Abstract
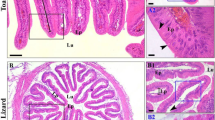
The main purpose of this study was to investigate the distribution of acid phosphatase (ACP), alkaline phosphatase (ALP), non-specific esterase (NSE), peroxidase (POD), and mucous cells in the intestine of the koi carp Cyprinus carpio var. koi. ACP activity was located in the striated border, enterocytes, and lamina propria of the anterior and middle intestines. The ACP activity in the anterior intestine was higher than that in the middle and posterior intestines. ALP existed in the striated border of enterocytes and lamina propria, serosa, muscular layer, and the junction between muscular layer and submucosa layer of the intestine. The ALP activity in the anterior intestine was higher than that in the middle and posterior intestines. NSE activity was localized in the cytoplasm of enterocytes in the whole intestine, and the middle intestine showed the lower NSE activity than the anterior and posterior intestines. POD activity was localized in the blood cells of the lamina propria and cytoplasm of enterocytes in all intestinal segments. The POD activity among the anterior, middle, and posterior intestines was non-significantly different. Alcian blue periodic acid–Schiff histochemical results revealed three types of mucous cells in the intestine. The total number of mucous cells and percentage of type I cells among the anterior, middle, and posterior intestines were non-significantly different. The percentage of the type II cells was the highest in the posterior intestine, while the lowest in the anterior intestine. The percentage of the type III cells was the highest in the anterior intestine, while the lowest in the posterior intestine.






Similar content being viewed by others
References
Akster HA (1985) Morphometry of muscle fibre types in the carp (Cyprinus carpio L.). Relationships between structural and contractile characteristics. Cell Tissue Res 241:193–201. https://doi.org/10.1007/BF00214641
Ben CE, Li SG (2001) Histochemistry. In: Pu YJ (ed) Hydrolase, 1st edn. the People’s Hygiene Press, Beijing, pp 271–364
Boonzaier J, Van der Merwe EL, Bennett NC, Kotzé SH (2013) A comparative histochemical study of the distribution of mucins in the gastrointestinal tracts of three insectivorous mammals. Acta Histochem 15:549–556. https://doi.org/10.1016/j.acthis.2012.12.003
Cao XJ, Wang WM (2009) Histology and mucin histochemistry of the digestive tract of yellow catfish, Pelteobagrus fulvidraco. Anat Histol Embryol 38:254–261. https://doi.org/10.1111/j.1439-0264.2009.00932.x
Cardoso N, Firmiano EM, Gomes ID, do Nascimento AA, Sales A, Araújo FG (2015) Histochemical and immunohistochemical study on endocrine cells (5HT, GAS, and SST) of the gastrointestinal tract of a teleost, the characin Astyanax bimaculatus. Acta Histochem 117:595–604. https://doi.org/10.1016/j.acthis.2015.05.007
Chakrabarti I, Gani MA, Chaki KK, Sur R, Mirsa KK (1995) Digestive enzymes in 11 freshwater teleost fish species in relation to food habit and niche segregation. Comp Biochem Phys A 112:167–177. https://doi.org/10.1016/0300-9629(95)00072-F
Díaz AO, García AM, Goldemberg AL (2008) Glycoconjugates in the mucosa of the digestive tract of Cynoscion guatucupa: a histochemical study. Acta Histochem 110:76–85. https://doi.org/10.1016/j.acthis.2007.08.002
Domeneghini C, Arrighi S, Radaelli G, Bosi G, Veggetti A (2005) Histochemical analysis of glycoconjugate secretion in the alimentary canal of Anguilla anguilla L. Acta Histochem 106:477–487. https://doi.org/10.1016/j.acthis.2004.07.007
Faggio C, Torre A, Lando G, Sabatino G, Trischitta F (2011) Carbonate precipitates and bicarbonate secretion in the intestine of sea bass, Dicentrarchus labrax. J Comp Physiol B 181:517–525. https://doi.org/10.1007/s00360-010-0538-y
Gajger IT, Nejedli S, Kozarić Z (2013) Histochemical distribution of digestive enzymes in the intestine of the common two-banded sea bream, Diplodus vulgaris, Geoffroy St-Hilaire 1817. Anat Histol Embryol 42:161–167. https://doi.org/10.1111/j.1439-0264.2012.01179.x
Hidalgo J, Velasco A, Sánchez AI, Amores P (1987) Light microscopic characterization of glycoconjugates in secretory cells of the carp (Cyprinus carpio) gill epithelium. Histochemistry 88:65–73. https://doi.org/10.1007/BF00490170
Kozarić Z, Kužir S, Nejedli S, Petrinec Z, Srebočan E (2004) Histochemical distribution of digestive enzymes in hake, Merluccius merluccius L. 1758. Vet Arhiv 74:299–308
Kozarić Z, Petrinec Z, Kužir S, Gjurčević E, Baždarić B (2011) Histochemical analyses of digestive enzymes in the intestine of adult large-scaled gurnard (Lepidotrigla cavillone, Lacepède, 1801). Anat Histol Embryol 40:314–320. https://doi.org/10.1111/j.1439-0264.2011.01074.x
Kužir S, Gjurčević E, Nejedli S, Baždarić B, Kozarić Z (2012) Morphological and histochemical study of intestine in wild and reared European eel (Anguilla anguilla L.). Fish Physiol Biochem 38:625–633. https://doi.org/10.1007/s10695-011-9543-7
Lauriano ER, Pergolizzi S, Capillo G, Kuciel M, Alesci A, Faggio C (2016) Immunohistochemical characterization of Toll-like receptor 2 in gut epithelial cells and macrophages of goldfish Carassius auratus fed with a high-cholesterol diet. Fish Shellfish Immun 59:250–255. https://doi.org/10.1016/j.fsi.2016.11.003
Leknes IL (2011) Histochemical studies on mucin-rich cells in the digestive tract of a teleost, the Buenos Aires tetra (Hyphessobrycon anisitsi). Acta Histochem 113:353–357. https://doi.org/10.1016/j.acthis.2010.01.010
Li WP, Jin WR (2006) Measurement of peroxidase activity in single neutrophils by combining catalyzed-enzyme reaction and epi-fluorescence microscopy. Talanta 70:251–256. https://doi.org/10.1016/j.talanta.2006.02.033
Løkka G, Austbø L, Falk K, Bjerkås I, Koppang EO (2013) Intestinal morphology of the wild Atlantic salmon (Salmo salar). J Morphol 274:859–876. https://doi.org/10.1002/jmor.20142
Loretz CA (1995) Electrophysiology of ion transport in teleost intestinal cells. Fish Physiol 14:25–56. https://doi.org/10.1016/S1546-5098(08)60241-1
Moretti DB, Nordi WM, Cruz TM, Cyrino JE, Machado-Neto R (2014) Histochemical distribution of intestinal enzymes of juvenile pacu (Piaractus mesopotamicus) fed lyophilized bovine colostrum. Fish Physiol Biochem 40:1487–1493. https://doi.org/10.1007/s10695-014-9942-7
Ni DS, Hong XF (1963) Histological study on digestive tract of grass carp Ctenopharyngodon idellus. Acta Hydrobiol Sin 30:1–25
Nuwansi KKT, Verma AK, Prakash C, Tiwari VK, Chandrakant MH, Shete AP, Prabhath GPWA (2015) Effect of water flow rate on polyculture of koi carp (Cyprinus carpio var. koi) and goldfish (Carassius auratus) with water spinach (Ipomoea aquatica) in recirculating aquaponic system. Aquac Int 24:1–9. https://doi.org/10.1007/s10499-015-9932-5
Pearse AGE (1972) Histochemistry. Theoretical and applied, vol 2, 3rd edn. Churchill Livingstone, Edinburgh, pp 1128–1170
Petrinec Z, Nejedli S, Kužir S, Opačak A (2005) Mucosubstances of the digestive tract mucosa in northern pike (Esox lucius L.) and European catfish (Silurus glanis L.). Vet Arhiv 75:317–327
Quan MX, Luo Y, Peng QQ, Peng F, Li E, Zhang SZ (2015) Histochemical localization of five kinds of enzymes in the digestive mucosa of megalobrama amblycephala. Chin J Histochem Cytochem 24:573–577. https://doi.org/10.16705/j.cnki.1004-1850.2015.06.013
Song H, Zhu HN, Guo H, Zhou NZ, Chen H, Zhang SZ (2012) Histochemical localization of six types of enzymes in the digestive tract of Elaphe taeniura. Chin J Histochem Cytochem 21:457–461. https://doi.org/10.3870/zgzzhx2012.05.007
Tengjaroenkul B, Smith BJ, Caceci T, Smith SA (2000) Distribution of intestinal enzyme activities along the intestinal tract of cultured Nile tilapia, Oreochromis niloticus L. Aquaculture 182:317–327. https://doi.org/10.1016/S0044-8486(99)00270-7
Vieira-Lopes DA, Pinheiro NL, Sales A, Ventura A, Araújo FG, Gomes ID, Nascimento AA (2013) Immunohistochemical study of the digestive tract of Oligosarcus hepsetus. World J Gastroenterol 19:1919–1929. https://doi.org/10.3748/wjg.v19.i12.1919
Wang AL, Liu JH, Wang WN (2005) Comparative studies on the total pigment and pigment components of color carp. Acta Hydrobiol Sin 29:694–698. https://doi.org/10.3321/j.issn:1000-3207.2005.06.016
Wang CH, Li SF, Liu ZG, Xiang SP, Wang J, Pang ZY, Duan JP (2006) Developmental quantitative genetic analysis of body weight and morphological traits in red common carp, Cyprinus carpio L. Aquaculture 251:219–230. https://doi.org/10.1016/j.aquaculture.2005.06.025
Williams JM, Duckworth CA, Vowell K, Burkitt MD, Pritchard DM (2016) Intestinal preparation techniques for histological analysis in the mouse. Curr Protoc Mouse Biol 6:148–168. https://doi.org/10.1002/cpmo.2
Funding
This study was partially supported by the Scientific Program of Tianjin City (18JCTPJC64700), Innovation Team of Tianjin Fisheries Research System (ITTFRS2017009), and Central Public-interest Scientific Institution Basal Research Fund for Chinese Academy of Tropical Agricultural Sciences (Nos. 1630052016011 and 1630052016021).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, J., Wang, Y., Lv, A. et al. Histochemical distribution of four types of enzymes and mucous cells in the intestine of koi carp (Cyprinus carpio var. koi). Fish Physiol Biochem 45, 1367–1376 (2019). https://doi.org/10.1007/s10695-019-00673-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-019-00673-y