Abstract
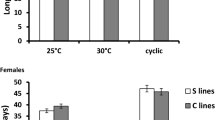
Despite obvious benefits, reproduction also imposes severe costs on females. Such costs and benefits are highly sensitive to environmental factors. Rapidly changing conditions may thus disturb a finely poised balance between the two and pose a challenge to natural populations. A more complete understanding of reproduction and population fitness across different environments is, hence, crucial. In particular, sexual selection could either be beneficial or detrimental when conditions change abruptly. Here Tribolium castaneum females were subjected to mating treatments with or without sexual selection (virginity, monogamy, polyandry) replicated at standard versus elevated temperatures. We found a substantial survival cost of reproduction at the standard, but not at the elevated temperature. Reproductive success was similar across mating treatments at the standard temperature, but at elevated temperature we detected a significant benefit of polyandry compared to monogamy. These findings indicate that environmental heterogeneity can strongly influence the balance between costs and benefits when sexual selection is allowed to act. Furthermore, reproduction may be critically affected by changes in temperature with potentially profound consequences for population fitness.



Similar content being viewed by others
References
Angilletta MJ (2009) Thermal adaptation: a theoretical and empirical synthesis. Oxford University Press, Oxford
Arnqvist G, Rowe L (2005) Sexual conflict. Princeton University Press, Princeton
Bale JS, Masters GJ, Hodkinson ID et al (2002) Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Glob Change Biol 8:1–16
Blanckenhorn WU, Hosken DJ, Martin OY et al (2002) The costs of copulating in the dung fly Sepsis cynipsea. Behav Ecol 13:353–358
Candolin U, Heuschele J (2008) Is sexual selection beneficial during adaptation to environmental change? TREE 23:446–452
Chevin LM, Lande R, Mace GM (2010) Adaptation, plasticity, and extinction in a changing environment: towards a predictive theory. PLoS Biol 8:1–8
Chihrane J, Lauge G (1994) Effects of high-temperature shocks on male germinal cells of Trichogramma brassicae (Hymenoptera, Trichogrammatidae). Entomophaga 39:11–20
Clutton-Brock TH (1984) Reproductive effort and terminal investment in iteroparous animals. Am Nat 123:212–229
Cohet YA, David JR (1976) Deleterious effects of copulation in Drosophila females as a function of growth temperature of both sexes. Experientia 32:696–697
Crudgington HS, Siva-Jothy MT (2000) Genital damage, kicking and early death—the battle of the sexes takes a sinister turn in the bean weevil. Nature 407:855–856
Edvardsson M (2007) Female Callosobruchus maculatus mate when they are thirsty: resource-rich ejaculates as mating effort in a beetle. Anim Behav 74:183–188
Fedina TY, Lewis SM (2004) Female influence over offspring paternity in the red flour beetle Tribolium castaneum. Proc Roy Soc Lond B 271:1393–1399
Fedina TY, Lewis SM (2007) Female mate choice across mating stages and between sequential mates in flour beetles. J Evol Biol 20:2138–2143
Fedina TY, Lewis SM (2008) An integrative view of sexual selection in Tribolium flour beetles. Biol Rev 83:151–171
Fox CW (1993) Multiple mating, lifetime fecundity and female mortality of the bruchid beetle, Callosobruchus maculatus (Coleoptera, Bruchidae). Funct Ecol 7:203–208
Fox CW, Rauter CM (2003) Bet-hedging and the evolution of multiple mating. Evol Ecol Res 5:273–286
Fricke C, Perry J, Chapman T et al (2009) The conditional economics of sexual conflict. Biol Lett 5:671–674
Hadley NF (1994) Water relations of terrestrial arthropods. Academic Press, San Diego
Holland B, Rice WR (1999) Experimental removal of sexual selection reverses intersexual antagonistic coevolution and removes a reproductive load. PNAS 96:5083–5088
Hosken DJ, Stockley P (2003) Benefits of polyandry: a life history perspective. Evol Biol 33:173–194
Hosken DJ, Garner TWJ, Ward PI (2001) Sexual conflict selects for male and female reproductive characters. Curr Biol 11:489–493
Hughes L (2000) Biological consequences of global warming: is the signal already apparent? TREE 15:56–61
Hunt J, Brooks R, Jennions MD et al (2004) High-quality male field crickets invest heavily in sexual display but die young. Nature 432:1024–1027
Hurst GDD, Sharpe RG, Broomfield AH et al (1995) Sexually-transmitted disease in a promiscuous insect, Adalia bipunctata. Ecol Entomol 20:230–236
Ingleby FC, Hunt J, Hosken DJ (2010) The role of genotype-by-environment interactions in sexual selection. J Evol Biol 23:2031–2045
Ivy TM, Johnson JC, Sakaluk SK (1999) Hydration benefits to courtship feeding in crickets. Proc Roy Soc Lond B 266:1523–1527
Jennions MD, Petrie M (2000) Why do females mate multiply? A review of the genetic benefits. Biol Rev 75:21–64
Johnstone RA, Keller L (2000) How males can gain by harming their mates: sexual conflict, seminal toxins, and the cost of mating. Am Nat 156:368–377
Lewis SM, Jutkiewicz E (1998) Sperm precedence and sperm storage in multiply mated red flour beetles. Behav Ecol Sociobiol 43:365–369
Lewis SM, Kobel A, Fedina T et al (2005) Sperm stratification and paternity success in red flour beetles. Physiol Entomol 30:303–307
Maes D, Titeux N, Hortal J et al (2010) Predicted insect diversity declines under climate change in an already impoverished region. J Ins Conserv 14:485–498
Mahroof R, Zhu KY, Neven L et al (2005) Expression patterns of three heat shock protein 70 genes among developmental stages of the red flour beetle, Tribolium castaneum (Coleoptera: Tenebrionidae). Comp Biochem Physiol A 141:247–256
Martin OY, Hosken DJ (2003a) Costs and benefits of evolving under experimentally enforced polyandry or monogamy. Evolution 57:2765–2772
Martin OY, Hosken DJ (2003b) The evolution of reproductive isolation through sexual conflict. Nature 423:979–982
Martin OY, Hosken DJ (2004) Copulation reduces male but not female longevity in Saltella sphondylli (Diptera: Sepsidae). J Evol Biol 17:357–362
Meehl GA, Stocker TF, Collins WD et al (2007) Global climate projections. In: Solomon S, Qin D, Manning M et al (eds) Climate change 2007: the physical science basis. Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, pp 747–845
Menendez R (2007) How are insects responding to global warming? Tijdschrift voor Entomol 150:355–365
Michalczyk L, Martin OY, Millard AL et al (2010) Inbreeding depresses sperm competitiveness, but not fertilization or mating success in male Tribolium castaneum. Proc Roy Soc Lond B 277:3483–3491
Michalczyk L, Millard AL, Martin OY et al (2011) Experimental evolution exposes female and male responses to sexual selection and conflict in Tribolium castaneum. Evolution 65:713–724
Morrow EH, Arnqvist G, Pitnick S (2003) Adaptation versus pleiotropy: why do males harm their mates? Behav Ecol 14:802–806
Narraway C, Hunt J, Wedell N et al (2010) Genotype-by-environment interactions for female preferences. J Evol Biol 23:2550–2557
Nilsson T, Fricke C, Arnqvist G (2003) The effects of male and female genotype on variance in male fertilization success in the red flour beetle (Tribolium castaneum). Behav Ecol Sociobiol 53:227–233
Pai A, Bernasconi G (2008) Polyandry and female control: the red flour beetle Tribolium castaneum as a case study. J Exp Zool (Mol Dev Evol) 310B:148–159
Pai A, Yan GY (2003) Rapid female multiple mating in red flour beetles (Tribolium castaneum). Can J Zool 81:888–896
Partridge L, Farquhar M (1981) Sexual activity reduces lifespan of male fruitflies. Nature 294:580–582
Pizzari T, Parker GA (2009) Sperm competition and sperm phenotype. In: Birkhead TR, Hosken DJ, Pitnick S (eds) Sperm biology: an evolutionary perspective. Elsevier Academic Press, Burlington, pp 207–245
Poiani A (2006) Complexity of seminal fluid: a review. Behav Ecol Sociobiol 60:289–310
Qazi MCB, Aprille JR, Lewis SM (1998) Female role in sperm storage in the red flour beetle, Tribolium castaneum. Comp Biochem Physiol A 120:641–647
Reed DH (2008) Effects of population size on population viability: from mutation to environmental catastrophes. In: Carroll SP, Fox CW (eds) Conservation biology: evolution in action. Oxford University Press, Oxford, pp 16–34
Rice WR (1996) Sexually antagonistic male adaptation triggered by experimental arrest of female evolution. Nature 381:232–234
Sadd B, Holman L, Armitage H et al (2006) Modulation of sexual signalling by immune challenged male mealworm beetles (Tenebrio molitor L.): evidence for terminal investment and dishonesty. J Evol Biol 19:321–325
Sokoloff A (1972) The biology of Tribolium with special emphasis on genetic aspects. Oxford University Press, Oxford
Svard L, Mcneil JN (1994) Female benefit, male risk—polyandry in the true armyworm Pseudaletia unipuncta. Behav Ecol Sociobiol 35:319–326
Tanaka Y (1996) Sexual selection enhances population extinction in a changing environment. J Theor Biol 180:197–206
Thomas CD, Cameron A, Green RE et al (2004) Extinction risk from climate change. Nature 427:145–148
Ward PI, Vonwil J, Scholte EJ et al (2002) Field experiments on the distributions of eggs of different phosphoglucomutase (PGM) genotypes in the yellow dung fly Scathophaga stercoraria (L.). Mol Ecol 11:1781–1785
Watson PJ, Arnqvist G, Stallmann RR (1998) Sexual conflict and the energetic costs of mating and mate choice in water striders. Am Nat 151:46–58
West PM, Packer C (2002) Sexual selection, temperature, and the lion’s mane. Science 297:1339–1343
Wigby S, Chapman T (2005) Sex peptide causes mating costs in female Drosophila melanogaster. Curr Biol 15:316–321
Wiklund C, Karlsson B, Leimar O (2001) Sexual conflict and cooperation in butterfly reproduction: a comparative study of polyandry and female fitness. Proc Roy Soc Lond B 268:1661–1667
Williams SE, Shoo LP, Isaac JL et al (2008) Towards an integrated framework for assessing the vulnerability of species to climate change. PLoS Biol 6:2621–2626
Zeh JA, Zeh DW (2001) Reproductive mode and the genetic benefits of polyandry. Anim Behav 61:1051–1063
Acknowledgments
We thank the SNF for support (standard research grant 31003A_125144/1, Ambizione grant PZ00P3_121777/1 to OYM), Sonja Sbilordo for help with the experiment and Marco Demont for statistical advice. We further thank Camillo Berenos, Luc Bussière, Niels Kerstes & Sara Goodacre for helpful comments and interesting discussions and Prof. Schmid-Hempel’s group for providing a stimulating research and social environment.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grazer, V.M., Martin, O.Y. Elevated temperature changes female costs and benefits of reproduction. Evol Ecol 26, 625–637 (2012). https://doi.org/10.1007/s10682-011-9508-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-011-9508-4