Abstract
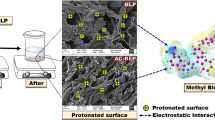
Chili stalk powder (CS), a non-conventional adsorbent, has been exercised for facile removal of cationic dyes from simulated and wastewater by batch technique. The prepared material has been characterized by Fourier-transform infrared spectroscopy (FTIR), Field emission scanning electron microscopy (FESEM), Brunauer–Emmett–Teller analysis (BET), powder X-ray diffraction (powder XRD), and pHZPC and tested best with methylene blue and crystal violet under ambient conditions. FTIR denotes the presence of carbonyl and polyphenolic groups, responsible for dye adsorption. BET surface area analysis evaluates the porous nature and specific surface area of the material, and powder XRD confirms its amorphous nature. The porous structure could be ascertained from the FESEM image, and energy dispersive X-ray analysis (EDX) confirms the elemental composition. The pH above pHzpc shows an increase in removal efficiency. The maximum adsorption capacities are 49.53 and 36.88 mg/g for methylene blue (MB) and crystal violet (CV) respectively. Linear as well as non-linear plots for kinetic and isotherm models were studied. Both dye uptake fits the linear plot of Langmuir adsorption isotherm (R2 = 0.999 and 0.995) and pseudo-second-order kinetics (R2 = 0.998 and 0.999). In the non-linear plot, the adsorption process for both dyes fit Langmuir (R2 = 0.999 for MB and R2 = 0.983 for CV) as well as Freundlich adsorption (R2 = 0.999 for MB and R2 = 0.994 for CV). 75.48% crystal violet (CV) and 73.35% methylene blue (MB) regeneration were successful in 1:1 methanol medium and reused for up to three cycles. The uptake mechanism is suggested to be a union of π-π stacking, electrostatic interaction, and weak hydrogen bonding. The material was tested with industrial effluent to prove its application in real wastewater treatment. Moreover, the material shows superior adsorption capacity than contemporary phytosorbents. To conclude, a zero-cost adsorbent using green chili stalk has been demonstrated for wastewater treatment.













Similar content being viewed by others
Data availability
Data produced and materials analyzed during the research are included in the manuscript and can be made available from the corresponding author on reasonable request.
References
Abbas, S., Javeed, T., Zafar, S., Taj, M. B., Ashraf, A. R., & Din, M. I. (2021). Adsorption of crystal violet dye by using a low-cost adsorbent–peanut husk. Desalination and Water Treatment, 233, 387–398. https://doi.org/10.5004/dwt.2021.27538
Adeyi, A. A., Jamil, S. N. A. M., Abdullah, L. C., Choong, T. S. Y., Lau, K. L., & Abdullah, M. (2019). Adsorptive removal of methylene blue from aquatic environments using thiourea-modified poly (acrylonitrile-co-acrylic acid). Materials, 12(11), 1734. https://doi.org/10.3390/ma12111734
Adeyi, A. A., Bitrus, N. K., Abdullah, L. C., Popoola, L. T., Chijioke-Okere, M., Omotara, O. O., & Saber, S. E. M. (2023). Effective sequestration of levofloxacin from wastewater by biochar-supported manganese dioxide composite: experimental study and modelling analyses. Environmental Engineering Research, 28(1). https://doi.org/10.4491/eer.2021.512.
Ahmad, M. A., Eusoff, M. A., Oladoye, P. O., Adegoke, K. A., & Bello, O. S. (2021). Optimization and batch studies on adsorption of methylene blue dye using pomegranate fruit peel based adsorbent. Chemical Data Collections, 32, 100676. https://doi.org/10.1016/j.cdc.2021.100676
Aziz, A., Hassan, H., Yahaya, N. K. E. M., Karim, J., Ahmad, M. A., & Karim, K. A. (2021). Carbonized chilli stalk adsorbent to remove methylene blue dye from aqueous solution: Equilibrium and kinetics studies. In IOP Conference Series: Earth and Environmental Science, 765(1), 012105. https://doi.org/10.1088/1755-1315/765/1/012105
Bayomie, O. S., Kandeel, H., Shoeib, T., Yang, H., Youssef, N., & El-Sayed, M. M. H. (2020). Novel approach for effective removal of methylene blue dye from water using fava bean peel waste. Scientific Reports, 10(1), 7824. https://doi.org/10.1038/s41598-020-64727-5
Cazetta, A. L., Vargas, A. M., Nogami, E. M., Kunita, M. H., Guilherme, M. R., Martins, A. C., ... & Almeida, V. C. (2011). NaOH-activated carbon of high surface area produced from coconut shell: kinetics and equilibrium studies from the methylene blue adsorption. Chemical Engineering Journal, 174(1), 117–125. https://doi.org/10.1016/j.cej.2011.08.058.
Daneshvar, E., Vazirzadeh, A., Niazi, A., Kousha, M., Naushad, M., & Bhatnagar, A. (2017). Desorption of methylene blue dye from brown macroalga: Effects of operating parameters, isotherm study and kinetic modeling. Journal of Cleaner Production, 152, 443–453. https://doi.org/10.1016/j.jclepro.2017.03.119
Das, M., Samal, A. K., & Mehar, N. (2020). Butea monosperma leaf as an adsorbent of methylene blue: Recovery of the dye and reuse of the adsorbent. International Journal of Environmental Science and Technology, 17(4), 2105–2112. https://doi.org/10.1007/s13762-019-02480-7
Dey, S., Pal, K., & Sarkar, S. (2007). Thermally induced reversible conformational changes in the host–guest adduct of meso-tetramethyltetrakis(ethyl)calix[4]pyrrole. Tetrahedron Letters, 48(31), 5481–5485. https://doi.org/10.1016/j.tetlet.2007.05.168
Dey, S., Bhagat, P., Mohanta, J., & Dey, B. (2022). Methylene blue removal using eucalyptus leaves: a low cost protocol towards environmental sustainability. European Journal of Advanced Chemistry Research, 3(1), 1–11. https://doi.org/10.24018/ejchem.2022.3.1.82
Dey, S., Chakraborty, R., Mohanta, J., & Dey, B. (2022). Tricosanthes cucumerina: a potential biomass for efficient removal of methylene blue from water. Bioremediation Journal, 1–15. https://doi.org/10.1080/10889868.2022.2086530
Elwakeel, K. Z., Elgarahy, A. M., Elshoubaky, G. A., & Mohammad, S. H. (2020). Microwave assist sorption of crystal violet and Congo red dyes onto amphoteric sorbent based on upcycled Sepia shells Chemistry. Journal of Environmental Health Science and Engineering, 18(1), 35–50. https://doi.org/10.1007/s40201-019-00435-1
Fiaz, R., Hafeez, M., & Mahmood, R. (2019). Ficcus palmata leaves as a low-cost biosorbent for methylene blue: Thermodynamic and kinetic studies. Water Environment Research, 91(8), 689–699. https://doi.org/10.1002/wer.1093
Hasan, M. M., Shenashen, M. A., Hasan, M. N., Znad, H., Salman, M. S., & Awual, M. R. (2021). Natural biodegradable polymeric bioadsorbents for efficient cationic dye encapsulation from wastewater. Journal of Molecular Liquids, 323, 114587. https://doi.org/10.1016/j.molliq.2020.114587
Ihaddaden, S., Aberkane, D., Boukerroui, A., & Robert, D. (2022). Removal of methylene blue (basic dye) by coagulation-flocculation with biomaterials (bentonite and Opuntia ficus indica). Journal of Water Process Engineering, 49, 102952. https://doi.org/10.1016/j.jwpe.2022.102952
Ilgin, P., Onder, A., Kıvanç, M. R., Ozay, H., & Ozay, O. (2023). Adsorption of methylene blue from aqueous solution using poly (2-acrylamido-2-methyl-1-propanesulfonic acid-co-2-hydroxyethyl methacrylate) hydrogel crosslinked by activated carbon. Journal of Macromolecular Science, Part A, 60(2), 135–149. https://doi.org/10.1080/10601325.2023.2165945
Imam, S. S., & Babamale, H. F. (2020). A short review on the removal of rhodamine B dye using agricultural waste-based adsorbents. Asian Journal of Chemical Sciences, 7(1), 25–37. https://doi.org/10.9734/ajocs/2020/v7i119013
Kumari, R., & Dey, S. (2019). A breakthrough column study for removal of malachite green using coco-peat. International Journal of Phytoremediation, 21(12), 1263–1271. https://doi.org/10.1080/15226514.2019.1633252
Kumari, R., Khan, M. A., Mahto, M., Qaiyum, M. A., Mohanta, J., Dey, B., & Dey, S. (2020a). Dewaxed honeycomb as an economic and sustainable scavenger for malachite green from water. ACS Omega, 5(31), 19548–19556. https://doi.org/10.1021/acsomega.0c02011
Kumari, R., Mohanta, J., Dey, B., & Dey, S. (2020b). Eucalyptus leaf powder as an efficient scavenger for Congo red from water: Comprehensive batch and column investigation. Separation Science and Technology, 55(17), 3047–3059. https://doi.org/10.1080/01496395.2019.1670208
Kumari, R., & Dey, S. (2018). Facile removal of congo red using Mahua (Madhuca longifolia) seeds, a low cost adsorbent. International Journal of Green and Herbal Chemistry, 7(2), 237–250. https://doi.org/10.24214/IJGHC/GC/7/2/23750.
Kumari, R., Mohanta, J., Sambasivaiah, B., Qaiyum, M. A., Dey, B., Samal, P. P., Dutta, S., & Dey, S. (2023). Dye sequestration from aqueous phase using natural and synthetic adsorbents in batch mode: present status and future perspectives. In International Journal of Environmental Science and Technology, 1–20. https://doi.org/10.1007/s13762-023-04782-3
Lee, T. C., Wang, S., Huang, Z., Mo, Z., Wang, G., Wu, Z., Liu, C., Han, H., & Ko, T. H. (2019). Tea stem as a sorbent for removal of methylene blue from aqueous phase. Advances in Materials Science and Engineering, 2019, 1–15. https://doi.org/10.1155/2019/9723763
Li, T., Liu, L., Zhang, Z., & Han, Z. (2020). Preparation of nanofibrous metal-organic framework filter for rapid adsorption and selective separation of cationic dye from aqueous solution. Separation and Purification Technology, 237, 116360. https://doi.org/10.1016/j.seppur.2019.116360
Loulidi, I., Boukhlifi, F., Ouchabi, M., Amar, A., Jabri, M., Kali, A., Chraibi, S., Hadey, C., & Aziz, F. (2020). Adsorption of crystal violet onto an agricultural waste residue: Kinetics, isotherm, thermodynamics, and mechanism of adsorption. Science World Journa, 2020(3), 1–9. https://doi.org/10.1155/2020/5873521
Mahato, R., Qaiyum, M. A., Samal, P. P., Dutta, S., Dey, B., & Dey, S. (2022). Exploring the promising potential of fallen bamboo leaves (Bambusa bambos) for efficient removal of crystal violet from wastewater. International Journal of Phytoremediation, 25(8), 1042–1051. https://doi.org/10.1080/15226514.2022.2125498
Mishra, S., Cheng, L., & Maiti, A. (2021). The utilization of agro–biomass/byproducts for effective bio-removal of dyes from dyeing wastewater: A comprehensive review. Journal of Environmental Chemical Engineering, 9(1), 104901. https://doi.org/10.1016/j.jece.2020.104901
Mohanta, J., Dey, B., & Dey, S. (2020). Sucrose-triggered, self-sustained combustive synthesis of magnetic nickel oxide nanoparticles and efficient removal of malachite green from water. ACS Omega, 5(27), 16510–16520. https://doi.org/10.1021/acsomega.0c00999
Morin-Crini, N., Lichtfouse, E., Fourmentin, M., Ribeiro, A. R. L., Noutsopoulos, C., Mapelli, F., ... & Crini, G. (2022). Removal of emerging contaminants from wastewater using advanced treatments. A review. Environmental Chemistry Letters, 20(2), 1333–1375. https://doi.org/10.1007/s10311-021-01379-5.
Mouni, L., Belkhiri, L., Bollinger, J. C., Bouzaza, A., Assadi, A., Tirri, A., Dahmoune, F., Madani, K., & Remini, H. (2018). Removal of methylene blue from aqueous solutions by adsorption on Kaolin: Kinetic and equilibrium studies. Applied Clay Science, 153, 38–45. https://doi.org/10.1016/j.clay.2017.11.034
Muhammad, U. L., Zango, Z. U., & Kadir, H. A. (2019). Crystal violet removal from aqueous solution using corn stalk biosorbent. Science World Journal, 14(1), 133–138. https://doi.org/10.13140/RG.2.2.10223.48808.
Nipa, S. T., Rahman, M. W., Saha, R., Hasan, M. M., & Deb, A. (2019). Jute stick powder as a potential low-cost adsorbent to uptake methylene blue from dye enriched wastewater. Desalination and Water Treatment, 153, 279–287. https://doi.org/10.5004/dwt.2019.23767
Nooraee Nia, N., Rahmani, M., Kaykhaii, M., & Sasani, M. (2017). Evaluation of eucalyptus leaves as an adsorbent for decolorization of methyl violet (2B) dye in contaminated waters: Thermodynamic and Kinetics model. Modeling Earth Systems and Environment, 3(2), 825–829. https://doi.org/10.1007/s40808-017-0338-4
Oloo, C. M., Onyari, J. M., Wanyonyi, W. C., Wabomba, J. N., & Muinde, V. M. (2020). Adsorptive removal of hazardous crystal violet dye form aqueous solution using Rhizophora mucronata stem-barks: Equilibrium and kinetics studies. Environmental Chemistry and Ecotoxicology, 2, 64–72. https://doi.org/10.1016/j.enceco.2020.05.001
Onder, A., & Ozay, H. (2021). Highly efficient removal of methyl orange from aqueous media by amine functional cyclotriphosphazene submicrospheres as reusable column packing material. Chemical Engineering and Processing-Process Intensification, 165, 108427. https://doi.org/10.1016/j.cep.2021.108427
Onder, A., Ilgin, P., Ozay, H. A. V. A., & Ozay, O. (2022). Preparation of composite hydrogels containing fly ash as low-cost adsorbent material and its use in dye adsorption. International Journal of Environmental Science and Technology, 19(8), 7031–7048. https://doi.org/10.1007/s13762-021-03622-6
Onder, A., Kıvanç, M. R., Ilgin, P., Ozay, H., & Ozay, O. (2023). Synthesis of p (HEMA-co-AETAC) nanocomposite hydrogel with vinyl-function montmorillonite nanoparticles and effective removal of methyl orange from aqueous solution. Journal of Macromolecular Science, Part A, 60(2), 108–123. https://doi.org/10.1080/10601325.2023.2169155
Ozsoy, F., Ozdilek, B., Onder, A., et al. (2022). Graphene nanoplate incorporated Gelatin/poly(2-(Acryloyloxy)ethyl trimethylammonium chloride) composites hydrogel for highly effective removal of Alizarin Red S from aqueous solution. Journal of Polymer Research, 29, 481. https://doi.org/10.1007/s10965-022-03327-5
Qaiyum, M. A., Mohanta, J., Kumari, R., Samal, P. P., Dey, B., & Dey, S. (2022). Alkali treated water chestnut (Trapa natans L.) shells as a promising phytosorbent for malachite green removal from water. International Journal of Phytoremediation, 24(8), 822–830. https://doi.org/10.1080/15226514.2021.1977912
Qaiyum, M. A., Sahu, P. R., Samal, P. P., Dutta, S., Dey, B., & Dey, S. (2022). Towards a win-win chemistry: extraction of C.I. orange from Kamala fruit (Mallotus philippensis), and simultaneous exercise of its peels for the removal of methylene blue from water. International Journal of Phytoremediation, 25(7), 907–916. https://doi.org/10.1080/15226514.2022.2119936
Rashidi, H. R., Sulaiman, N. M. N., Hashim, N. A., Hassan, C. R. C., & Ramli, M. R. (2015). Synthetic reactive dye wastewater treatment by using nano-membrane filtration. Desalination and Water Treatment, 55(1), 86–95. https://doi.org/10.1080/19443994.2014.912964
Rathi, B. S., Kumar, P. S., & Vo, D. V. N. (2021). Critical review on hazardous pollutants in water environment: Occurrence, monitoring, fate, removal technologies and risk assessment. Science of the Total Environment, 797, 149134. https://doi.org/10.1016/j.scitotenv.2021.149134
Rout, A., Qaiyum, M. A., Samal, P. P., Dutta, S., Dey, B., & Dey, S. (2022). Brinjal (Solanum melongena) stalk waste as an effective scavenger for Eriochrome Black-T from water and wastewater: An approach towards waste to best. International Journal of Phytoremediation, 25(8), 956–964. https://doi.org/10.1080/15226514.2022.2123445
Sakr, F., Alahiane, S., Sennaoui, A., Dinne, M., Bakas, I., & Assabbane, A. (2020). Removal of cationic dye (methylene blue) from aqueous solution by adsorption on two type of biomaterial of South Morocco. Materials Today: Proceedings, 22(1), 93–96. https://doi.org/10.1016/j.matpr.2019.08.101
Samal, P. P., Kumari, J., Qaiyum, M. A., Mohanta, J., Kumari, R., Dutta, S., Dey, B., & Dey, S. (2022). Thiosulfate impregnated spent tea leaves for the remarkable uptake of malachite green. International Journal of Phytoremediation, 25(11), 1413–1422. https://doi.org/10.1080/15226514.2022.2161465
Sharma, S., & Kaur, A. (2018). Various methods for removal of dyes from industrial effluents - a review. Indian Journal of Science and Technology, 11(12), 1–21. https://doi.org/10.17485/ijst/2018/v11i12/120847.
Sharma, S., Dutta, V., Raizada, P., Hosseini-Bandegharaei, A., Thakur, V., Nguyen, V. H., ... & Singh, P. (2021). An overview of heterojunctioned ZnFe2O4 photocatalyst for enhanced oxidative water purification. Journal of Environmental Chemical Engineering, 9(5), 105812. https://doi.org/10.1016/j.jece.2021.105812.
Singh, R., Singh, T. S., Odiyo, J. O., Smith, J. A., & Edokpayi, J. N. (2020). Evaluation of methylene blue sorption onto low-cost biosorbents: Equilibrium, kinetics, and thermodynamics. Journal of Chemistry, 2020, 1–11. https://doi.org/10.1155/2020/8318049
Solangi, N. H., Kumar, J., Mazari, S. A., Ahmed, S., Fatima, N., & Mubarak, N. M. (2021). Development of fruit waste derived bio-adsorbents for wastewater treatment: A review. Journal of Hazardous Materials, 46, 125848. https://doi.org/10.1016/j.jhazmat.2021.125848
Sulyman, M., & Gierak, A. (2020). Green environmental approach for adsorption of hazardous dye from water using tree and sea plant leaves (Dead L.). Acta Scientific Agriculture, 4(2), 01–10. https://doi.org/10.31080/asag.2020.04.green-environmental-approach-for-adsorption-of-hazardous-dye-from-water-using-tree-and-sea-plant-leaves-dead-l.
Taqui, S. N., Cs, M., Goodarzi, M. S., Elkotb, M. A., Khatoon, B. A., Soudagar, M. E. M., Koki, I. B., Elfasakhany, A., Khalifa, A. S., Ali, M. A., Saifullah, Z., Siddiqui, M. I. H., Safaei, M. R., & Saleel, C. A. (2021). Sustainable adsorption method for the remediation of crystal violet dye using nutraceutical industrial fenugreek seed spent. Applied Sciences, 11(16), 7635. https://doi.org/10.3390/app11167635
Wiriyathamcharoen, S., Sarkar, S., Jiemvarangkul, P., Nguyen, T. T., Klysubun, W., & Padungthon, S. (2020). Synthesis optimization of hybrid anion exchanger containing triethylamine functional groups and hydrated Fe (III) oxide nanoparticles for simultaneous nitrate and phosphate removal. Chemical Engineering Journal, 381, 122671. https://doi.org/10.1016/j.cej.2019.122671
Yakutha, S. A., Taqui, S. N., Syed, U. T., & Syed, A. A. (2019). Nutraceutical industrial chillies stalk waste as a new adsorbent for the removal of acid violet 49 from water and textile industrial effluent: Adsorption isotherms and kinetic models. Desalination and Water Treatment, 155, 94–112. https://doi.org/10.5004/dwt.2019.23997
Zhang, G., Yi, L., Deng, H., & Sun, P. (2014). Dyes adsorption using a synthetic carboxymethyl cellulose-acrylic acid adsorbent. Journal of Environmental Sciences, 26(5), 1203–1211. https://doi.org/10.1016/S1001-0742(13)60513-6
Acknowledgements
The author acknowledges the MRC MNIT Jaipur for FTIR analysis.
Author information
Authors and Affiliations
Contributions
Ankita Panda: wrote the original manuscript, methodology, and artwork; Priyanka Priyadarsini Samal: referencing, editing, and data curation; Md. Atif Qaiyum: data curation and validation; Banashree Dey: formal analysis and review; Soumen Dey: conceptualization, review, editing, and overall supervision.
Corresponding author
Ethics declarations
Ethics approval
No approval is needed.
Consent to participate
Not applicable.
Consent for publication
The authors give their consent for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Novelty statement
Green chili stalks powder has been demonstrated for scavenging methylene blue and crystal violet. It is available all over the seasons, organic, economical, free of hazardous chemicals, uncontaminated nature, easily accessible in bulk amounts and simple disposal makes the whole adsorption process environmentally benign. Maximum adsorption capacities were obtained as 49.53 and 36.88 mg/g for methylene blue (MB) and crystal violet (CV) respectively, which is greater than that of other adsorbents. The adsorption process follows the linear Langmuir model and linear pseudo-second-order kinetics. Dye-loaded spent material could be regenerated using aqueous methanolic solution, and be reused. This greener method sets a perfect example of waste to the best.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Panda, A., Samal, P.P., Qaiyum, M.A. et al. Think before throw: waste chili stalk powder for facile scavenging of cationic dyes from water. Environ Monit Assess 196, 118 (2024). https://doi.org/10.1007/s10661-023-12243-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-12243-0