Abstract
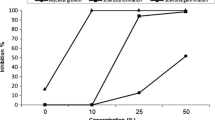
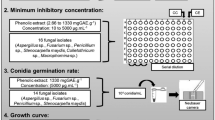
Organic solvents are widely used in bioassay experiments to dissolve water-insoluble technical grade fungicides, and the added organic solvents may substantially influence toxicity of the fungicides tested. In this study, toxicity of the four organic solvents methanol, ethanol, acetone, and dimethyl sulfoxide (DMSO) to Sclerotinia sclerotiorum and Botrytis cinerea were determined. EC50 values of methanol, ethanol, acetone and DMSO against S. sclerotiorum were 13.5, 24.3, 21.8, and 20.1 μl/ml, respectively, and EC50 values of the four solvents against B. cinerea were 20.7, 30.2, 57.2, and 20.6 μl/ml, respectively. At solvent concentrations from 1 to 10 μl/ml (0.1 % to 1 % v/v), the EC50 values of the fungicides dimethachlon and pyraclostrobin decreased along with the increase of solvent concentration. Interaction analysis based on inhibition percentages of mycelial growth on potato dextrose agar (PDA) identified synergistic, additive, and rarely antagonistic interactions between fungicide and solvent. Permeability of cell membrane of S. sclerotiorum grown on PDA amended with ethanol, acetone or DMSO at 1 % was significantly higher than at 0.1 % and that of the solvent-free control. This study demonstrated that solvent concentrations may greatly influence toxicity of fungicides and therefore, solvent concentrations should be kept as low as possible. These results are valuable for choosing appropriate solvents and concentrations in assays of fungicide toxicity.



Similar content being viewed by others
References
Bangham, A. D., Standish, M. M., & Miller, N. (1965). Cation permeability of phospholipid model membranes: effect of narcotics. Nature, 208, 1295–1297.
Boland, G. J., & Hall, R. (1994). Index of plant hosts of Sclerotinia sclerotiorum. Canadian Journal of Plant Pathology, 16, 93–108.
Bolton, D. M., Thomma, B. P., & Nelson, B. D. (2006). Sclerotinia sclerotiorum (lib.) de bray: biology and molecular traits of a cosmopolitan pathogen. Molecular Plant Pathology, 7, 1–16.
Bowman, M. C., Oller, W. L., Cairns, T., Gosnell, A. B., & Oliver, K. H. (1981). Stressed bioassay systems for rapid screening of pesticide residues. Archives of Environmental Contamination and Toxicology, 10, 9–24.
Burrell, R. E. & Corke, C. T. (1980). Interactions of the solvent acetone with the fungicides benomyl and captan in fungal assays. Bulletin of Environmental Contamination and Toxicology, 25, 554–561.
Duan, Y. B., Ge, C. Y., & Zhou, M. G. (2014). Molecular and biochemical characterization of Sclerotinia sclerotiorum laboratory mutants resistant to dicarboximide and phenylpyrrole fungicides. Journal of Pest Science, 87, 221–230.
Isken, S. & de Bont, J. A. (1998). Bacteria tolerant to organic solvents. Extremophiles, 2, 229–238.
Liang, H. J., Di, Y. L., Li, J. L., You, H., & Zhu, F. X. (2015). Baseline sensitivity of pyraclostrobin and toxicity of SHAM to Sclerotinia sclerotiorum. Plant Disease, 99, 267–273.
Ma, H. X., Feng, X. J., Chen, Y., Chen, C. J., & Zhou, M. G. (2009). Occurrence and characterization of dimethachlon insensitivity in Sclerotinia sclerotiorum in Jiangsu Province of China. Plant Disease, 93, 36–42.
Mariscal, A., Peinado, M. T., Carnero-Varo, M., & Fernández-Crehuet, J. (2003). Influence of organic solvents on the sensitivity of a bioluminescence toxicity test with Vibrio harveyi. Chemosphere, 50, 349–54.
Purdy, L. H. (1979). Sclerotinia sclerotiorum: history, diseases and symptomatology, host range, geographic distribution and impact. Phytopathology, 69, 875–880.
Rekha, R., Mouli, B. C., Kumari, S. G. V., Indumathi, C., & Mithyantha, M. S. (2006). Studies on the identification of suitable solvents for microbial bioassay. Current Science, 90, 1663–1667.
Sikkema, J., de Bont, J. A., & Poolman, B. (1995). Mechanisms of membrane toxicity of hydrocarbons. Microbiological Reviews, 59, 201–222.
Stratton, G. W. (1985). The influence of solvent type on solvent-pesticide interactions in bioassays. Archives of Environmental Contamination and Toxicology, 14, 651–658.
Stratton, G. W. (1989). Factors affecting magnitude of toxicological interactions in microbial bioassays. Environmental Toxicology and Water Quality, 4, 425–435.
Stratton, G. W., Burrell, R. E., & Corke, C. T. (1982). Technique for identifying and minimizing solvent-pesticide interactions in bioassays. Archives of Environmental Contamination and Toxicology, 11, 437–445.
Stratton, G. W., Burrell, R. E., Kurp, M. L., & Corke, C. T. (1980). Interactions between the solvent acetone and the pyrethroid insecticide permethrin on activities of the blue-green alga Anabaena. Bulletin of Environmental Contamination and Toxicology, 24, 562–569.
Stratton, G. W., & Corke, C. T. (1981). Effect of acetone on the toxicity of atrazine towards photosynthesis in Anabaena. Journal of Environmental Science and Health (Part B), 16, 21–33.
Veloukas, T. & Karaoglanidis, G. S. (2012). Biological activity of the succinate dehydrogenase inhibitor fluopyram against Botrytis cinerea and fungal baseline sensitivity. Pest Management Science, 68, 858–864.
Wadhwani, T., Desai, K., Patel, D., Lawani, D., Bahaley, P., Johsi, P., & Kothari, V. (2008). Effect of various solvents on bacterial growth in context of determining MIC of various antimicrobials. The Internet Journal of Microbiology, 7. http://ispub.com/IJMB/7/1/5909. Accessed 22 May 2015.
Weber, F. J. & de Bont, J. A. (1996). Adaptation mechanisms of microorganisms to the toxic effects of organic solvents on membranes. Biochimica et Biophysica Acta, 1286, 225–245.
Wise, K. A., Bradley, C. A., Pasche, J. S., Gudmestad, N. C., Dugan, F. M., & Chen, W. (2008). Baseline sensitivity of Ascochyta rabiei to azoxystrobin, pyraclostrobin, and boscalid. Plant Disease, 92, 295–300.
Yang, L. J. & Bai, Y. L. (2012). Strobilurin fungicide – pyraclostrobin. Modern Agriculture, 11, 46–56.
Yuan, N. N., Chen, S. N., Zhai, L. X., Schnabel, G., Yin, L. F., & Luo, C. X. (2013). Baseline sensitivity of Monilia yunnanensis to the DMI fungicides tebuconazole and triadimefon. European Journal of Plant Pathology, 136, 651–655.
Zahran, H. E. M., Kawanna, M. A., & Bosly, H. A. (2013). Larvicidal activity and joint action toxicity of certain combating agents on Culex pipiens L. mosquitoes. Annual Review and Research in Biology, 3, 1055–1065.
Zhou, F., Zhang, X. L., Li, J. L., & Zhu, F. X. (2014). Dimethachlon resistance in Sclerotinia sclerotiorum in China. Plant Disease, 98, 1221–1226.
Acknowledgments
This study was supported by National Natural Science Foundation of China (31371964) and the Special Fund for Agro-scientific Research in the Public Interest (201103016). The authors are grateful to Prof. Daohong Jiang and Guoqing Li of Huazhong Agricultural University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liang, HJ., Lu, XM., Zhu, ZQ. et al. Effect of organic solvent on fungicide toxicity to Sclerotinia sclerotiorum and Botrytis cinerea . Eur J Plant Pathol 146, 37–45 (2016). https://doi.org/10.1007/s10658-016-0889-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-0889-7