Abstract
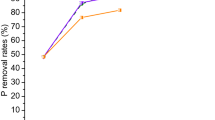
Heavy metal pollution has always been a serious environmental problem widely concerned by researchers all around the world. On the other side, the accumulation of biowastes has also occupied a large amount of space and caused a series of environment pollution. In this study, the waste oyster shell, was applied as a type of biogenic carbonate material for Pb(II) removal from the aquatic environment, and further as a remediation agent for metal stabilization in the contaminated river sediment. After simple pretreatment, the oyster shell powder (OSP) was characterized, and the results showed that the prepared OSP is mainly composed of calcite with particle size of micron-level. The OSP exhibited excellent Pb(II) adsorption performance, with the adsorption capacity as 639.9 mg/g through adsorption isotherm study. Furthermore, the OSP was applied to remediate the collected river sediment artificially contaminated by Pb(II). It was found that the proportion of residual Pb fraction (F4) was greatly increased from 39.6% of the original sediment to 76.7% in the 14-day incubated sediment with OSP. The Pb(II) concentration after leaching procedure was decreased from 810.7 to 108.6 μg/L even after 5-day incubation. Therefore, this study shows the potential of using waste oyster shell as adsorbent and amendment agent for effective metal immobilization in both aquatic and sediment systems.
Graphic abstract





Similar content being viewed by others
References
Alvarenga, R. A. F. D., Galindro, B. M., Helpa, C. D. F., & Soares, S. R. (2012). The recycling of oyster shells: An environmental analysis using life cycle assessment. Journal of Environmental Management, 106, 102–109. https://doi.org/10.1016/j.jenvman.2012.04.017
Audry, S., Schäfer, J., Blanc, G., & Jouanneau, J. M. (2004). Fifty-year sedimentary record of heavy metal pollution (Cd, Zn, Cu, Pb) in the lot river reservoirs (France). Environmental Pollution, 132(3), 413–426. https://doi.org/10.1016/j.envpol.2004.05.025
Basu, M., Guha, A. K., & Ray, L. (2017). Adsorption of lead on cucumber peel. Journal of Cleaner Production, 151, 603–615. https://doi.org/10.1016/j.jclepro.2017.03.028
Bozbaş, S. K., & Boz, Y. (2016). Low-cost biosorbent: Anadara inaequivalvis shells for removal of Pb(II) and Cu(II) from aqueous solution. Process Safety and Environmental Protection, 103, 144–152. https://doi.org/10.1016/j.psep.2016.07.007
Cerbo, A. A. V., Ballesteros, F., Chen, T. C., & Lu, M. C. (2017). Solidification/stabilization of fly ash from city refuse incinerator facility and heavy metal sludge with cement additives. Environmental Science and Pollution Research, 24(2), 1748–1756. https://doi.org/10.1007/s11356-016-7943-z
Che, Y., He, Q., & Lin, W. Q. (2003). The distributions of particulate heavy metals and its indication to the transfer of sediments in the Changjiang Estuary and Hangzhou Bay, China. Marine Pollution Bulletin, 46(1), 123–131. https://doi.org/10.1016/s0025-326x(02)00355-7
Dai, L., Wang, L., Li, L., Liang, T., Zhang, Y., Ma, C., et al. (2018). Multivariate geostatistical analysis and source identification of heavy metals in the sediment of Poyang Lake in China. Science of the Total Environment, 621, 1433–1444. https://doi.org/10.1016/j.scitotenv.2017.10.085
Du, Y., Lian, F., & Zhu, L. (2011). Biosorption of divalent Pb, Cd and Zn on aragonite and calcite mollusk shells. Environmental Pollution, 159(7), 1763–1768. https://doi.org/10.1016/j.envpol.2011.04.017
Fonti, V., Beolchini, F., Rocchetti, L., & Dell’Anno, A. (2015). Bioremediation of contaminated marine sediments can enhance metal mobility due to changes of bacterial diversity. Water Research, 68, 637–650. https://doi.org/10.1016/j.watres.2014.10.035
Guru, P. S., & Dash, S. (2014). Sorption on eggshell waste—A review on ultrastructure, biomineralization and other applications. Advances in Colloid and Interface Science, 209, 49–67. https://doi.org/10.1016/j.cis.2013.12.013
Hu, H., Zhang, Q., Yuan, W., Li, Z., Zhao, Y., & Gu, W. (2019). Efficient Pb removal through the formations of (basic) carbonate precipitates from different sources during wet stirred ball milling with CaCO3. Science of the Total Environment, 664, 53–59. https://doi.org/10.1016/j.scitotenv.2019.01.424
Imamoglu, M., Şahin, H., Aydın, Ş, Tosunoğlu, F., Yılmaz, H., & Yıldız, S. Z. (2016). Investigation of Pb(II) adsorption on a novel activated carbon prepared from hazelnut husk by K2CO3 activation. Desalination and Water Treatment, 57(10), 4587–4596. https://doi.org/10.1080/19443994.2014.995135
Ip, C. C. M., Li, X. D., Zhang, G., Wai, O. W. H., & Li, Y. S. (2007). Trace metal distribution in sediments of the pearl river estuary and the surrounding coastal area, South China. Enviromental Pollution, 147(2), 311–323. https://doi.org/10.1016/j.envpol.2006.06.028
Islam, M. S., Choi, W. S., Nam, B., Yoon, C., & Lee, H. J. (2017a). Needle-like iron oxide@CaCO3 adsorbents for ultrafast removal of anionic and cationic heavy metal ions. Chemical Engineering, 307, 208–219. https://doi.org/10.1016/j.cej.2016.08.079
Islam, M. N., Taki, G., Nguyen, X. P., Jo, Y. T., Kim, J., & Park, J. H. (2017b). Heavy metal stabilization in contaminated soil by treatment with calcined cockle shell. Environmental Science and Pollution Research, 24, 7177–7183. https://doi.org/10.1007/s11356-016-8330-5
Jacob, J. J., Varalakshmi, R., Gargi, S., Jayasri, M. A., & Suthindhiran, K. (2018). Removal of Cr (III) and Ni (II) from tannery effluent using calcium carbonate coated bacterial magnetosomes. NPJ Clean Water, 1(1), 1. https://doi.org/10.1038/s41545-018-0001-2
Jung, S., Heo, N. S., Kim, E. J., Oh, S. Y., Lee, H. U., Kim, I. T., et al. (2016). Feasibility test of waste oyster shell powder for water treatment. Process Safety and Environmental Protection, 102, 129–139. https://doi.org/10.1016/j.psep.2016.03.004
Köhler, S. J., Cubillas, P., Rodríguez-Blanco, J. D., Bauer, C., & Prieto, M. (2007). Removal of cadmium from wastewaters by aragonite shells and the influence of other divalent cations. Environmental Scicence & Technology, 41(1), 112–118. https://doi.org/10.1021/es060756j
Lee, M. E., Park, J. H., & Chung, J. W. (2019). Comparison of the lead and copper adsorption capacities of plant source materials and their biochars. Journal of Environmental Management, 236, 118–124. https://doi.org/10.1016/j.jenvman.2019.01.100
Liao, D., Zheng, W., Li, X., Yang, Q., Yue, X., Guo, L., et al. (2010). Removal of lead(II) from aqueous solutions using carbonate hydroxyapatite extracted from eggshell waste. Journal of Hazardous Materials, 177(1–3), 126–130. https://doi.org/10.1016/j.jhazmat.2009.12.005
Lim, A. P., Zulkeflee, Z., & Aris, A. Z. (2016). Effective removal of lead (II) ions by dead calcareous skeletons: Sorption performance and influencing factors. Water Science and Technology, 74(7), 1577–1584. https://doi.org/10.2166/wst.2016.368
Liu, Y., Tang, Y., Wang, P., & Zeng, H. (2019a). Carbonaceous halloysite nanotubes for the stabilization of Co, Ni, Cu and Zn in river sediments. Environmental Science: Nano, 6(8), 2420–2428.
Liu, Y., Tang, Y., Zhong, G., & Zeng, H. (2019b). A comparison study on heavy metal/metalloid stabilization in Maozhou river sediment by five types of amendments. Journal of Soils and Sediments, 19, 3922–3933. https://doi.org/10.1007/s11368-019-02310-w
Melichová, Z., & Ľuptáková, A. (2016). Removing lead from aqueous solutions using different low-cost abundant adsorbents. Desalination and Water Treatment, 57(11), 5025–5034. https://doi.org/10.1080/19443994.2014.999713
Moon, D. H., Cheong, K. H., Khim, J., Grubb, D. G., & Ko, I. (2011). Stabilization of Cu-contaminated army firing range soils using waste oyster shells. Environmental Geochemistry and Health, 33(S1), 159–166. https://doi.org/10.1007/s10653-010-9358-y
Ok, Y. S., Lee, S. S., Jeon, W.-T., Oh, S.-E., Usman, A. R. A., & Moon, D. H. (2011a). Application of eggshell waste for the immobilization of cadmium and lead in a contaminated soil. Environmental Geochemistry and Health, 33(S1), 31–39. https://doi.org/10.1007/s10653-010-9362-2
Ok, Y. S., Lim, J. E., & Moon, D. H. (2011b). Stabilization of Pb and Cd contaminated soils and soil quality improvements using waste oyster shells. Environmental Geochemistry and Health, 33(1), 83–91. https://doi.org/10.1007/s10653-010-9329-3
Ok, Y. S., Oh, S.-E., Ahmad, M., Hyun, S., Kim, K.-R., Moon, D. H., et al. (2010). Effects of natural and calcined oyster shells on Cd and Pb immobilization in contaminated soils. Environmental Earth Sciences, 61(6), 1301–1308. https://doi.org/10.1007/s12665-010-0674-4
Peng, W., Li, X., Xiao, S., & Fan, W. (2018). Review of remediation technologies for sediments contaminated by heavy metals. Journal of Soils and Sediments, 18(4), 1701–1719. https://doi.org/10.1007/s11368-018-1921-7
Rađenović, D., Kerkez, Đ, Pilipović, D. T., Dubovina, M., Grba, N., Krčmar, D., et al. (2019). Long-term application of stabilization/solidification technique on highly contaminated sediments with environment risk assessment. Science of the Total Environment, 684, 186–195. https://doi.org/10.1016/j.scitotenv.2019.05.351
Wang, L., Chen, L., Cho, D.-W., Tsang, D. C. W., Yang, J., Hou, D., et al. (2019). Novel synergy of Si-rich minerals and reactive MgO for stabilisation/solidification of contaminated sediment. Journal of Hazardous Materials, 365, 695–706. https://doi.org/10.1016/j.jhazmat.2018.11.067
Wu, Q., Chen, J., Clark, M., & Yu, Y. (2014). Adsorption of copper to different biogenic oyster shell structures. Applied Surface Science, 311, 264–272. https://doi.org/10.1016/j.apsusc.2014.05.054
Xia, W.-Y., Feng, Y.-S., Jin, F., Zhang, L.-M., & Du, Y.-J. (2017). Stabilization and solidification of a heavy metal contaminated site soil using a hydroxyapatite based binder. Construction and Building Materials, 156, 199–207. https://doi.org/10.1016/j.conbuildmat.2017.08.149
Zhang, M., Wang, X., Yang, L., & Chu, Y. (2019a). Research on progress in combined remediation technologies of heavy metal polluted sediment. International Journal of Environmental Research and Public Health, 16(24), 5098. https://doi.org/10.3390/ijerph16245098
Zhang, Z., Chen, H., Guo, G., & Li, F. (2019b). Amendments for simultaneous stabilization of lead, zinc, and cadmium in smelter-contaminated topsoils. Environmental Engineering Science, 36(3), 326–334. https://doi.org/10.1089/ees.2018.0231
Zhou, Q., Liao, B., Lin, L., Qiu, W., & Song, Z. (2018). Adsorption of Cu(II) and Cd(II) from aqueous solutions by ferromanganese binary oxide–biochar composites. Science of the Total Environment, 615, 115–122. https://doi.org/10.1016/j.scitotenv.2017.09.220
Acknowledgements
This work is supported financially by the National Natural Science Foundation of China (NSFC) (41977329; 21707063) and the Science and Technology Innovation Committee of Shenzhen Municipality (JCYJ20160429191618506). This work is also sponsored by the Guangdong Provincial Key Laboratory of Soil and Groundwater Pollution Control (No. 2017B030301012) and the State Environmental Protection Key Laboratory of Integrated Surface Water-Groundwater Pollution Control.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This work does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhong, G., Liu, Y. & Tang, Y. Oyster shell powder for Pb(II) immobilization in both aquatic and sediment environments. Environ Geochem Health 43, 1891–1902 (2021). https://doi.org/10.1007/s10653-020-00768-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-020-00768-z