Abstract
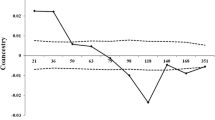
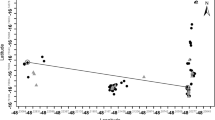
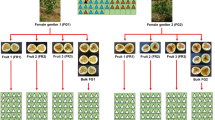
Genebank accessions stored as seed populations require periodic rejuvenation in order to maintain sufficient numbers of viable seeds. During rejuvenation the genetic composition of accessions may be altered for a variety of reasons, of which variation in pollination rates between plants is the least understood. In the present study, a paternity exclusion analysis was performed on a rejuvenated accession of perennial ryegrass. In addition, flowering data of the 49 parental plants were collected during the flowering season. The aim of the study was to determine how accurate variation in pollination rates between plants can be predicted from data on the spatial and temporal distribution of pollen release. The parental population and a total of 551 offspring from 12 progeny arrays were genotyped by means of molecular analysis. Using 25 microsatellites, paternity was identified for 81.9% of the offspring, while remaining ambiguities were resolved by AFLP analysis, except in four cases. Within the total sample 9 cases of contamination were observed. Mating within the study population was clearly non-random, as 61.9% of the identified pollen donors were located within 1 m distance from the mother plant. Observed pollination rates were very well described by an inverse quadratic function of inter-plant distance between potential mating pairs. Incorporation of the recorded flowering data in the calculation of expected pollination rates improved the goodness of fit with observed values by only 0.77%. Suggestions to reduce the variance in paternal contributions were presented. However, contamination was considered more threatening to the genetic integrity of perennial ryegrass germplasm than variation in pollination rates between plants, and indicated the need for improved measures to avoid gene flow from other germplasm.
Similar content being viewed by others
References
Barker, R.E., J.A. Kilgore, R.L. Cook, A.E. Garay & S.E. Warnke, 2001. Use of flow cytometry to determine ploidy level of ryegrass. Seed Sci Technol 29: 493–502.
Brownstein, M.J., J.D. Carpten & J.R. Smith, 1996. Modulation of non-templated nucleotide addition by Taq DNA polymerase: Primer modifications that facilitate genotyping. Biotechniques 20: 1004–1010.
Cunliffe, K.V., A.C. Vecchies, E.S. Jones et al., 2004. Assessment of gene flow using tetraploid genotypes of perennial ryegrass (Lolium perenne L.). Austr J Agr Res 55: 389–396.
Darlington, C.D., 1929. Chromosome behaviour and structural hybridity in the Tradescantiae. J Genet 21: 207–286.
De Nettancourt, D., 2000. Incompatibility and Incongruity in Wild and Cultivated Plants. Springer-Verlag, Berlin.
Elgersma, A., 1990. Heritability estimates of spaced-plant traits in three perennial ryegrass (Lolium perenne L.) cultivars. Euphytica 51: 163–172.
Falconer, D.S., 1981. Introduction to Quantitative Genetics. Longman, London.
Forster, J.W., E.S. Jones, R. Kölliker et al., 2001. DNA profiling in outbreeding forage species. In: R. Henry (Ed.), Plant Genotyping – the DNA Fingerprinting of Plants, pp. 299–320. CABI Press.
Fulton, T.M., J. Chunwongse & S.D. Tanksley, 1995. Microprep protocol for extraction of DNA from tomato and other herbaceous plants. Plant Mol Biol Rep 13: 207–209.
Galbraith, D.W., K.R. Harkins, J.M. Maddox et al., 1983. A rapid flow cytometric analysis of the plant cell in intact plant tissues. Science 220: 1049–1051.
Giddings, G.D., N.R. Sackville Hamilton & M.D. Hayward, 1997a. The release of genetically modified grasses. 1: Pollen dispersal to traps in Lolium perenne. Theor Appl Genet 94: 1000–1006.
Giddings, G.D., N.R. Sackville Hamilton & M.D. Hayward, 1997b. The release of genetically modified grasses. 2: The influence of wind direction on pollen dispersal. Theor Appl Genet 94: 1007–1014.
Guthridge, K.M., M.P. Dupal, R. Kölliker, E.S. Jones, K.F. Smith & J.W. Forster, 2001. AFLP analysis of genetic diversity within and between populations of perennial ryegrass (Lolium perenne L.). Euphytica 122: 191–201
Holmes, W., 1980. Grass, its Production and Utilization. Blackwell scientific publications, Oxford, UK.
ICONFORS, 2001. Improving germplasm conservation methods for perennial European forage species. http://www.igergru.bbsrc.ac.uk/iconfors/projectannouncement.htm.
Jansen, R.C. & A.P.M. den Nijs, 1993. A statistical method for estimating the proportion of unreduced pollen grains in perennial ryegrass (Lolium perenne L.) via the size of the pollen grains. Euphytica 70: 205–215.
Jarne, P. & P.J.L. Lagoda, 1996. Microsatellites, from molecules to populations and back. Trends Ecol Evol 11: 424–429.
Johnson, R.C., V.L. Bradley & M.A. Evans, 2002. Effective population size during grass germplasm seed regeneration. Crop Sci 42: 286–290.
Johnson, R.C., V.L. Bradley & M.A. Evans, 2004. Inflorescence sampling improves effective population size of grasses. Crop Sci 44: 1450–1455.
Jones, E.S., M.P. Dupal, R. Kölliker, M.C. Drayton & J.W. Forster, 2001. Development and characterisation of simple sequence repeat (SSR) markers for perennial ryegrass (Lolium perenne L.). Theor Appl Genet 102: 405–415.
Kubik, C., W.A. Meyer & B.S. Gaut, 1999. Assessing the abundance and polymorphism of simple sequence repeats in perennial ryegrass. Crop Sci 39: 1136–1141.
Kubik, C., M. Sawkins, W.A. Meyer & B.S. Gaut, 2001. Genetic diversity in seven perennial ryegrass (lolium perenne L.) cultivars based on SSR markers. Crop Sci 41: 1565–1572.
Lexer, C., B. Heinze, H. Steinkellner, S. Kampfer, B. Ziegenhagen & J. Glossl, 1999. Microsatellite analysis of maternal half-sib families of Quercus robur, pedunculate oak: Detection of seed contamination and inference of the seed parents from the offspring. Theor Appl Genet 99: 185–191.
Marum, P., I.D. Thomas & M. Veteläinen, 1998. Summary of germplasm holdings. In: Report of a Working Group on Forages. Sixth meeting, 6–8 March 1997, Beitostø len, Norway, European Co-operative Programme for Crop Genetic Resources Networks (ECP/GR), pp. 184–190. International Plant Genetic Resources Institute, Rome, Italy.
Mather, K., 1936. Segregation and linkage in autotetraploids. J Genet 32: 287–314.
Pemberton, J.M., J. Slate, D.R. Bancroft & J.A. Barrett, 1995. Nonamplifying alleles at microsatellite loci: A caution for parentage and population studies. Mol Ecol 4: 249–252.
Queller, D.C., J.E. Strassmann & C.R. Hughes, 1993. Microsatellites and kinship. Trends Ecol Evol 8: 285–288.
Ramsey,J. & D.W. Schemske, 2002. Neopolyploidy in flowering plants. Ann Rev Ecol Syst 33: 589–639.
Sackville Hamilton, N.R., 1998. The rationalization of regeneration methods: how far can we go? The example of forage grasses. In: T. Gass, L. Frese, F. Begemann & E. Lipman (Eds.), Implementation of the Global Plan of Action in Europe – Conservation and Sustainable Utilization of Plant Genetic Resources for Food and Agriculture. IPGRI, Rome, Italy.
Thorogood, D., W.J. Kaiser, J.G. Jones & I. Armstead, 2002. Self-incompatibility in ryegrass 12. Genotyping and mapping the S and Z loci of Lolium perenne L. Heredity 88: 385-390.
Van Treuren, R., N. Bas, P.J. Goossens, H. Jansen & L.J.M. van Soest, 2005. Genetic diversity in perennial ryegrass and white clover among old Dutch grasslands as compared to cultivars and nature reserves. Mol Ecol 14: 39–52.
Vos, P., R. Hogers, M. Bleeker et al., 1995. AFLP: a new technique for DNA fingerprinting. Nucl Acids Res 23: 4407–4414.
Weber, J.L. & C. Wong, 1993. Mutation of human short tandem repeats. Hum Mol Genet 2: 1123–1128.
Zeven, A.C. & J.M.J. de Wet, 1982. Dictionary of Cultivated Plants and their Regions of Diversity. Pudoc, Wageningen, the Netherlands.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Van Treuren, R., Goossens, P.J. & Ševčíková, M. Variation in effective pollination rates in relation to the spatial and temporal distribution of pollen release in rejuvenated perennial ryegrass. Euphytica 147, 367–382 (2006). https://doi.org/10.1007/s10681-005-9033-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-005-9033-z