Abstract
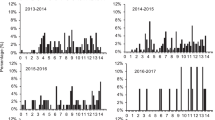
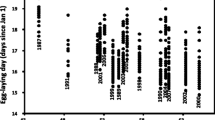
In several species of short-lived Australian agamid lizards, an individual’s sex is determined by the nest temperatures encountered during incubation. The adaptive significance of such systems remains unclear. Here, we explore the hypothesis that (1) the optimal timing of hatching differs between the sexes, and thus (2) temperature-dependent sex determination (TSD) enhances maternal and offspring fitness by generating seasonal shifts in offspring sex ratios. Our model predicts that TSD can indeed enhance maternal fitness returns in short-lived lizards if (1) male–male competition is intense, thus reducing mating success of newly-matured males (but not females), and (2) the nesting season is prolonged, such that seasonal effects become significant. Available data on the distribution of TSD in Australian agamid lizards broadly support these predictions. Because both the level of male–male competition and the length of nesting season can vary at small spatial and temporal scales, selective forces on sex-determining mechanisms also should vary. Hence, our model predicts extensive small-scale (intraspecific) variation in sex-determining systems within agamid lizards, as well as among species.


Similar content being viewed by others
References
Andrews RM (2005) Incubation temperature and sex ratio of the veiled chameleon (Chamaeleo calyptratus). J Herpetol 39:515–518
Aragón P, López P, Martín J (2006) Roles of male residence and relative size in the social behavior of Iberian rock lizards, Lacerta monticola. Behav Ecol Sociobiol 59:762–769
Baroiller JF, Chourrout D, Fostier A, Jalabert B (1995) Temperature and sex chromosomes govern sex ratios of the mouthbrooding cichlid fish Oreochromis niloticus. J Exp Zool 273:216–223
Brattstrom BH (1971) Social and thermoregulatory behaviour of the bearded dragon, Amphibolurus barbatus. Copiea 1971:484–497
Bull JJ, Vogt RC (1979) Temperature-dependent sex determination in turtles. Science 206:1186–1188
Bull JJ (1983) Evolution of sex determining mechanisms. Benjamin/Cummings, Menlo Park, California, USA
Bull JJ, Charnov EL (1989) Enigmatic reptilian sex ratios. Evolution 43:1561–1566
Carpenter CC, Badham JA, Kimble B (1970) Behavior patterns of three species of Amphibolurus (Agamidae). Copeia 1970:497–505
Charnov EL, Bull JJ (1977) When is sex environmentally determined? Nature 266:828–830
Cogger HG (1969) A study of the ecology and biology of the mallee dragon (Amphibolurus fordi) and its adaptation to survival in an arid environment. Dissertation, Macquarie University
Cogger HG (1978) Reproductive cycles, fat body cycles and socio-sexual behaviour in the mallee dragon, Amphibolurus fordi (Lacertilia: Agamidae). Aust J Zool 26:653–672
Cogger HG (2000) Reptiles and amphibians of Australia. Ralph Curtis Publishing, Florida, USA
Conover DO (1984) Adaptive significance of temperature-dependent sex determination in a fish. Am Nat 123:297–313
Conover DO, Heins SW (1987) Adaptive variation in environmental and genetic sex determination in a fish. Nature 326:496–498
Cooper WE Jr (2003) Sexual dimorphism in distance from cover but not escape behavior by the keeled earless lizard Holbrookia propinqua. J Herpetol 37:374–378
Cree AM, Thompson MB, Daugherty CH (1995) Tuatara sex determination. Nature 375:543
Daan S, Dijkstra C, Weissing FJ (1996) An evolutionary explanation for seasonal trends in avian sex ratios. Behav Ecol 7:426–430
Doody JS, Georges A, Young JE (2004) Determinants of reproductive success and offspring sex in a turtle with environmental sex determination. Biol J Linn Soc 81:1–16
Doody SJ, Guarino E, Georges A, Corey B, Murray G, Ewert M (2006) Nest site choice compensates for climate effects on sex ratios in a lizard with environmental sex determination. Evol Ecol 20:307–330
El Mouden EH, Znari M, Pieau C (2001) Effects of incubation temperature on embryonic development and sex determination in the northern African agamid lizard, Agama impalearis. Herpetol J 11:101–108
Ewert MA, Nelson CE (1991) Sex determination in turtles: diverse patterns and some possible adaptive values. Copeia 1991:50–69
Fisher RA (1930) The genetical theory of natural selection. Clarendon Press, Oxford, UK
Ferguson MWJ, Joanen T (1982) Temperature of egg incubation determines sex in Alligator mississippiensis. Nature 296:850–853
Franco MG, Rubini PG Vecchi M (1982) Sex-determinants and their distribution in various populations of Musca domestica L. of western Europe. Genet Res 40:279–293
Greer AE (1989) The biology and evolution of Australian lizards. Surrey Beatty and Sons, Chipping Norton, Australia
Griffiths AD (1999) Demography and home range of the frillneck lizard, Chlamydosaurus kingii (Agamidae), in northern Australia. Copeia 1999:1089–1096
Harlow PS (2000) Incubation temperature determines hatchling sex in Australian rock dragons (Agamidae: genus Ctenophorus). Copeia 2000:958–964
Harlow PS (2001) The ecology of sex-determining mechanisms in Australian agamid lizards. Dissertation, Macquarie University
Harlow PS (2004) Temperature-dependent sex determination in lizards. In: Valenzuela N, Lance VA (eds) Temperature-dependent sex determination in vertebrates. Smithsonian Institution Press, Washington DC, pp 42–52
Harlow PS, Harlow MF (1997) Captive reproduction and longevity in the eastern water dragon (Physignathus lesueurii). Herpetofauna 27:14–19
Harlow PS, Shine R (1999) Temperature-dependent sex determination in the frillneck lizard, Chlamydosaurus kingii (Agamidae). Herpetologica 55:205–212
Harlow PS, Taylor JE (2000) Reproductive ecology of the jacky dragon (Amphibolurus muricatus): an agamid lizard with temperature-dependent sex determination. Aust Ecol 25:640–652
Janzen FJ, Paukstis GL (1991a) Environmental sex determination in reptiles: ecology, evolution, and experimental design. Quart Rev Biol 66:149–179
Janzen FJ, Paukstis GL (1991b) A preliminary test of the adaptive significance of temperature-dependent sex determination in reptiles. Evolution 45:435–440
Janzen FJ, Krenz JG (2004) Phylogenetics: which was first, TSD or GSD? In: Valenzuela N, Lance VA (eds) Temperature-dependent sex determination in vertebrates. Smithsonian Institution Press, Washington DC, pp 121–130
Janzen FJ, Phillips PC (2006) Exploring the evolution of environmental sex determination, especially in reptiles. J Evol Biol 19:1775–1784
Jenssen TA, Decourcy KR, Congdon JD (2005) Assessment in contests of male lizards (Anolis carolinensis): how should smaller males respond when size matters? Anim Behav 69:1325–1336
Jun-Yi JW, Kau-Hung L (1982) Population ecology of the lizard Japalurua swinhonis formosensis (Sauria: Agamidae) in Taiwan. Copeia 1982:425–434
Kozielska M, Pen I, Beukeboom LW, Weissing FJ (2006) Sex ratio selection and multi-factorial sex determination in the house fly: a dynamic model. J Evol Biol 19:879–888
Lagomarsino IV, Conover DO (1993) Variation in environmental and genotypic sex-determining mechanisms across a latitudinal gradient in the fish, Menidia menidia. Evolution 47:487–494
LeBas NR (2002) Mate choice, genetic incompatibility, and outbreeding in the ornate dragon lizard, Ctenophorus ornatus. Evolution 56:371–377
Lister BC, Aguayo AG (1992) Seasonality, predation, and the behaviour of a tropical mainland anole. J Anim Ecol 61:717–733
Manning A, Ehmann H (1991) A study of the activity and behaviour of the southern angle-headed dragon using the spool tracking technique. Herpetofauna 21:5–16
Olsson M (1992) Contest success in relation to body size and residency in male sand lizards, Lacerta agilis. Anim Behav 44:386–388
Olsson M, Shine R (1997) The seasonal timing of oviposition in sand lizards (Lacerta agilis): why early clutches are better. J Evol Biol 10:369–381
Olsson M, Madsen T (1998) Sexual selection and sperm competition in reptiles. In: Birkhead TR, Møller AP (eds) Sperm competition and sexual selection. Academic Press, San Diego, California, USA, pp 503–577
Osborne L (2005) Rival recognition in the territorial tawny dragon (Ctenophorus decresii). Acta Ethol 8:45–50
Pen I, Weissing FJ, Daan S (1999) Seasonal sex ratio trend in the European kestrel: an evolutionarily stable strategy analysis. Am Nat 153:384–397
Peters RA, Ord TJ (2003) Display response of the jacky dragon, Amphibolurus muricatus (Lacertilia: Agamidae), to intruders: a semi-markovian Process. Aust Ecol 28:449–506
Qualls FJ, Shine R (1998) Geographic variation in lizard phenotypes: the importance of incubation temperatures. Biol J Linn Soc 64:477–491
Qualls FJ, Shine R (2000) Post-hatching environment contributes greatly to phenotypic variation between two populations of the Australian garden skink, Lampropholis guichenoti. Biol J Linn Soc 71:315–341
Robert KA, Thompson MB (2001) Viviparous lizard selects sex of embryos. Nature 412:698–699
Sarre SD, Georges A, Quinn A (2004) The ends of a continuum: genetic and temperature-dependent sex determination in reptiles. Bioessays 26:639–645
Shine R (1999) Why is sex determined by nest temperature in many reptiles? Trend Ecol Evol 14:186–189
Shine R, Elphick MJ, Harlow PS (1995) Sisters like it hot. Nature 378:451–452
Shine R, Elphick MJ, Donnellan S (2002) Co-occurrence of multiple, supposedly incompatible modes of sex determination in a lizard population. Ecol Lett 5:486–489
Smallwood PD, Smallwood JA (1998) Seasonal shifts in sex ratios of fledgling American kestrels (Falco sparverius paulus): the early bird hypothesis. Evol Ecol 12:839–853
Stuart-Smith JN, Swain R, Welling A (2005) Reproductive ecology of the mountain dragon, Rankinia (Tympanocryptus) diemensis (Reptilia: Squamata: Agamidae) in Tasmania. Pap Proc R Soc Tas 139:23–28
Thompson GG, Thompson SA (2001) Behaviour and spatial ecology of Gilbert’s dragon Lophognathus gilberti (Agamidae: Reptilia). J R Soc West Aust 84:153–158
Tinkle DW (1967) The life history and demography of the side-blotched lizard, Uta stansburiana. Misc Publ Mus Zool Univ Michigan 132:1–182
Uller T, Olsson M (2006) No seasonal sex ratio shift despite sex-specific fitness returns of hatching date in a lizard with genotypic sex determination. Evolution 60:2131–2136
Uller T, Mott B, Odierna G, Olsson M (2006) Consistent sex ratio bias of individual female dragon lizards. Biol Lett 2:569–572
Uller T, Pen I, Wapstra E, Beukeboom L, Komdeur J (2007) Evolution of sex ratios and sex-determining systems. Trends Ecol Evol 22:292–297
Valenzuela N (2004) Evolution and maintenance of temperature-dependent sex determination. In: Valenzuela N, Lance VA (eds) Temperature-dependent sex determination in vertebrates. Smithsonian Institution Press, Washington DC, pp 131–147
Valenzuela N, Lance VA (2004) Temperature-dependent sex determination in vertebrates. Smithsonian Institution Press, Washington DC
Viets BE, Ewert MA, Talent LG, Nelson CE (1994) Sex-determining mechanisms in squamate reptiles. J Exp Zool 270:45–56
Wapstra E, Olsson M, Shine R, Edwards A, Swain R, Joss JMP (2004) Maternal basking determines offspring sex in a viviparous reptile. Biol Lett, Proc R Soc Lond B (suppl) 271:S230–S232
Warner DA, Shine R (2005) The adaptive significance of temperature-dependent sex determination: experimental tests with a short-lived lizard. Evolution 59:2209–2221
Warner DA, Shine R (2007a) Maternal nest-site choice in a lizard with temperature-dependent sex determination. Anim Behav (in press)
Warner DA, Shine R (2007b) Fitness of juvenile lizards depends on seasonal timing of hatching, not offspring body size. Oecologia 154:65–73
Werren JH, Charnov EL (1978) Facultative sex ratios and population dynamics. Nature 272:349–350
Witten GJ (1974) Population movements of the agamid lizard Amphibolurus nobbi. Aust Zool 18:129–132
Witten GJ, Heatwole H (1978) Preferred temperature of the agamid lizard Amphibolurus nobbi nobbi. Copeia 1978:362–364
Wright DD, Ryser JT, Kiltie RA (1995) First-cohort advantage hypothesis: a new twist on facultative sex ratio adjustment. Am Nat 145:133–145
Acknowledgements
Comments by M. Olsson, S. Parker, I. Pen, R. Radder, F. Seebacher, M. Thompson, and E. Wapstra improved this manuscript. DAW was supported by an International Postgraduate Research Scholarship and an International Postgraduate Award, TU by the Wenner-Gren Foundations and the Australian Research Council, and RS by the Australian Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Warner, D.A., Uller, T. & Shine, R. Fitness effects of the timing of hatching may drive the evolution of temperature-dependent sex determination in short-lived lizards. Evol Ecol 23, 281–294 (2009). https://doi.org/10.1007/s10682-007-9222-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-007-9222-4