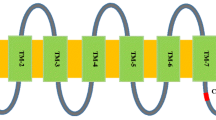
Abstract
Powdery mildew (PM), caused by Podosphaera xanthii, is one of the most serious disease in bitter gourd (Momordica charantia). Specific members of the Mildew Locus O (MLO) gene family act as susceptibility factors by recognizing PM pathogens and the corresponding loss-of-function mutations confer broad-spectrum durable host resistance. However, the potential PM-susceptibility genes in bitter gourd have not been identified yet. Therefore, genome-wide characterization and expression analysis of the bitter gourd MLO gene family were carried out in the present study. The results suggested that 15 MLO homologs were present in the bitter gourd genome. Gene organization, cis-elements, conserved amino acid residues and motifs were also investigated and analyzed. Phylogenetic analysis revealed that these identified MLO proteins clustered in six well-defined clades. It was noteworthy that McMLO7b, a bitter gourd MLO homolog falling in clade V which clustered all the known PM susceptibility factors of dicots, was up-regulated more than 6-fold at an early infection stage (6 h) and was inferred to be a candidate gene of PM susceptibility factor. In addition, exogenous salicylic acid (SA) treatment enhanced the PM resistance of bitter gourd, accompanied by a significantly elevated level of endogenous SA. However, McMLO7b showed nearly equivalent expression in SA-treated plants compared to the mock-treated seedlings at the early PM infection stage. MLO proteins were directly targeted by PM to trigger pathogenesis, therefore, a weak effect of exogenous SA on interaction between bitter gourd and PM could be inferred. To conclude, the results will be helpful in proposing better strategies to control PM disease with either resistant cultivars or cultivation management.






Similar content being viewed by others
References
Ablazov, A., & Tombuloglu, H. (2016). Genome-wide identification of the mildew resistance locus O (MLO) gene family in novel cereal model species Brachypodium distachyon. European Journal of Plant Pathology, 145, 239–253.
Acevedo-Garcia, J., Kusch, S., & Panstruga, R. (2014). Magical mystery tour: MLO proteins in plant immunity and beyond. New Phytologist, 204(2), 273–281.
Aist, J. R., Gold, R. E., & Bayles, C. J. (1987). Evidence for the involvement of molecular components of papillae in mlo resistance to barley powdery mildew. Phytopathology, 77, 17–32.
Appiano, M., Pavan, S., Catalano, D., Zheng, Z., Bracuto, V., Lotti, C., Visser, R. G. F., Ricciardi, L., & Bai, Y. (2015). Identification of candidate MLO powdery mildew susceptibility genes in cultivated Solanaceae and functional characterization of tobacco NtMLO1. Transgenic Research, 24, 847–858.
Bai, Y., Pavan, S., Zheng, Z., Zappel, N., Reinstädler, A., Lotti, C., De Giovanni, C., Ricciardi, L., Lindhout, P., Visser, R. G. F., Theres, K., & Panstruga, R. (2008). Naturally occurring broad-spectrum powdery mildew resistance in a central American tomato accession is caused by loss of MLO function. Molecular Plant-Microbe Interactions, 21, 30–39.
Becker, B., & Marin, B. (2009). Streptophyte algae and the origin of embryophytes. Annals of Botany, 103, 999–1004.
Büschges, R., Hollricher, K., Panstruga, R., Simons, G., Wolter, M., Frijters, A., Daelen, R., Lee, T., Diergaarde, P., Groenendijk, J., Töpsch, S., Vos, P., Salamini, F., & Schulze-Lefert, P. (1997). The barley Mlo gene: A novel control element of plant pathogen resistance. Cell, 88(5), 695–705.
Chen, Z., Noir, S., Kwaaitaal, M., Hartmann, H. A., Wu, M. J., Mudgil, Y., Sukumar, P., Muday, G., Panstruga, R., & Jones, A. M. (2009). Two seven-transmembrane domain MILDEW RESISTANCE LOCUS O proteins cofunction in Arabidopsis root thigmomorphogenesis. The Plant Cell, 21(7), 1972–1991.
Chen, C., Chen, H., Zhang, Y., Thomas, H. R., Frank, M. H., He, Y., & Xia, R. (2020). TBtools: an integrative toolkit developed for interactive analyses of big biological data. Molecular Plant, 13(8), 1194–1202.
Consonni, C., Humphry, M. E., Hartmann, H. A., Livaja, M., Durner, J., Westphal, L., Vogel, J., Lipka, V., Kemmerling, B., Schulze-Lefert, P., Somerville, S. C., & Panstruga, R. (2006). Conserved requirement for a plant host cell protein in powdery mildew pathogenesis. Nature Genetics, 38(6), 716–720.
Dean, R., van Kan, J. A. L., Pretorius, Z. A., Hammond-Kosack, K. E., Di Pietro, A., Spanu, P. D., Rudd, J. J., Dickman, M., Kahmann, R., Ellis, J., & Foster, G. D. (2012). The top 10 fungal pathogens in molecular plant pathology. Molecular Plant Pathology, 13, 414–430.
Deshmukh, R., Singh, V. K., & Singh, B. D. (2014). Comparative phylogenetic analysis of genome-wide Mlo gene family members from Glycine max and Arabidopsis thaliana. Molecular Genetics and Genomics, 289(3), 345–359.
Devoto, A., Piffanelli, P., Nilsson, I., Wallin, E., Panstruga, R., von Heijne, G., & Schulze-Lefert, P. (1999). Topology, subcellular localization, and sequence diversity of the Mlo family in plants. The Journal of Biological Chemistry, 274, 34993–35004.
Devoto, A., Hartmann, H. A., Piffanelli, P., Elliott, C., Simmons, C., Taramino, G., Goh, C. S., Cohen, F. E., Emerson, B. C., Schulze-Lefert, P., & Panstruga, R. (2003). Molecular phylogeny and evolution of the plants-specific seven-transmembrane MLO family. Journal of Molecular Evolution, 56, 77–88.
Dhillon, N. S., Sanguansil, S., Srimat, S., Schafleitner, R., Manjunath, B., Agarwal, P., Xiang, Q., Masud, M., Myint, T., Hanh, N., Cuong, T., Balatero, C. H., Salutan-Bautista, V., Pitrat, M., Lebeda, A., & McCreight, J. D. (2018). Cucurbit powdery mildew-resistant bitter gourd breeding lines reveal four races of Podosphaera xanthii in Asia. HortScience, 53(3), 337–341.
Dhillon, N. S., Sanguansil, S., Srimat, S., Laenoi, S., Schafleitner, R., Pitrat, M., & McCreight, J. D. (2019). Inheritance of resistance to cucurbit powdery mildew in bitter gourd. HortScience, 54(6), 1013–1016.
Elliott, C., Zhou, F., Spielmeyer, W., Panstruga, R., & Schulze-Lefert, P. (2002). Functional conservation of wheat and rice Mlo orthologs in defense modulation to the powdery mildew fungus. Molecular Plant-Microbe Interactions, 15(10), 1069–1077.
Elliott, C., Müller, J., Miklis, M., Schulze-lefert, P., & Panstruga, R. (2005). Conserved extracellular cysteine residues and cytoplasmic loop-loop interplay are required for functionality of the heptahelical MLO protein. Biochemical Journal, 385, 243–254.
Feechan, A., Jermakow, A. M., Torregrosa, L., Panstruga, R., & Dry, I. B. (2008). Identification of grapevine MLO gene candidates involved in susceptibility to powdery mildew. Functional Plant Biology, 35, 1255–1266.
Feechan, A., Jermakow, A. M., Ivancevic, A., Godfrey, D., Pak, H., Panstruga, R., & Dry, I. B. (2013). Host cell entry of powdery mildew is correlated with endosomal transport of antagonistically acting VvPEN1 and VvMLO to the papilla. Molecular Plant-Microbe Interactions, 26, 1138–1150.
Feng, J., Zhang, M., Yang, K., & Zheng, C. (2020). Salicylic acid-primed defence response in octoploid strawberry ‘Benihoppe’ leaves induces resistance against Podosphaera aphanis through enhanced accumulation of proanthocyanidins and upregulation of pathogenesis-related genes. BMC Plant Biology, 20, 149.
Filiz, E., & Vatansever, R. (2018). Genome-wide identification of mildew resistance locus O (MLO) genes in tree model poplar (Populus trichocarpa): Powdery mildew management in woody plants. European Journal of Plant Pathology, 152(1), 95–109.
Fung, R. W., Gonzalo, M., Fekete, C., Kovacs, L. G., He, Y., Marsh, E., McIntyre, L. M., Schachtman, D. P., & Qiu, W. (2008). Powdery mildew induces defense-oriented reprogramming of the transcriptome in a susceptible but not in a resistant grapevine. Plant Physiology, 146(1), 236–249.
Gaj, T., Gersbach, C. A., & Barbas III, C. F. (2013). ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends in Biotechnology, 31, 397–405.
Humphry, M., Reinstaedler, A., Ivanov, S., Bisseling, T., & Panstruga, R. (2011). Durable broad-spectrum powdery mildew resistance in pea er1 plants is conferred by natural loss-of-function mutations in PsMLO1. Molecular Plant Pathology, 12, 866–878.
Iovieno, P., Andolfo, G., Schiavulli, A., Catalano, D., Ricciardi, L., Frusciante, L., Ercolano, M. R., & Pavan, S. (2015). Structure, evolution and functional inference on the Mildew Locus O (MLO) gene family in three cultivated Cucurbitaceae spp. BMC Genomics, 16(1), 1112.
Jiao, Y. T., Xu, W. R., Duan, D., Wang, Y. J., & Nick, P. (2016). A stilbene synthase allele from a Chinese wild grapevine confers resistance to powdery mildew by recruiting salicylic acid signalling for efficient defence. Journal of Experimental Botany, 67, 5841–5856.
Jiwan, D., Roalson, E. H., Main, D., & Dhingra, A. (2013). Antisense expression of peach mildew resistance locus O (PpMlo1) gene confers cross-species resistance to powdery mildew in Fragaria × ananassa. Transgenic Research, 22, 1119–1131.
Kessler, S. A., Shimosato-Asano, H., Keinath, N. F., Wuest, S. E., Ingram, G., Panstruga, R., & Grossniklaus, U. (2010). Conserved molecular components for pollen tube reception and fungal invasion. Science, 330, 968–971.
Kim, D. S., & Hwang, B. K. (2012). The pepper MLO gene, CaMLO2, is involved in the susceptibility cell-death response and bacterial and oomycete proliferation. Plant Journal, 72, 843–855.
Kim, M. C., Panstruga, R., Elliott, C., Muller, J., Devoto, A., Yoon, H. W., Park, H. C., Cho, M. J., & Schulze-Lefert, P. (2002). Calmodulin interacts with MLO protein to regulate defence against mildew in barley. Nature, 416, 447–451.
Konishi, S., Sasakuma, T., & Sasanuma, T. (2010). Identification of novel MLO family members in wheat and their genetic characterization. Genes & Genetic Systems, 85(3), 167–175.
Kusch, S., Pesch, L., & Panstruga, R. (2016). Comprehensive phylogenetic analysis sheds light on the diversity and origin of the MLO family of integral membrane proteins. Genome Biology and Evolution, 8(3), 878–895.
Lim, C. W., & Lee, S. C. (2013). Functional roles of the pepper MLO protein gene, CaMLO2, in abscisic acid signaling and drought sensitivity. Plant Molecular Biology, 85, 1–10.
Liu, Q., & Zhu, H. (2008). Molecular evolution of the MLO gene family in Oryza sativa and their functional divergence. Gene, 409(1–2), 1–10.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods, 25, 402–408.
Meuwly, P., & Metraux, J. P. (1993). Ortho-anisic acid as internal standard for the simultaneous quantitation of salicylic acid and its putative biosynthetic precursors in cucumber leaves. Analytical Biochemistry, 214, 500–505.
Nekrasov, V., Wang, C., Win, J., Lanz, C., Weigel, D., & Kamoun, S. (2017). Rapid generation of a transgene-free powdery mildew resistant tomato by genome deletion. Scientific Reports, 7(1), 482.
Panstruga, R. (2005a). Serpentine plant MLO proteins as entry portals for powdery mildew fungi. Biochemical Society Transactions, 33, 389–392.
Panstruga, R. (2005b). Discovery of novel conserved peptide domains by ortholog comparison within plant multi-protein families. Plant Molecular Biology, 59, 485–500.
Pavan, S., Schiavulli, A., Appiano, M., Marcotrigiano, A. R., Cillo, F., Visser, R. G., Bai, Y., Lotti, C., & Ricciardi, L. (2011). Pea powdery mildew er1 resistance is associated to loss-of-function mutations at a MLO homologous locus. Theoretical and Applied Genetics, 123, 1425–1431.
Pessina, S., Pavan, S., Catalano, D., Gallotta, A., Visser, R. G., Bai, Y., Malnoy, M., & Schouten, H. J. (2014). Characterization of the MLO gene family in Rosaceae and gene expression analysis in Malus domestica. BMC Genomics, 15, 618.
Qin, B., Wang, M., He, H. X., Xiao, H. X., Zhang, Y., & Wang, L. F. (2019). Identification and characterization of a potential candidate Mlo gene conferring susceptibility to powdery mildew in rubber tree. Phytopathology, 109(7), 1236–1245.
Reinstädler, A., Müller, J., Czembor, J. H., Piffanelli, P., & Panstruga, R. (2010). Novel induced mlo mutant alleles in combination with site-directed mutagenesis reveal functionally important domains in the heptahelical barley Mlo protein. BMC Plant Biology, 10(1), 31.
Sakata, Y., Kubo, N., Morishita, M., Kitadani, E., Sugiyama, M., & Hirai, M. (2006). QTL analysis of powdery mildew resistance in cucumber (cucumis sativus L.). Theoretical and Applied Genetics, 112(2), 243.
Talieva, M. N., & Kondrat'eva, V. V. (2002). Influence of exogenous salicylic acid on the level of phytohormones in tissues of Phlox paniculata and Phlox setacea leaves with special reference to resistance against the powdery mildew causative agent Erysiphe cichoracearum DC. f. phlogis Jacz. Biology Bulletin of the Russian Academy of Sciences, 29(6), 551–554.
Tayeh, C., Randoux, B., Bourdon, N., & Reignault, P. (2013). Lipid metabolism is differentially modulated by salicylic acid and heptanoyl salicylic acid during the induction of resistance in wheat against powdery mildew. Journal of Plant Physiology, 170(18), 1620–1629.
Thompson, J. D., Higgins, D. G., & Gibson, T. J. (1994). CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Research, 22, 4673–4680.
Urasaki, N., Takagi, H., Natsume, S., Uemura, A., Taniai, N., Miyagi, N., Fukushima, M., Suzuki, S., Tarora, K., Tamaki, M., Sakamoto, M., Terauchi, R., & Sakamoto, M. (2017). Draft genome sequence of bitter gourd (Momordica charantia), a vegetable and medicinal plant in tropical and subtropical regions. DNA Research, 24(1), 51–58.
Wang, Y., Cheng, X., Shan, Q., Zhang, Y., Liu, J., Gao, C., & Qiu, J. (2014). Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nature Biotechnology, 32, 947–951.
Wang, X., Ma, Q., Dou, L., Liu, Z., Peng, R., & Yu, S. (2016). Genome-wide characterization and comparative analysis of the MLO gene family in cotton. Plant Physiology and Biochemistry, 103, 106–119.
Zheng, Z., Nonomura, T., Appiano, M., Pavan, S., Matsuda, Y., Toyoda, H., Wolters, A. M., Visser, R. G., & Bai, Y. (2013). Loss of function in Mlo orthologs reduces susceptibility of pepper and tomato to powdery mildew disease caused by Leveillula taurica. PLoS One, 8(7), e70723.
Acknowledgements
This work was supported by the Natural Science Foundation of Jiangsu Province (SBK2020042988).
Author information
Authors and Affiliations
Contributions
Longzheng Chen designed and wrote this manuscript. Jing Liu performed the bioinformatics analysis. Zhiyang Liu performed the experiments. Pengfei Xia and Xihan Yuan made contributions to reply the reviewers’ comments. Yu Ning critically revised this manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of financial and non-financial interests.
Ethical statement
This research does not involve any animal or human participants.
Supplementary Information
Table S1
Information of MLO homologs in Cucurbitaceae species used for construction of phylogenetic tree. (XLSX 14 kb)
Table S2
Detailed information of primer sequences used for qRT-PCR analysis. (XLSX 10 kb)
Table S3
Conserved amino acids analysis of McMLO proteins against the reported 30 conserved amino acids. (XLSX 13 kb)
Rights and permissions
About this article
Cite this article
Chen, L., Liu, J., Liu, Z. et al. Genome-wide identification and expression analysis of the MLO gene family reveal a candidate gene associated with powdery mildew susceptibility in bitter gourd (Momordica charantia). Eur J Plant Pathol 159, 163–178 (2021). https://doi.org/10.1007/s10658-020-02152-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-020-02152-0