Synopsis
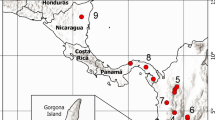
We analyzed variation in allozymes and mating preferences in 12 populations across much of the range of the sailfin molly, Poecilia latipinna. Sailfin mollies can be sympatric with its sexual parasite Amazon mollies, P. formosa. Amazon mollies must co-exist and mate with bisexual males of closely related species (including sailfin mollies) to induce embryogenesis but inheritance is strictly maternal. Where sailfin and Amazon mollies are sympatric there is evidence of reproductive character displacement as males show a significantly stronger mating preference for sailfin molly females over Amazon mollies compared to preferences of males from allopatric populations. From the allozyme data we found a moderate amount of genetic variation across all populations but this variation did not reveal significant partitioning between sympatric and allopatric populations. Additionally, we found no evidence for isolation by distance as genetic distance was not significantly correlated with geographic distance. While allozyme variation also did not significantly correlate with male mating preferences, there was a significant correlation between male mating preferences and geographic distance. This correlation between mating preferences and geographic distance may have arisen from coevolution with Amazon mollies resulting in reproductive character displacement. Taken together, the distribution of genetic and behavioral variation among sympatric and allopatric populations suggests that behavioral evolution has outpaced evolution at the allozyme loci we examined in P. latipinna.
Similar content being viewed by others
References
J.C. Avise J.C. Trexler J. Travis W. Nelson (1991) ArticleTitlePoeciliamexicanais the recent female parent of the unisexual fish P. formosa Evolution 45 1530–1533
J.S. Balsano E.M. Rasch P.J. Monaco (1989) The evolutionary ecology of Poeciliaformosaand its triploid associates G.K. Meffe F.F. Snelson (Eds) Ecology and Evolution of Livebearing Fishes (Poeciliidae). Prentice Hall New Jersey 277–297
S.R. Bordenstein M.D. Drapeau J.H. Werren (2000) ArticleTitleIntraspecific variation in sexual isolation in the jewel wasp Nasonia Evolution 54 567–573 Occurrence Handle1:STN:280:DC%2BD3cvgtlKruw%3D%3D Occurrence Handle10937233
W.H. Brown (1953) ArticleTitleIntroduced fish species of the Guadalupe River Basin Texas J. Sci. 5 245–251
W.L. Brown E.O. Wilson (1956) ArticleTitleCharacter displacement Syst. Zool. 5 49–64
J.A. Coyne H.A. Orr (1989) ArticleTitlePatterns of speciation in Drosophila Evolution 43 362–381
J.A. Coyne H.A. Orr (1997) ArticleTitlePatterns of speciation in Drosophilarevisited Evolution 51 295–303
R.M. Darnell P. Abramoff (1968) ArticleTitleDistribution of the gynogenetic fish, Poeciliaformosa, with remarks on the evolution of the species Copeia 1968 354–361
J. Felsenstein (1995) PHYLIP: Phylogenetic Inference Package, Version 3.572 University of Washington Seattle, Washington, USA
M. Ferreira J.W.H. Ferguson (2002) ArticleTitleGeographic variation in the calling song of the field cricket Gryllusbimaculatus(Orthoptera: Gryllidae) and its relevance to mate recognition and mate choice J. Zool. 257 163–170
C.R. Gabor M.J. Ryan (2001) ArticleTitleGeographical variation in reproductive character displacement in mate choice by male sailfin mollies Proc. Roy. Soc. Ser. B. Biol. Sci. 268 1063–1070 Occurrence Handle1:STN:280:DC%2BD3M3pt1OrtA%3D%3D
H.C. Gerhardt (1994) ArticleTitleReproductive character displacement of female mate choice in the grey treefrog, Hyla chrysoscelis Anim. Behav. 47 959–969
H.C. Gerhardt (1999) Reproductive character displacement and other sources of selection on acoustic communication systems M.D. Hauser M. Konishi (Eds) The Design of Animal Communication. Massachusetts Institute of Technology Press Boston, MA 515–534
H. Harris D.A. Hopkinson (1978) Handbook of Enzyme Electrophoresis in Human Genetics North-Holland Publishing Company Amsterdam, Holland
G. Höbel H.C. Gerhardt (2003) ArticleTitleReproductive character displacement in the acoustic communication system of green tree frogs (Hyla cinerea) Evolution 57 894–904 Occurrence Handle12778558
D.J. Howard (1993) Reinforcement: origin, dynamics and fate of an evolutionary hypothesis R.G. Harrison (Eds) Hybrid Zones and the Evolutionary Process. Oxford University Press New York 46–69
C. Hubbs L.C. Hubbs (1932) ArticleTitleApparent parthenogenesis in nature in a form of fish of hybrid origin Science 76 628–630
J.P. Kruskal M. Wish (1978) Multidimensional Scaling Sage Publications Newbury Park, California, USA
R. Lande (1982) ArticleTitleRapid origin of sexual isolation and character divergence in a cline. Evolution 36 213–223
P. Legendre A. Vaudor (1991) The R-package: Multidimensional Analysis, Spatial Analysis University of Montreal Québec, Canada
E.P. Lessa (1990) ArticleTitleMultidimensional analysis of geographic genetic structure Syst. Zool. 39 242–252
M.J. Littlejohn J.D. Robertson (1975) ArticleTitleAcoustic analysis of an intergrade zone between two call races of the Limnodynastes tasmaniensiscomplex (Anura: Leptocactylidae) in southeastern Australia Aust. J. Zool. 23 113–122
R. Márquez J. Bosch (1997) ArticleTitleMale advertisement call and female preference in sympatric and allopatric midwife toads Anim. Behav. 54 1333–1345 Occurrence Handle9521791
D.C. Marshall J.R. Cooley (2000) ArticleTitleReproductive character displacement and speciation in periodical cicadas, with description of a new species, a 13-year Magicicada neotredecima Evolution 54 1333–1345
M. Mateos R.C. Vrijenhoek (2002) ArticleTitleAncient versus reticulate origin of a hemiclonal lineage Evolution 56 985–992 Occurrence Handle12093033
T.C. Mendelson (2003) ArticleTitleSexual isolation evolves faster than hybrid inviability in a diverse and sexually dimorphic genus of fish (Percidae: Etheostoma) Evolution 57 317–327 Occurrence Handle12683528
Milstead, E. (1980). Genetic differentiation among subpopulations of three Gambusiaspecies (Pisces: Poeciliidae) in the Pecos River, Texas and New Mexico. M.S. thesis, Baylor University, Waco, Texas.
D.C. Morizot M.E. Schmidt (1990) Starch gel electrophoresis and histochemical visualization of proteins D.H. Whitmore (Eds) Electrophoretic and Isoelectric Focusing Techniques in Fisheries Management. CRC Press Boca Raton, FL 23–80
M. Nei (1978) ArticleTitleEstimation of average heterozygosity and genetic distance from a small number of individuals Proc. Nat. Acad. Sci. USA 70 3321–3323
J.E. Neigel J.C. Avise (1986) Phylogenetic relationships of mitochondrial DNA under various demographic models of speciation E. Nevo S. Karlin (Eds) Evolutionary Processes and Theory. Academic Press New York 515–534
Nevo, E., A. Beiles & R. Ben-Shlomo (1984) The evolutionary significance of genetic diversity: ecological, demographic and life history correlates 53: 13–213. In: S. Levin (ed.), Lecture Notes in Biomathematics: Evolutionary Dynamics of Genetic Diversity, Springer-Verlag, Berlin.
M.A. Noor (1995) ArticleTitleSpeciation driven by natural selection in Drosophila Nature 375 674–675 Occurrence Handle1:CAS:528:DyaK2MXmsFKrt74%3D Occurrence Handle7791899
K.S. Pfennig (2000) ArticleTitleFemale spadefoot toads compromise on mate quality to ensure conspecific matings Behav. Ecol. 11 220–227
J.M. Quattro J.C. Avise R.C. Vrijenhoek (1992) ArticleTitleAn ancient clonal lineage in the fish genus Poeciliopsis(Atheriniformes: Poeciliidae) Proc. Nat. Acad. Sci. USA 89 348–352 Occurrence Handle1:STN:280:DC%2BD3MrmtFahtQ%3D%3D Occurrence Handle11607248
M.G. Ritchie R.K. Butlin G.M. Hewitt (1992) ArticleTitleFitness consequences of potential assortative mating inside and outside a hybrid zone in Chorthippus parallelus(Orthoptera: Acrididae): Implications for reinforcement and sexual selection theory Biol. J. Linn. Soc. 45 219–234
J.S. Rogersm (1972) ArticleTitleMeasures of genetic similarity and genetic distance Studies in Genetics, University of Texas Publishers 7213 145–153
H.D. Rundle D. Schluter (1998) ArticleTitleReinforcement of stickleback mate preferences: sympatry breeds contempt Evolution 52 200–208
M.J. Ryan W. Wilczynski (1991) ArticleTitleEvolution of intraspecific variation in the advertisement call of a cricket frog (Acris crepitans, Hylidae) Biol. J. Linn. Soc. 44 249–271
M.J. Ryan L.A. Dries P. Batra D.M. Hillis (1996) ArticleTitleMale mate preferences in a gynogenetic species complex of Amazon mollies Anim. Behav. 52 1225–1236
M.J. Ryan A.S. Rand L.A. Weigt (1996b) ArticleTitleAllozyme and advertisement call variation in the Túngara frog, Physalaemus pustulosus Evolution 50 2435–2453 Occurrence Handle1:CAS:528:DyaK2sXnvFamtg%3D%3D
G.-P. Sætre T. Moum S. Bures M. Král M. Adamjan J. Moreno (1997) ArticleTitleA sexually selected character displacement in flycatchers reinforces premating isolation Nature 387 589–592
N. Sanderson J.M. Szymura N.H. Barton (1992) ArticleTitleVariation in mating call across the hybrid zone between the fire-bellied toads Bombinabombinaand B. variegata Evolution 46 595–607
M. Schartl B. Wilde I. Schlupp J. Parzefall (1995) ArticleTitleEvolutionary origin of a parthenoform, the Amazon molly, Poecilia formosa, on the basis of a molecular genealogy Evolution 49 827–835 Occurrence Handle1:CAS:528:DyaK28XktlyisA%3D%3D
I. Schlupp J. Parzefall M. Schartl (2002) ArticleTitleBiogeography of the Amazon molly, Poecilia formosa J. Biogeogr. 29 1–6
S. Schneider D. Roessli L. Excoffier (2000) Arlequin version 2.00: A software for populations genetics data analysis Genetics and Biometry Laboratory, University of Geneva Switzerland
R.K. Selander M.H. Smith S.Y. Yang W.E. Johnson J.B. Gentry (1971) ArticleTitleBiochemical polymorphism and systematics in the genus Peromyscus. I. Variation in the old-field mouse (Peromyscus polionotus) Studies in Genetics VI. University of Texas Publications 7103 49–90
D.E. Simanek (1978a) ArticleTitleGenetic variability and population structure of Poecilia latipinna Nature 276 612–614
Simanek, D.E. 1978b. Population genetics and evolution in the Poecilia formosacomplex (Pisces: Poeciliidae). Ph.D. dissertation, Yale University, New Haven.
M. Slatkin (1987) ArticleTitleGene flow and the geographic structure of natural populations Science 236 787–792 Occurrence Handle1:STN:280:BiiB3cnhvV0%3D Occurrence Handle3576198
P.E. Smouse J.C. Long R.R. Sokal (1986) ArticleTitleMultiple regression and correlation extensions of the Mantel test of matrix correspondence Syst. Zool. 35 627–632
P.H.A. Sneath R.R. Sokal (1973) Numerical Taxonomy Freeman W.B. San Francisco, California
D.L. Swofford R.B. Selander (1981) ArticleTitleBIOSYS-1: A FORTRAN computer program for the comprehensive analysis of electrophoretic data in population genetics and systematics J. Hered. 72 281–283
S.G. Tilley P.A. Verrell S.J. Arnold (1990) ArticleTitleCorrespondence between sexual isolation and allozyme differentiation: a test in the salamander Desmognathus ochrophaeus Proc. Nat. Acad. Sci. USA 87 2715–2719 Occurrence Handle1:STN:280:By%2BB3cjis1M%3D Occurrence Handle2320584
J.C. Trexler (1988) ArticleTitleHierarchical organization of genetic variation in the sailfin molly, Poecilialatipinna(Pisces: Poeciliidae) Evolution 42 1006–1017
B.J. Turner B.H. Brett E.M. Rasch J.S. Balsano (1980) ArticleTitleEvolutionary Genetics of a Gynogenetic Fish, Poecilia formosa, the Amazon Molly Evolution 34 246–258
J.K. Waage (1979) ArticleTitleReproductive character displacement in Calopteryx(Odonata: Calopterygidae) Evolution 33 104–116
B.S. Weir (1996) Genetic Data Analysis II Sinauer and Associates Sunderland, Massachusetts
B.S. Weir C.C. Cockerham (1984) ArticleTitleEstimating F-statistics for the analysis of population structure Evolution 38 1358–1370
M.C. Whitlock (1999) ArticleTitleNeutral additive genetic variance in a metapopulation Genet. Res. 74 215–221 Occurrence Handle1:STN:280:DC%2BD3c7lsVeruw%3D%3D Occurrence Handle10689799
R.J. Wootten K.T. Scribner M.H. Smith (1988) ArticleTitleGenetic variability and systematics of Gambusiain the southeastern United States Copeia 1988 283–289
Acknowledgements
We thank A. Aspbury, L. Dries, L. Higgins, C. Nice, and J. Ott for helpful discussion concerning this research. L. Dries, L. Higgins, J. Krejca, C. Papp, and I. Schlupp are thanked for helping to collect fishes. We also thank F. Aboudi and J. Stahl for preparing fishes for allozyme analyses and J. Cruchfield at Brackenridge Field Laboratory for use of their facilities for maintaining fishes. Research was fully supported by funds from National Science Foundation Post-doctoral Research Fellowship grant DBI-9750278 to C.R.G. C.R.G. was also supported by Texas Higher Education Coordinating Board Advanced Research Program grant # 003615-0004-2001 during manuscript preparation. We also thank the Mexican Government for permission to collect these fishes.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gabor, C.R., Ryan, M.J. & Morizot, D.C. Character displacement in sailfin mollies, Poecilia latipinna: allozymes and behavior. Environ Biol Fish 73, 75–88 (2005). https://doi.org/10.1007/s10641-004-5354-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10641-004-5354-3