaculture systems. Aquaculture 458:113–120. https://doi.org/10.1016/j.aquaculture.2016.03.001
Aschbacher K, O'Donovan A, Wolkowitz OM, Dhabhar FS, Su Y, Epel E (2013) Good stress, bad stress and oxidative stress: Insights from anticipatory cortisol reactivity. Psychoneuroendocrinol. 38:1698–1708. https://doi.org/10.1016/j.psyneuen.2013.02.004
Barton BA, Morgan JD, Vijayan MM (2002) Physiological and condition-related indicators of environmental stress in fish. In: Adams SM (ed) Biological Indicators of Aquatic Ecosystem Stress. American Fisheries Society, Bethesda, pp 111–148
Beutler E, Durom O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–890
Biller-Takahashi JD, Takahashi LS, Saita MV, Gimbo RY, Urbinati EC (2013) Leucocytes respiratory burst activity as indicator of innate immunity of pacu Piaractus mesopotamicus. Braz J Biol 73:425–429. https://doi.org/10.1590/S1519-69842013000200026
Birnie-Gauvin K, Costantini D, Cooke SJ, Willmore WJ (2017) A comparative and evolutionary approach to oxidative stress in fish: a review. Fish and Fish 18:928–942. https://doi.org/10.1111/faf.12215
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Camejo G, Wallin B, Enojärvi M (1998) Analysis of oxidation and antioxidants using microtiter plates. Methods Mol Biol 108:377–387. https://doi.org/10.1385/0-89603-472-0:377
Camougrand N, Rigoulet M (2001) Aging and oxidative stress: studies of some genes involved both in aging and in response to oxidative stress. Respir Physiol 15:393–401. https://doi.org/10.1016/S0034-5687(01)00314-0
Ceriello A (2000) Oxidative stress and glycemic regulation. Metabolism 49:27–29. https://doi.org/10.1016/S0026-0495(00)80082-7
Chadzinska M, Tertil E, Kepka M, Hermsen T, Scheer M, Verburg-Van Kemenade BM (2012) Adrenergic regulation of the innate immune response in common carp (Cyprinus carpio L.). Dev Comp Immunol 36:306–316. https://doi.org/10.1016/j.dci.2011.04.010
Clotfelter ED, Lapidus SJH, Brown AC (2013) The effects of temperature and dissolved oxygen on antioxidant defences and oxidative damage in the fathead minnow Pimephales promelas. J Fish Biol 82:1086–1092. https://doi.org/10.1111/jfb.12050
Demers NE, Bayne CJ (1997) The immediate effects of stress on hormones and plasma lysozyme in rainbow trout. Dev Comp Immunol 21:363–373. https://doi.org/10.1016/S0145-305X(97)00009-8
Du J, Wang Y, Hunter R, Wei Y, Blumenthal R, Falke C, Khairova R, Zhou R, Yuan P, Machado-Vieira R, Mc Ewen BS, Manji HK (2009) Dynamic regulation of mitochondrial function by glucocorticoids. PNAS 106:3543–3548. https://doi.org/10.1073/pnas.0812671106
Du F, Xu G, Gao J, Nie Z, Xu P, Gu R (2016) Transport-induced changes in hypothalamic–pituitary–interrenal axis gene expression and oxidative stress responses in Coilia nasus. Aquac Res 46:3599–3607. https://doi.org/10.1111/are.12812
Elabd H, Wang H-P, Shaheen A, Yao H, Abbass A (2017) Anti-oxidative effects of some dietary supplements on Yellow perch (Perca flavescens) exposed to different physical stressors. Aquacult Rep 8:21–30. https://doi.org/10.1016/j.aqrep.2017.09.002
Eslamloo K, Akhavan SR, Fallah FJ, Henry MA (2014) Variations of physiological and innate immunological responses in goldfish (Carassius auratus) subjected to recurrent acute stress. Fish Shellfish Immunol 37:147–153. https://doi.org/10.1016/j.fsi.2014.01.014
Filiciotto F, Cecchini S, Buscaino G, Mac Carrone V, Piccione G, Fazio F (2017) Impact of aquatic acoustic noise on oxidative status and some immune parameters in gilthead sea bream Sparus aurata (Linnaeus, 1758) juveniles. Aquacult Res 48:1895–1903. https://doi.org/10.1111/are.13027
Gimbo RY, Favero GC, Franco Montoya NL, Urbinati EC (2015) Energy deficit does not affect immune responses of experimentally infected pacu (Piaractus mesopotamicus). Fish Shellfish Immunol 43:295–300. https://doi.org/10.1016/j.fsi.2015.01.005
Hafeman DG, Sunde RA, Hoekstra WC (1974) Effect of dietary selenium on erythrocyte and liver glutathione peroxidase in the rat. J Nutr 104:580–587. https://doi.org/10.1093/jn/104.5.580
Harris J, Bird DJ (2000) Modulation of the fish immune system by hormones. Vet Immunol Immunopathol 77:163–176. https://doi.org/10.1016/S0165-2427(00)00235-X
Hermenegildo C, Raya A, Roma J, Romero FJ (1993) Decreased glutathione peroxidase activity in sciatic nerve of alloxan-induced diabetic mice and its correlation with blood glucose levels. Neurochem Res 18:893–896. https://doi.org/10.1007/BF00998274
Jensen F, Nielsen M, Nielsen R (2014) Increased competition for aquaculture from fisheries: does improved fisheries management limit aquaculture growth? Fish Res 159:25–33. https://doi.org/10.1016/j.fishres.2014.05.004
Liu B, Liu Y, Wang X (2015) The effect of stocking density on growth and seven physiological parameters with assessment of their potential as stress response indicators for the Atlantic salmon (Salmo salar). Mar Freshw Behav Physiol 48:177–192. https://doi.org/10.1080/10236244.2015.1034956
Lushchak VI (2016) Contaminant-induced oxidative stress in fish: a mechanistic approach. Fish Physiol Biochem 42:711–747. https://doi.org/10.1007/s10695-015-0171-5
Mccord JE, Fridovich I (1969) Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244:6049–6055
Mommsen TP, Vijayan MM, Moon TW (1999) Cortisol in teleosts: dynamics, mechanisms of action, and metabolic regulation. Rev Fish Biol Fish 9:211–268. https://doi.org/10.1023/A:1008924418720
Nardocci G, Navarro C, Cortés PP, Imarai M, Montoya M, Valenzuela B, Jara P, Acuña-Castillo P, Fernández R (2014) Neuroendocrine mechanisms for immune system regulation during stress in fish. Fish Shellfish Immunol 40:531–538. https://doi.org/10.1016/j.fsi.2014.08.001
Nimse SB, Pal D (2015) Free radicals, natural antioxidants, and their reaction mechanisms. RSC Advances 5:27986–28006
Ramos Filho MM, Ramos MIL, Hiane PA, Souza EMT (2008) Perfil lipídico de quatro espécies de peixes da região pantaneira de Mato Grosso do Sul. Ciênc Tecnol Aliment 2:361–365. https://doi.org/10.1590/S0101-20612008000200014
Robinson JM (2008) Reactive oxygen species in phagocytic leukocytes. Histochem Cell Biol 130:281–297. https://doi.org/10.1007/s00418-008-0461-4
Roy B, Rai U (2009) Genomic and non-genomic effect of cortisol on phagocytosis in freshwater teleost Channa punctatus: an in vitro study. Steroids 74:449–455. https://doi.org/10.1016/j.steroids.2008.12.013
Secombes CJ (1996) The nonspecific immune system: cellular defences. In: Iwama G, Nakanishi T (eds) The fish immune system: organism, pathogen and environment. Academic Press, San Diego, pp 63–103
Secombes CJ, Wang T (2012) The innate and adaptive immune system of fish. In: Austin B (ed) Infectious disease in aquaculture: prevention and control. Woodhead Publishing Series in Food Science, Technology and Nutrition, pp 3–68
Sierra-Flores R, Atack T, Migaud H, Davie A (2015) Stress response to anthropogenic noise in Atlantic cod Gadus morhua L. Aquacult Eng 67:67–76. https://doi.org/10.1016/j.aquaeng.2015.06.003
Tort L (2011) Stress and immune modulation in fish. Dev Comp Immunol 35:1366–1375. https://doi.org/10.1016/j.dci.2011.07.002
Urbinati EC, Takahashi LS (2020) Pacu (Piaractus mesopotamicus). In: Espécies nativas para pisciculture no Brasil. Baldisserotto, B (Ed.). Editora UFSM, 169-191.
Valladão GMR, Gallani SU, Pilarski F (2018) South American fish for continental aquaculture. Rev Aquacult 10:351–369. https://doi.org/10.1111/raq.12164
Welker TL, Congleton JL (2004) Oxidative stress in juvenile chinook salmon, Oncorhynchus tshawytscha (Walbaum). Aquacult Res 35:881–887. https://doi.org/10.1111/j.1365-2109.2004.01080.x
Wendelaar Bonga SE (1997) The stress response in fish. Physiol Rev 77:591–625. https://doi.org/10.1152/physrev.1997.77.3.591
Zanuzzo FS, Zaiden SF, Senhorini JA, Marzocchi-Machado CM, Urbinati EC (2015) Aloe vera bath improved physical and humoral protection in breeding stock of matrinxã (Brycon amazonicus) after induced spawning. Fish Shellfish Immunol 45:132–140. https://doi.org/10.1016/j.fsi.2015.02.017
Zanuzzo FS, Sabioni RE, Montoya LNF, Favero G, Urbinati EC (2017) Aloe vera enhances the innate immune response of pacu (Piaractus mesopotamicus) after transport stress and combined heat killed Aeromonas hydrophila infection. Fish Shellfish Immunol 65:198–205. https://doi.org/10.1016/j.fsi.2017.04.013
Zanuzzo FS, Sabioni RE, Marzocchi-Machado CM, Urbinati EC (2019) Modulation of stress and innate immune response by corticosteroids in pacu (Piaractus mesopotamicus). Comp Biochem Physiol Part A 231:39–48. https://doi.org/10.1016/j.cbpa.2019.01.019
Code availability
Not applicable.
Funding
This research was granted by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Grant number 130587/2015-0).
Author information
Authors and Affiliations
Contributions
The experimental trials were conducted by C.F.P.F. and T.P.M. and supervised by E.C.U. Assays and data analysis were performed by C.F.P.F. with the help from E.C.U. and C.B.R.M. M.M.M.M and L.S.T. helped in the statistical analysis. The manuscript was written or revised by all authors.
Corresponding author
Ethics declarations
Ethics approval
The experimental procedures were conducted in accordance with the Brazilian Law on Experimental Animals and were approved by the Animal Ethics and Welfare Committee of the Universidade Estadual Paulista (UNESP) (number 2092/16), Jaboticabal campus.
Conflict of interest
The authors declare no competing interests.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Fátima Pereira de Faria, C., dos Reis Martinez, C.B., Takahashi, L.S. et al. Modulation of the innate immune response, antioxidant system and oxidative stress during acute and chronic stress in pacu (Piaractus mesopotamicus). Fish Physiol Biochem 47, 895–905 (2021). https://doi.org/10.1007/s10695-021-00940-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-00940-x