Abstract
Understanding the relation between genetic variation and fitness remains a key question in evolutionary biology. Although heterozygosity has been reported to correlate with many fitness-related traits, the strength of the heterozygosity–fitness correlations (HFCs) is usually weak and it is still difficult to assess the generality of these associations in natural populations. It has been suggested that HFCs may become meaningful only under particular environmental conditions. Moreover, existing evidence suggests that HFCs may also differ between sexes. The aim of this study was to investigate correlations between heterozygosity in neutral markers (microsatellites) and fitness-related traits in a natural population of blue tits (Cyanistes caeruleus). Additionally, we tested whether sex and environmental conditions may influence the magnitude and direction of HFCs. We found a positive relationship between heterozygosity and body mass of 14 days post-hatching nestlings, but only among females. Our results suggest that the correlation between heterozygosity and nestling body mass observed among female offspring could be attributed to within-brood effects. We failed to find any evidence that environmental conditions as simulated by brood size manipulation affect HFCs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Associations between individual genetic diversity and fitness-related traits are commonly known as heterozygosity–fitness correlations (HFCs) and have been intensively studied in the last decades using neutral markers (reviewed in, e.g. Hansson and Westerberg 2002; Chapman et al. 2009). Two hypotheses have been put forward to explain the expected HFCs. First, the general effect hypothesis holds that inbreeding effects cause associations between neutral markers and genome-wide heterozygosity resulting in HFCs (Hansson and Westerberg 2002). Second, the local effect hypothesis suggests that linkage disequilibrium between neutral markers and selected genes leads to the observed correlations (David 1998; Hansson and Westerberg 2002).
There is currently an extensive empirical evidence for a relationship between individual heterozygosity and fitness in both plants and animals (e.g. Coltman and Slate 2003; Chapman et al. 2009; Olano-Marin et al. 2011a, b). However, a recent meta-analysis of heterozygosity–fitness correlations has noted that although HFCs are widespread, they tend to be weak and unstable (Chapman et al. 2009). The inconsistency between studies may be partially explained by the fact that HFCs could become meaningful only under particular environmental conditions; (Chapman et al. 2009) these may be revealed in competitive environments and appear non-significant under favorable conditions. Indeed, environmental factors such as thermal stress, limited food availability, parasite presence and harsh weather conditions have been found to strengthen the association between heterozygosity and fitness with more pronounced correlations arising under stressful environmental conditions (Lesbarreres et al. 2005; Da Silva et al. 2006; Marr et al. 2006; Fox and Reed 2011; Voegeli et al. 2012; Forcada and Hoffman 2014; Ferrer et al. 2016). However, this avenue of research has received limited attention to date and has rarely been tested in experimental studies. Interestingly, differences in the strength and direction of HFCs may also differ between sexes and these differences may result from sex-specific genetic structure or different sensitivity of males and females to environmental limitations (Rossiter et al. 2001; Foerster et al. 2003). Sex-specific effects of inbreeding have indeed been found in a variety of species (Saccheri et al. 2005; Charpentier et al. 2006; Reid et al. 2007; Rioux-Paquette et al. 2011). In most cases, the negative effects of inbreeding (or the positive effects of outbreeding) have been found more often among females than males. The differences in the effects of heterozygosity or inbreeding between males and females may be explained by sex-specific gene expression (Yun and Agrawal 2014), maternal investments (Charpentier et al. 2006), food acquisition (Charpentier et al. 2006), growth strategies (Coulson et al. 1998), sexual selection pressure and life history (Ebel and Phillips 2016).
Here, we tested the effect of heterozygosity on measures of offspring quality in the blue tit (Cyanistes caeruleus) nestlings. Because functionality of the markers used to measure heterozygosity in HFCs studies can have important implications for formulating predictions and interpreting results we used only neutral microsatellite markers (sensu Olano-Marin et al. 2011a, b). Neutral microsatellite markers sensu (Olano-Marin et al. 2011a, b) are expected to reflect general phenomena affecting the whole genome (like inbreeding or outbreeding) (Olano-Marin et al. 2011a, b).
We measured body mass, tarsus length and cell-mediated immune response in the nestlings and correlated these traits with the level of individual heterozygosity. Cell-mediated immunity appears to provide an adequate framework to study our question because it has frequently been shown to be a heritable and condition-dependent trait (Saino et al. 1997; Cichoń et al. 2006; Drobniak et al. 2010) and to predict survival in nestling blue tits (Cichoń and Dubiec 2005) and longevity in zebra finches (Birkhead et al. 1999). In order to test, whether environmental conditions may influence the magnitude and direction of HFCs, we manipulated environmental conditions during nestling growth by altering brood sizes. Such a manipulation appears to be an effective way of increasing within-brood competition, as negative effects of brood size enlargement on various nestling characteristics has been repeatedly reported in many species including blue tit (e.g. Cichoń and Dubiec 2005; Voegeli et al. 2012). We expected the HFCs to be particularly expressed among offspring from experimentally enlarged broods. Based on previous studies (e.g. Olano-Marin et al. 2011a, b), we also predicted the HFCs to be particularly expressed among females compared to males. In statistical sense we expected a significant interaction between experimental treatment/sex and individual level of heterozygosity.
Materials and methods
Study species and field techniques
The study was carried out in the spring 2011 in a blue tit population breeding in nest boxes on the Swedish island, Gotland [57°03′N, 18°17′E; see Pärt and Gustafsson (1989) for more detailed description of the study area]. From the end of April, we regularly inspected nest boxes to determine the laying date, clutch size and hatching success. On the second day post-hatching all nestlings were individually marked by clipping their nails, blood-sampled and weighed with an electronic balance (to the nearest 0.1 g). Nestlings were weighed again on the day 11, 12 and 14 after hatching. Tarsus length was measured 14 days post-hatching with an electronic caliper to the nearest 0.1 mm. Adults were caught inside nest boxes or by mist nets while feeding 14-day-old nestlings. Adult sex was determined by the presence of a brood patch (only females are incubating in this species). Blood samples (ca. 20 μl) were collected from all nestlings and adults and stored in 96% ethanol for further genetic analyses.
The data upon which this study is based have been obtained following the Swedish guidelines for work on natural populations and under permit from the Swedish Ringing Centre.
Brood size manipulation experiment and cross-fostering procedure
To create standard differences in rearing conditions, we manipulated brood size. We selected pairs of broods with the same hatching date and similar brood size (±1 chick). One randomly selected brood in each pair was enlarged (experimental nest) by adding three nestlings from a donor nest. These extra nestlings from a donor nest served only to increase within-nest competition and were not used in the analyses. The brood enlargement causes ca. 30% increase in brood size. In addition to brood size manipulation, half of randomly chosen nestlings were cross-fostered between the control and the enlarged nest within each pair (only original nestlings, excluding donor ones). Thus, nestlings originating from one family were raised under different environmental conditions. Brood size manipulation and cross-fostering were performed on the second day after hatching.
Cell-mediated immune response
On the day 11 post-hatch we measured cell-mediated immune response induced by non-pathogenic antigen, phytohaemagglutinin (PHA). PHA is a plant-derived lectin that has a strong mitogenic effect on T lymphocytes, serving as a model non-pathogenic antigen (Goto et al. 1978). In our experiment 0.2 mg of PHA (Sigma Aldrich, Poznań, Poland) suspended in 0.04 mL of saline was injected to the right wing web. The thickness of the wing web was measured in triplicate at two time points: immediately prior to and 24 h after the injection using pressure-sensitive spessimeter (Mitutoyo, Tokyo, Japan). The level of immune response was expressed as the intensity of swelling and was calculated as the difference between wing-web thickness prior to and after the injection.
Genetic and paternity analyses
DNA was extracted from blood samples with Chelex according to a standard protocol. Nestling sex was determined using P2 and P8 primers (Griffiths et al. 1998). Fifteen autosomal microsatellite markers were amplified (Ase18, Cdi31, Mcyµ4, Pca3, Pca4, Pca7, Pca9, PK12, Pma303, PmaC25, PmaGAn27, PmaGAn40, PmaTGAn45, Pocc1, Pocc6) by polymerase chain reaction using Qiagen multiplex PCR kit (Qiagen AG, Hombrechtikon, Switzerland) as described in the study of Olano-Marin et al (2010). These markers were classified as presumably neutral sensu Olano-Marin et al. (2011a, b). We used Fstat (version 2.9.3; Goudet 1995) to calculate deviation from Hardy–Weinberg equilibrium and linkage disequilibrium, using genetic data from adults only to avoid bias due to family structures. After Bonferroni correction, none of the loci deviated from Hardy–Weinberg equilibrium, and there was no evidence for linkage disequilibrium between any pair of microsatellite markers used. To avoid bias due to family structures, we based calculations of allele frequency on the adult data set only. Microsatellite loci were also used for paternity analyses. Nestlings were considered as extra-pair offspring (EPO) if their genotype mismatched their putative father’s at two or more loci. In total we genotyped 67 females, 66 males and 313 nestlings from 33 nests with both parents known across a panel of 15 microsatellite loci. We used Cervus 3.0 (Kalinowski et al. 2007) for parentage assignment. The analysis revealed moderate levels of extra-pair paternity. We found 12 broods (36.36%) with mixed paternity and 21 broods with no extra-pair young. 38 out of the 313 genotyped offspring (12.14%) were not sired by their social father.
Heterozygosity
Homozygosity by loci (HL) was used as an estimate of individual genetic diversity. HL takes into account the allelic variability of each locus and thus improves heterozygosity estimate by weighing the contribution of each locus to the homozygosity index, giving more weight to more informative loci (Aparicio et al. 2006). HL values range from 0 (all loci heterozygous) to 1 (complete homozygosity). We computed a heterozygosity parameter as 1-HL. HL was calculated using Rhh, an extension package for R (Alho et al. 2010). We tested for identity disequilibrium with two approaches in R (R Development Core Team 2013) using the function in the ‘inbreedR’ package (Stoffel et al. 2016). Following Balloux et al. (2004) we calculated the heterozygosity–heterozygosity correlation (HHC). All the markers were randomly divided into two subsets of fifteen loci that were separately used to calculate multi-locus heterozygosity and then the correlation coefficient between the heterozygosity of the two subsets was calculated. If neutral markers such as microsatellites carry information about genome-wide levels of heterozygosity, then comparing two random subsets of such markers should yield a positive, significant correlation (Balloux et al. 2004; Alho et al. 2010). We ran 1000 randomizations of the markers for heterozygosity by loci (HL) estimate of heterozygosity and estimated the average HHC coefficient (r) and the 95% confidence intervals (Alho et al. 2010). To detect identity disequilibrium (correlations in heterozygosity among loci) due to variance in inbreeding, we also calculated the parameter, g2, because this gives a more powerful statistic than HHC (Szulkin et al. 2010). Then, we assessed the expected power of our microsatellite markers set to detect general-effect HFCs using formulae from Miller et al. (2014; eqn 5).
Statistics
We used generalized linear mixed models (GLMMs) in R (R Development Core Team 2013) with the add-on R package lme4 (Bates et al. 2012). The association between parental heterozygosity and offspring heterozygosity was assessed using another linear mixed-effect model with genetic father and genetic mother heterozygosity as explanatory variables. To account for the dependences among siblings, we included nest identity as a random factor. Nestling response to PHA, tarsus length, and body mass were modeled using linear mixed-effect models with restricted maximum-likelihood estimation (REML). Explanatory variables in the starting models were heterozygosity levels of nestlings (HLnestlings), nestling sex, and brood size manipulation. We included heterozygosity levels of foster fathers (HLfostermale) and foster mothers (HLfosterfemale) and original brood size as covariates. When modeling the response to PHA, we additionally included nestling body mass on the day 12 after hatching. To account for the common environment dependences among siblings, we included nest of origin as a random factor. Moreover, nest of rearing was also included as a random factor to account for the cross-fostering procedure; hence, we controlled for the fact that some chicks remained in their parental nests but some were raised in foster nests. All interactions were tested but discarded if non-significant to increase the power of the test, which is particularly important given the relatively small sample size. In the final analysis we excluded extra-pair nestlings, thus we used a full-sibling approach, i.e. we controlled the level of inbreeding. Sample sizes differ among the analyses as some measurements were not always available for all nestlings. We used “within-group centering” (Van de Pol and Wright 2009) to separate within- from between-brood effects of nestling heterozygosity. The within-brood effect was assessed by subtracting the brood mean heterozygosity level from each individual nestling heterozygosity estimator. The between-brood effect was simply assessed by the mean values for each brood. To this end, we fitted (1) a model with body mass on the day 14 post-hatching of female nestlings as response variable (the significant effects of HL on body mass were detected in the starting models only among females), introducing HLnestlings, experimental treatment, original brood size, HLfosterfemale and HLfostermale as predictors, and nest of origin and nest of rearing as random effects (model 1), and (2) a model with the individually centered values of HLnestlings within a nest (for within-nest effects) and the mean HLnestlings of each nest (for between-nest effects) as predictors, and nest of origin and nest of rearing as random effects (model 2, van de Pol and Wright 2009). A stronger statistical support for (e.g. lower AIC value) would indicate whether effects of HLnestlings are differentially driven by within- and between nest effects. We used the ‘MuMIn’ package in R to calculate effect sizes (R 2) from mixed models (Bartoń 2009).
Single-locus effects
To test for the possibility that local, rather than general, effects were driving the observed HFCs in case of the body mass on the day 14 post-hatching among female nestlings, we ran a multiple regression following Szulkin et al. (2010) where each locus (n = 15) was included as an individual predictor and coded as 0 or 1 for homozygous or heterozygous respectively. If this model explains more variation than a basic model where multi locus heterozygosity is included as a single predictor, then this will lend support to the local effects hypothesis.
Results
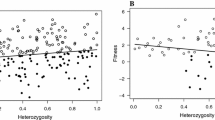
The mean HL measured with all markers ranged from to 0.50 to 1.0 (mean ± SD = 0.825 ± 0.098). Heterozygosity–heterozygosity correlations (HHC) and g2 for markers were not significantly different from zero (i.e. 95% quantiles crossed zero) [all markers: rHHC = −0.06, 95% CI = −0.068–0.196; g2 = 0.001, 95% CI = 0.002–0.006, P = 0.17]. The expected power to detect HFCs according to Miller et al. (2014) was small (r 2 = 0.06). Parental heterozygosity was not significantly associated with offspring heterozygosity (Table 1). We found a significant interaction between sex and nestlings heterozygosity in the model explaining variation in nestling body mass (Table 2). The relationship between nestling heterozygosity and body mass was significant only among females, while no significant relationship was detected among males (Table 3; Fig. 1, effect size R 2 = 0.10). There was a significant interaction between experimental treatment and sex in case of tarsus length (Table 2). The interaction between experimental treatment and heterozygosity of nestlings appeared non-significant for all measured traits (Table 2).
Nestling body mass on the day 14 post-hatching in relation to nestling heterozygosity and sex. The lines are the best-fit regressions through the data. (See “Materials and methods” section for the details on statistics)
The centered model, fitted to discriminate effects of heterozygosity within a nest (e.g. between full-siblings, sharing the same inbreeding coefficient) was better supported (lower AIC values) than the non-centered model (e.g. between individuals with different levels of inbreeding), in case of body mass among female offspring (Body mass: AIC: 480.68 vs 481.97). Nevertheless it has to be kept in mind that models with delta AIC <2 are considered as alternative (Burnham and Anderson 2002), therefore non-centered model would not be statistically incorrect in this case. The relationships between heterozygosity levels and body mass observed among females could be attributed to the effects of heterozygosity within broods, as indicated by the significant within-nest variable in the centered model (Table 4).
There was no support for a local effect of heterozygosity acting on body mass of the females (F14,101 = 1.04; P = 0.579).
Discussion
Here we found that brood size manipulation experiment did not significantly affect the correlation between heterozygosity and nestling performance in blue tits. In this study, we manipulated growth conditions of nestling blue tits and investigated the mediating effect of treatment on the occurrence of HFCs. To our knowledge the effect of brood size manipulation experiment on the occurrence of the HFCs has been experimentally investigated in only a single study so far. In accordance to our study, Voegeli et al. (2012) did not find any effect of brood size manipulation on the incidence of HFCs. This may potentially stem from the relatively small sample size used in our study and in the study of Voegeli et al. (2012), but it is possible that HFCs are sensitive to stressful environmental conditions.
We found heterozygosity to have a positive effect on body mass in female nestlings, but no effect was detected among males. Differences in the strength and direction of HFCs between sexes have been reported previously (e.g. Rossiter et al. 2001; Foerster et al. 2003; Olano-Marin et al. 2011b). It has been shown that females with higher body mass relative to their siblings enjoyed larger longevity, suggesting that prevailing in brood competition for food has long-term, besides obvious short-term, effects on viability (Mock and Parker 1997). In blue tit nestlings, females are smaller than males, and this sexual size dimorphism results in asymmetric sibling competition, and nestlings of the smaller sex are expected to prioritize the development of those morphological characters that maximize effective sibling competition (Mainwaring et al. 2012). Thus, our results suggest that more heterozygous female nestlings possibly may benefit from their larger genetic diversity and are therefore able to invest more into development and growth. In our study we did not observe any relation between heterozygosity and immune response. Similarly, Voegeli et al. (2013) manipulated the harshness of the rearing conditions of great tit nestlings by experimentally manipulating brood size and did not find effect of brood size manipulation on the relationship between heterozygosity and immune responsiveness. However, they found that heterozygosity was positively related to the immune response to a novel antigen among nests experiencing experimental parasite removal. This study suggests that heterozygosity–fitness correlations may become meaningful only under particular environmental conditions, not necessary related to direct resource limitation.
According to our results the correlation between heterozygosity and nestling body mass observed among female offspring could be attributed to within-brood effects. As full-siblings share their ancestry and inbreeding history, HFCs within full-sibling are commonly interpreted as evidence for local effects (Hansson et al. 2001; Da Silva et al. 2006; Fossøy et al. 2009). However, it has recently been put forward that the existence of HFCs among full-siblings should be interpreted with care, as full-siblings will vary in the proportion of the genome, which is identical by descent due to chance events during Mendelian segregation (Forstmeier et al. 2012; but see Hansson and Westerberg 2008). This variation may already be sufficient to cause HFCs even among full-siblings. If local or direct effects underlie the detected HFCs, we would expect to detect single-locus heterozygosity (SLH) effects. However, there was no evidence for SLH among our microsatellite markers set, but it must be kept in mind that the local effects are extremely difficult to detect with this methodology if the multi-locus heterozygosity effects are already weak (Szulkin et al. 2010). In fact, Szulkin et al. (2010) were not aware of any HFCs data that passed this rigorous test and detected significant local effects (but see e.g. García-Navas et al. 2014; Minias et al. 2015). Alternatively, the detected HFCs may arise due to genome-wide effects of heterozygosity and hence indicate inbreeding depression. We did not detect positive heterozygosity–heterozygosity correlations (HHCs; Balloux et al. 2004) for our microsatellite markers set, also the parameter g2 gave no indication for inbreeding in our population, giving no support to the general effect hypothesis. However, according to formulae from Miller et al. (2014) the power of our markers to estimate inbreeding in our study system was rather small. It must be kept in mind that the nonsignificant values should not be misinterpreted as disproving general effects, as the effects of weak inbreeding are more readily detected on the phenotypic level than on the level of a limited number of markers (Szulkin et al. 2010).
In conclusion, our results provide empirical evidence for heterozygosity effects on fitness-related trait in the blue tit nestlings. The fact that we did not find any evidence for the local effects and that the association between fitness-related traits and individual genetic diversity suggests that the observed effects may be mediated by inbreeding and/or by genome-wide genetic diversity. In our study, the strength and the shape of the relationship between fitness-related traits and heterozygosity differ between sexes and measured traits suggests that overall effect of heterozygosity are context-dependent. On the other hand, our study do not support the idea that HFCs are more pronounced under elevated environmental stress levels than under optimal conditions, suggesting that the environmental stress not necessary enhances HFCs.
References
Alho JS, Valimaki K, Merila J (2010) Rhh: an R extension for estimating multilocus heterozygosity and heterozygosity–heterozygosity correlation. Mol Ecol Res 10:720–722
Aparicio JM, Ortego J, Cordero PJ (2006) What should we weigh to estimate heterozygosity, alleles or loci? Mol Ecol 15:4659–4665
Balloux F, Amos W, Coulson T (2004) Does heterozygosity estimate inbreeding in real populations? Mol Ecol 13:3021–3031
Bartoń K (2009) MuMIn: multi-model inference. R package version 0.12.2/r18. http://R-Forge.R-project.org/projects/mumin/
Bates D, Maechler M, Bolker B (2012) lme4: linear mixed-effects models using S4 classes. R package version 0.999999-0. http://CRAN.R-project.org/package=lme4
Birkhead TR, Fletcher F, Pellatt EJ (1999) Nestling diet, secondary sexual traits and fitness in the zebra finch. Proc R Soc Lond B 266:385–390
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Chapman JR, Nakagawa S, Coltman DW, Slate J, Sheldon BC (2009) A quantitative review of heterozygosity–fitness correlations in animal populations. Mol Ecol 18:2746–2765
Charpentier M, Setchell JM, Prugnolle F et al (2006) Life history correlates of inbreeding depression in mandrills (Mandrillus sphinx). Mol Ecol 15:21–28
Cichoń M, Dubiec A (2005) Cell-mediated immunity predicts the probability of local recruitment in nestling blue tits. J Evol Biol 18:962–966
Cichoń M, Sendecka J, Gustafsson L (2006) Genetic and environmental variation in immune response of collared flycatcher nestlings. J Evol Biol 19:1701–1706
Coltman DW, Slate J (2003) Microsatellite measures of inbreeding: a meta-analysis. Evolution 57:971–983
Coulson TN, Pemberton JM, Albon SD et al (1998) Microsatellites reveal heterosis in red deer. Proc R Soc Lond B 265:489–495
Da Silva A, Luikart G, Yoccoz NG, Cohas A, Allaine D (2006) Genetic diversity–fitness correlation revealed by microsatellite analyses in European alpine marmots (Marmota marmota). Conserv Genet 7:371–382
David P (1998) Heterozygosity–fitness correlations: new perspectives on old problems. Heredity 80:531–537
Drobniak SM, Wiejaczka D, Arct A, Dubiec A, Gustafsson L, Cichoń M (2010) Sex-specific heritability of cell-mediated immune response in the blue tit nestlings (Cyanistes caeruleus). J Evol Biol 23:1286–1292
Ebel ER, Phillips PC (2016) Intrinsic differences between males and females determine sex-specific consequences of inbreeding. BMC Evol Biol 16(1):1
Ferrer ES, García-Navas V, Sanz JJ, Ortego J (2016) The strength of the association between heterozygosity and probability of inter-annual local recruitment increases with environmental harshness in blue tits. Ecol Evol 6:8857–8869
Foerster K, Delhey K, Johnsen A, Lifjeld JT, Kempenaers B (2003) Females increase offspring heterozygosity and fitness through extra-pair matings. Nature 425:714–717
Forcada J, Hoffman JI (2014) Climate change selects for heterozygosity in a declining fur seal population. Nature 511:462–465
Forstmeier W, Schielzeth H, Mueller JC, Ellegren H, Kempenaers B (2012) Heterozygosity–fitness correlations in zebra finches: microsatellite markers can be better than their reputation. Mol Ecol 21:3237–3249
Fossøy F, Johnsen A, Lifjeld JT (2009) Cell-mediated immunity and multi-locus heterozygosity in bluethroat nestlings. J Evol Biol 22:1954–1960
Fox CW, Reed DH (2011) Inbreeding depression increases with environmental stress: an experimental study and meta-analysis. Evolution 65:246–258
García-Navas V, Cáliz-Campal C, Ferrer ES, Sanz JJ, Ortego J (2014) Heterozygosity at a single locus explains a large proportion of variation in two fitness-related traits in great tits: a general or a local effect? J Evol Biol 27(12):2807–2819
Goto N, Kodama H, Okada K, Fujimoto Y (1978) Suppression of phytohemagglutinin skin response in thymectomized chickens. Poult Sci 57:246–250
Goudet J (1995) FSTAT (Version 1.2): a computer program to calculate F-statistics. J Hered 86:485–486
Griffiths R, Double M, Orr K, Dawson R (1998) A DNA test to sex most birds. Mol Ecol 7:1071–1075
Hansson B, Westerberg L (2002) On the correlation between heterozygosity and fitness in natural populations. Mol Ecol 11:2467–2474
Hansson B, Westerberg L (2008) Heterozygosity–fitness correlations within inbreeding classes: local or genome-wide effects? Conserv Genet 9:73–78
Hansson B, Bensch S, Hasselquist D, Akesson M (2001) Microsatellite diversity predicts recruitment of sibling great reed warblers. Proc R Soc Lond B 268:1287–1291
Kalinowski ST, Taper ML, Marshall TC (2007) Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol Ecol 16:1099–1106
Lesbarreres D, Primmer SR, Laurila A, Merila J (2005) Environmental and population dependency of genetic variability-fitness correlations in Rana temporaria. Mol Ecol 14:311–323
Mainwaring MC, Dickens M, Hartley IR (2012) Sexual dimorphism and offspring growth: smaller female Blue Tit nestlings develop relatively larger gapes. J Ornithol 153(4):1011–1016
Marr AB, Arcese P, Hochachka WM, Reid JM, Keller LF (2006) Interactive effects of environmental stress and inbreeding on reproductive traits in a wild bird population. J Anim Ecol 75:1406–1415
Miller JM, Malenfant RM, David P, Davis CS, Poissant J, Hogg JT, Festa-Bianchet M, Coltman DW (2014) Estimating genome-wide heterozygosity: effects of demographic history and marker type. Heredity 112(3):240–247
Minias P, Wojczulanis-Jakubas K, Rutkowski R, Kaczmarek K (2015) Local heterozygosity effects on nestling growth and condition in the great cormorant. Evol Biol 42(4):452–460
Mock DW, Parker GA (1997) The evolution of sibling rivalry. Oxford University Press, Oxford
Olano-Marin J, Dawson DA, Girg A, Hansson B, Ljungqvist M, Kempenaers B et al (2010) A genome-wide set of 106 microsatellite markers for the blue tit (Cyanistes caeruleus). Mol Ecol Resour 10:516–532
Olano-Marin J, Mueller JC, Kempenaers B (2011a) Correlations between heterozygosity and reproductive success in the blue tit (Cyanistes caeruleus): an analysis of inbreeding and single locus effects. Evolution 65:3175–3194
Olano-Marin J, Mueller JC, Kempenaers B (2011b) Heterozygosity and survival in blue tits (Cyanistes caeruleus): contrasting effects of presumably functional and neutral loci. Mol Ecol 20:4028–4041
Pärt Y, Gustafsson L (1989) Breeding dispersal in the collared flycatcher (Ficedula albicollis): possible causes and reproductive consequences. J Anim Ecol 58:305–320
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reid JM, Arcese P, Keller LF, Elliott KH, Sampson L, Hasselquist D (2007) Inbreeding effects on immune response in free-living song sparrows (Melospiza melodia). Proc R Soc Lond B 274:697–706
Rioux-Paquette E, Festa-Bianchet M, Coltman DW (2011) Sex differential effects of inbreeding on overwinter survival, birth date and mass of bighorn lambs. J Evol Biol 24:121–131
Rossiter SJ, Jones G, Ransome RD, Barratt EM (2001) Outbreeding increases offspring survival in wild greater horseshoe bats (Rhinolophus ferrumequinum). Proc R Soc Lond B 268:1055–1061
Saccheri IJ, Lloyd HD, Helyar SJ, Brakefield PM (2005) Inbreeding uncovers fundamental differences in the genetic load affecting male and female fertility in a butterfly. Proc R Soc Lond B 272:39–46
Saino N, Calza S, Möller AP (1997) Immunocompetence of nestling barn swallows in relation to brood size and parental effort. J Anim Ecol 66:827–836
Stoffel MA, Esser M, Kardos M, Humble E, Nichols H, David P, Hoffman JI (2016) inbreedR: an R package for the analysis of inbreeding based on genetic markers. Methods Ecol Evol 7(11):1331–1339
Szulkin M, Bierne N, David P (2010) Heterozygosity–fitness correlations: a time for reappraisal. Evolution 64:1202–1217
Van de Pol M, Wright J (2009) A simple method for distinguishing within- versus between-subject effects using mixed models. Anim Behav 77:753–758
Voegeli B, Saladin V, Wegmann M, Richner H (2012) Parasites as mediators of heterozygosity–fitness correlations in the Great Tit (Parus major). J Evol Biol 25:584–590
Voegeli B, Saladin V, Wegmann M, Richner H (2013) Heterozygosity is linked to the costs of immunity in nestling great tits (Parus major). Ecol Evol 3(14):4815–4827
Yun L, Agrawal AF (2014) Variation in the strength of inbreeding depression across environments: effects of stress and density dependence. Evolution 68:3599–3606
Acknowledgements
We thank Ewa Śliwińska for the preliminary genetic analyses. This research was funded by the National Science Centre (Poland) allocated on the basis of the decision number DEC-2013/09/B/NZ8/03322 (OPUS 9).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Arct, A., Sudyka, J., Podmokła, E. et al. Heterozygosity–fitness correlations in blue tit nestlings (Cyanistis caeruleus) under contrasting rearing conditions. Evol Ecol 31, 803–814 (2017). https://doi.org/10.1007/s10682-017-9911-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-017-9911-6