Abstract
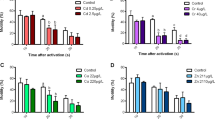
The effects of the world wide-distributed chemical bisphenol A (BPA) on the endocrine system of vertebrates have been demonstrated in several studies. Here, we report on the impact of BPA (0, 5, 50 and 500 μg/l nominally, deduced effective concentrations 0, 0.24, 2.4, and 24.1 μg/l, respectively, all at 15 °C) on the 70 kD stress protein family (hsp70), the 90 kD stress protein family (hsp90), and gonad histology of the crustacean Gammarus fossarum exposed in artificial indoor streams. The animals were exposed for a maximum of 103 days and samples were taken at the beginning and at days 34, 69 and 103 of the experiment. Exposure to BPA resulted in accelerated maturation of oocytes in females and in a decline in the number and size of early vitellogenic oocytes. The level of hsp90, which plays a pivotal role in vertebrate sex steroid signal transduction, was significantly reduced by BPA. In all five streams, measured parameters did not indicate any captivity stress for a period of 69 days. Beyond this time, the mortality rate and proteotoxic effects, the latter measured by hsp70 expression, were found to be elevated.








Similar content being viewed by others
References
Andersen H.R., Halling-Soerensen B., Kusk K.O., (1999). A parameter for detecting estrogenic exposure in the copepod Acartia tonsaEcotoxicol. Environ. Safety 44: 56–61
Borgmann U., (1996). Systematic analysis of aqueous ion requirements of Hyaella azteca: a standard artificial medium including the essential bromide ionArch. Environ. Contam. Toxicol. 30: 356–63
Bradford M.M., (1976). A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principles of protein-dye bindingAnal. Biochem. 72: 248–54
Colburn T., (2002). Clues from wildlife to create an assay from thyroid system disruptionEnv. Health Persp. 110(3): 363–7
de Fur P.L., Crane M., Ingersoll C., Tattersfield L., (1999). Endocrine Disruption in Invertebrates: Endocrinology, Testing, and Assessment SETAC Press Pensacola, FL
de Waal M., Portman J., Voogt P.A., (1982). Steroid receptors in invertebrates. A specific 17β-estradiol binding protein in a sea starMar. Biol. Lett. 3: 317–23
di Cosmo A., di Cristo C., Paolucci M., (2002). A Estradiol−17β receptor in the reproductive system of the female of Octopus vulgaris: characterization and immunolocalizationMol. Reprod. Dev. 61: 367–75
Duft M., Schulte-Oehlmann U., Weltje L., Tillmann M., Oehlmann J., (2003). Stimulated embryo production as a parameter of estrogenic exposure via sediments in the freshwater mudsnail Potamopyrgus antipodarumAquatic Toxicol. 64: 437–49
European Commission – European Chemical Bureau (2003). Risk assessment report bisphenol A. Online available at: http://ecb.jrc.it/existing-chemicals
Gullette L.J., Gunderson M.P., (2001). Alterations in development of reproductive and endocrine system of wildlife populations exposed to endocrine-disrupting contaminants Reproduction 122: 857–64
Jobling S., Casey D., Rodgers-Gray T., Oehlmann J., Schulte-Oehlmann U., Pawlowski S., Braunbeck T., Turner A.P., Tyler C.R., (2002). Comparitative responses of molluscs and fish to environmental estrogens and an estrogenic effluentAquatic Toxicol. 65: 205–20
Jungmann D., Brust K., Licht O., Mählmann J., Schmidt J., Nagel R., (2001). Artificial indoor streams as a method to investigate the impact of chemicals on lotic communitiesEnviron. Sci. Pollut. Res. 8(1): 49–55
Köhler A., Triebskorn R., (2004). Histologische Untersuchungen In: Jungmann D., Köhler A., Köhler H.-R., Ladewig V., Licht O., Ludwichowski K.-U., Schirling M., Triebskorn R., Nagel R., (eds), Umweltchemikalien mit Wirkung auf das Hormonsystem – TV 5: Wirkung von Xenohormonen in aquatischen Ökosystemen Teil I: Freilanduntersuchungen. German Federal Environmental Agency (UBA) Report Berlin, Germany pp. 89–115
Ladewig V., (2004). Der Einfluss von hormonell wirksamen Umweltchemikalien auf die Populationsökologie von Gammarus fossarum. PhD thesis Dresden University of Technology Germany
Licht O., Jungmann D., Ladewig V., Ludwichowksi K.-U., Nagel R., (2004). Bisphenol-A in artificial indoor streams: I. Substance dynamics and effects on aufwuchsEcotoxicology 13(7): 657–66
McLachlen J.A., (2001). Environmental signaling: what embryos and evolution teach us about endocrine disrupting chemicalsEndocr. Rev. 22(3): 319–41
Oehlmann J., Schulte-Oehlmann U., Tillmann M., Markert B., (2000). Effects of endocrine disruptors on prosobranch snails (Mollusca: Gastropoda) in the laboratory. Part 1: bisphenol A and octylphenol as xeno-estrogensEcotoxicology 9: 383–97
Papaconstantinou A.D., Fisher B.R., Umbreit T.H., Goering P.L., Lappas N.T., Brown K.M., (2001). Effects of beta estradiol and bisphenol A on heat shock protein levels and localization in the mouse uterus are antagonized by the antiestrogen ICI 182,780 Toxicol. Sci. 63: 173–80
Pascoe D., Caroll K., Karntanut W., Watts M.M., (2002). Toxicity of 17α-Enthinylestradiol and Bisphenol A to the freshwater cnidarian Hydra vulgarisArch. Environ. Contam. Toxicol. 43: 56–63
Pöckl M., Humpesch U.H., (1990). Intra- and inter-specific variations in egg survival and brood development time for Austrian populations of Gammarus fossarum and G. roeseli (Crustacea Amphipoda)Freshwater Biol. 23: 441–55
Pratt W.B., Toft D.O., (1997). Steroid receptor interactions with heat shock protein and immunophilin chaperonesEndocr. Rev. 18(3): 306–60
Rachmandran C., Catelli M.G., Schneider W., Shyamala G., (1988). Estrogenic regulation of uterin 90-kilodalton heat shock proteinEndocrinology 123(2): 956–61
Richardson K.C., Jarret L., Finke E.H., (1960). Embedding in epoxy resin for ultrathin sectioning in electron microscopyStain Technol. 35: 313–25
Sabbah M., Radanyi C., Redeuilh G., Baulieu E.-E., (1996). The 90 kDa heat shock protein (hsp90) modulates the binding of the oestrogen receptor to its cognate DNA Biochem. J. 314: 205–13
Schirling, M., Jungmann, D., Ladewig, V., Nagel, R., Triebskorn, R., Köhler and H.-R. (in press) Endocrine effects in Gammarus fossarum (Amphipoda)? Importance of wastewater effluents, temporal variability and spatial aspects for free-living populations. Arch. Environ. Contam. Toxicol
Schirling M., Triebskorn R., Köhler H.-R., (2004). Variation in stress protein levels (hsp70 and hsp90) in relation to oocyte development in Gammarus fossarum (Koch 1835) Invertebr. Reprod. Dev. 45(2): 161–7
Schönfelder G., Wittfoht W., Hopp H., Talsness C.E., Paul M., Chahoud I., (2002). Parental bisphenol A accumulation in the human maternal-fetal-placental unit Environ. Health Perspect. 110(11): 703–7
Segner H., Caroll K., Fenske M., Janssen C.R., Maack G., Pascoe D., Schaefers C., Vandenbergh G.F., Watts M.M., Wenzel A., (2003). Identification of endocrine effects in aquatic vertebrates and invertebrates. Report from the European IDEA projectEcotoxicol. Environ. Safety 54(4): 302–14
Staples C.A., Dorn P.B., Klecka G.M., O’Block S.T., Harris L.R., (1998). A review on the environmental fate, effects, and exposure of bisphenol AChemosphere 36(10): 2149–73
Staples C.A., Woodburn K., Caspers N., Hall A.T., Klécka G.M., (2002). A weight of evidence approach to aquatic hazard assessment of bisphenol AHum. Ecol. Risk Assess. 8(5): 1083–105
Stoker C., Rey F., Rodriguez H., Ramos J.G., Sirosky P., Larriera A., Luque E.H., Muñoz-de-Toro H., (2003). Sex reversal effects on Caiman latirostris exposed to environmentally relevant doses of the xenoestrogen bisphenol AGen. Comp. Endocrinol. 133: 287–96
Shymala G., Gauthier Y., Morre S.K., Cateli M.G., Ulrich S.J., (1989). Estrogenic regulation of murin uterin 90 kDa heat shock protein gene expressionMol. Cell Biol. 9(8): 3567–70
Tan-Fermin J.D., Pudadera R.A., (1989). Ovarian maturation stages in Wild Giant Tiger Prawn, Penaeus monodont (Fabricius)Aquaculture 77: 229–42
Thornton J.W., Need E., Crews D., (2003). Resurrecting the ancestral steroid receptor: ancient origin of estrogen signalling Science 301: 1714–7
Triebskorn R., Böhmer J., Braunbeck T., Honnen W., Köhler H.-R., Lehmann R., Oberemm A., Schwaiger J., Segner H., Schüürmann G., Traunspurger W., (2001). The project VALIMAR (VALIdation of bioMARkers for the assessment of small stream pollution): objectives, experimental design, summary of results, and recommendations for the application of biomarkers in risk assessmentJ. Aquat. Ecosys. Stress Recov. 8: 161–78
Watts M.M., Pascoe D., Caroll K., (2001a). Survival and precopulatory behaviour of Gammarus pulex (L.) exposed to two xenoestrogensWater Res. 35(10): 2347–52
Watts M.M., Pascoe D., Caroll K., (2001b). Chronic exposure to 17α-Ethinylestradiol and Bisphenol A – effects on development and reproduction in the freshwater invertebrate Chironomus riparius (Diptera: Chironomidae)Aquatic Toxicol. 55: 113–26
Watts M.M., Pascoe D., Caroll K., (2002). Population responses of the freshwater amphipod, Gammarus pulex (L.) to an environmental estrogen, 17α-ethinylestradiolEnv. Toxicol. Chem. 21: 445–50
Acknowledgments
The authors thank A. Köhler for assistance in sectioning and histological quantification, and A. Woitschella for the modified design of Fig. 2. Furthermore, we are grateful to the German Federal Environmental Agency (Umweltbundesamt, Berlin, FKZ 299 65 221/05) for funding. The helpful comments of two anonymous referees are acknowledged as well.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schirling, M., Jungmann, D., Ladewig, V. et al. Bisphenol A in Artificial Indoor Streams: II. Stress Response and Gonad Histology in Gammarus fossarum (Amphipoda). Ecotoxicology 15, 143–156 (2006). https://doi.org/10.1007/s10646-005-0044-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-005-0044-2