Summary
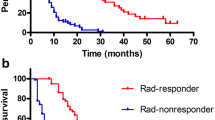
Alpha-fetoprotein (AFP) response (relative decline in AFP) is associated with imaging response evaluated by response evaluation criteria in solid tumors ver1.1 (RECIST) and survival in treatment for hepatocellular carcinoma (HCC). However, the optimal threshold of AFP response is still unknown, especially in atezolizumab and bevacizumab (Atez/Bev) treatment. In this prospective multicenter study, we aimed to investigate an optimal threshold of AFP response in Atez/Bev treatment. Out of 284 patients with unresectable HCC who were treated with Atez/Bev, 91 patients with AFP ≥ 10 ng/ml were enrolled in the multicenter study. We investigated the relationship between various AFP response thresholds (relative decline ≥ 20%, ≥ 50%, and ≥ 75%) and treatment response and progression-free survival (PFS). An AFP relative decrease of ≥ 50% was associated with an overall response rate (ORR) with an odds ratio (95% confidence interval [CI]) of 5.7 (1.9–17). Disease control rate (DCR) was associated with an AFP relative decrease of ≥ 20%, with a 100% positive predictive value and a 52.0% sensitivity. AFP relative decreases of ≥ 50% and ≥ 20% were significantly associated with PFS with a hazard ratio (HR) of 5.60 (95% CI: 1.6–19, p = 0.006) and a HR of 4.44 (95% CI: 1.9–10, p < 0.001), respectively. AFP response of ≥ 50% and ≥ 20% were related to ORR and DCR, respectively, and both of these responses were also associated with PFS. AFP can be used as a real-time monitor during Atez/Bev treatment and is helpful for treatment optimization.


Similar content being viewed by others
References
Higuchi M, Tamaki N, Kurosaki M et al (2022) Longitudinal association of magnetic resonance elastography-associated liver stiffness with complications and mortality. Aliment Pharmacol Ther 55:292–301
Tamaki N, Kurosaki M, Huang DQ et al (2022) Noninvasive assessment of liver fibrosis and its clinical significance in nonalcoholic fatty liver disease. Hepatol Res 52:497–507
Huang DQ, Singal AG, Kono Y et al (2022) Changing global epidemiology of liver cancer from 2010 to 2019: NASH is the fastest growing cause of liver cancer. Cell Metab 34:969–77.e2
Marrero JA, Kulik LM, Sirlin CB et al (2018) Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 68:723–750
Bruix J, Chan SL, Galle PR et al (2021) Systemic treatment of hepatocellular carcinoma: An EASL position paper. J Hepatol 75:960–974
Kokudo N, Takemura N, Hasegawa K et al (2019) Clinical practice guidelines for hepatocellular carcinoma: The Japan Society of Hepatology 2017 (4th JSH-HCC guidelines) 2019 update. Hepatol Res 49:1109–1113
Kuzuya T, Ishigami M, Ito T et al (2020) Sorafenib vs. Lenvatinib as First-line Therapy for Advanced Hepatocellular Carcinoma With Portal Vein Tumor Thrombosis. Anticancer Res 40:2283–90
Kuzuya T, Ishigami M, Ito T et al (2020) Favorable radiological antitumor response at 2 weeks after starting lenvatinib for patients with advanced hepatocellular carcinoma. Hepatol Res 50:374–381
Rimini M, Shimose S, Lonardi S et al (2021) Lenvatinib versus Sorafenib as first-line treatment in hepatocellular carcinoma: A multi-institutional matched case-control study. Hepatol Res 51:1229–1241
Hiraoka A, Kumada T, Tada T et al (2021) Efficacy of lenvatinib for unresectable hepatocellular carcinoma based on background liver disease etiology: multi-center retrospective study. Sci Rep 11:16663
Sekiguchi S, Tsuchiya K, Yasui Y et al (2022) Clinical usefulness of geriatric assessment in elderly patients with unresectable hepatocellular carcinoma receiving sorafenib or lenvatinib therapy. Cancer Rep (Hoboken) e1613
Yasui Y, Kurosaki M, Tsuchiya K et al (2022) Real-World Data on Ramucirumab Therapy including Patients Who Experienced Two or More Systemic Treatments: A Multicenter Study. Cancers (Basel) 14
Kudo M, Finn RS, Qin S et al (2018) Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet 391:1163–1173
Ochi H, Tani J, Tomonari T et al (2021) Sequential therapy including regorafenib for unresectable hepatocellular carcinoma: Effect of early relative changes in hepatic functional reserve after regorafenib administration on prognosis. Hepatol Res 51:1219–1228
Kim S, Kim KH, Kim BK et al (2021) Lenvatinib is independently associated with the reduced risk of progressive disease when compared with sorafenib in patients with advanced hepatocellular carcinoma. J Gastroenterol Hepatol 36:1317–1325
Tsuchiya K, Kurosaki M, Sakamoto A et al (2021) The Real-World Data in Japanese Patients with Unresectable Hepatocellular Carcinoma Treated with Lenvatinib from a Nationwide Multicenter Study. Cancers (Basel) 13
Kudo M, Tsuchiya K, Kato N et al (2021) Cabozantinib in Japanese patients with advanced hepatocellular carcinoma: a phase 2 multicenter study. J Gastroenterol 56:181–190
Terashima T, Yamashita T, Takata N et al (2021) Safety and efficacy of sorafenib followed by regorafenib or lenvatinib in patients with hepatocellular carcinoma. Hepatol Res 51:190–200
Finn RS, Qin S, Ikeda M et al (2020) Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med 382:1894–1905
Tada T, Kumada T, Hiraoka A et al (2022) Neutrophil-lymphocyte ratio predicts early outcomes in patients with unresectable hepatocellular carcinoma treated with atezolizumab plus bevacizumab: a multicenter analysis. Eur J Gastroenterol Hepatol 34:698–706
Chuma M, Uojima H, Hattori N et al (2022) Safety and efficacy of atezolizumab plus bevacizumab in patients with unresectable hepatocellular carcinoma in early clinical practice: A multicenter analysis. Hepatol Res 52:269–280
Maesaka K, Sakamori R, Yamada R et al (2022) Hyperprogressive disease in patients with unresectable hepatocellular carcinoma receiving atezolizumab plus bevacizumab therapy. Hepatol Res 52:298–307
Sho T, Suda G, Ogawa K et al (2021) Early response and safety of atezolizumab plus bevacizumab for unresectable hepatocellular carcinoma in patients who do not meet IMbrave150 eligibility criteria. Hepatol Res 51:979–989
Hayakawa Y, Tsuchiya K, Kurosaki M et al (2022) Early experience of atezolizumab plus bevacizumab therapy in Japanese patients with unresectable hepatocellular carcinoma in real-world practice. Invest New Drugs 40:392–402
Ochi H, Kurosaki M, Joko K et al (2022) Usefulness of neutrophil-to-lymphocyte ratio in predicting progression and survival outcomes after atezolizumab-bevacizumab treatment for hepatocellular carcinoma. Hepatol Res
Kuzuya T, Kawabe N, Hashimoto S et al (2022) Early Changes in Alpha-Fetoprotein Are a Useful Predictor of Efficacy of Atezolizumab plus Bevacizumab Treatment in Patients with Advanced Hepatocellular Carcinoma. Oncology 100:12–21
He C, Peng W, Liu X et al (2019) Post-treatment alpha-fetoprotein response predicts prognosis of patients with hepatocellular carcinoma: A meta-analysis. Medicine (Baltimore) 98:e16557
Zhu AX, Dayyani F, Yen CJ et al (2022) Alpha-fetoprotein as a potential surrogate biomarker for atezolizumab + bevacizumab treatment of hepatocellular carcinoma. Clin Cancer Res
Ichikawa T, Machida N, Sasaki H et al (2016) Early Prediction of the Outcome Using Tumor Markers and mRECIST in Unresectable Hepatocellular Carcinoma Patients Who Underwent Transarterial Chemoembolization. Oncology 91:317–330
Rungsakulkij N, Suragul W, Mingphruedhi S et al (2018) Prognostic role of alpha-fetoprotein response after hepatocellular carcinoma resection. World J Clin Cases 6:110–120
Yu SJ, Kwon JH, Kim W et al (2018) Initial Alpha-Fetoprotein Response Predicts Prognosis in Hepatitis B-related Solitary HCC Patients After Radiofrequency Ablation. J Clin Gastroenterol 52:e18–e26
Kim BK, Ahn SH, Seong JS et al (2011) Early α-fetoprotein response as a predictor for clinical outcome after localized concurrent chemoradiotherapy for advanced hepatocellular carcinoma. Liver Int 31:369–376
Lee S, Kim BK, Kim SU et al (2015) Early α-fetoprotein response predicts survival in patients with advanced hepatocellular carcinoma treated with sorafenib. J Hepatocell Carcinoma 2:39–47
Personeni N, Bozzarelli S, Pressiani T et al (2012) Usefulness of alpha-fetoprotein response in patients treated with sorafenib for advanced hepatocellular carcinoma. J Hepatol 57:101–107
Liu B, Shang X, Shi JY et al (2022) Early Alpha-Fetoprotein Response Is Associated With Survival in Patients With HBV-Related Hepatocellular Carcinoma Receiving Lenvatinib. Front Oncol 12:807189
Kaneko S, Tsuchiya K, Yasui Y et al (2022) Conversion surgery for hepatocellular carcinoma after tyrosine kinase inhibitor treatment. JGH Open 6:301–308
Kuzuya T, Kawabe N, Hashimoto S et al (2022) Clinical Outcomes of Ramucirumab as Post-treatment Following Atezolizumab/Bevacizumab Combination Therapy in Advanced Hepatocellular Carcinoma. Anticancer Res 42:1905–1910
Kudo M, Kawamura Y, Hasegawa K et al (2021) Management of Hepatocellular Carcinoma in Japan: JSH Consensus Statements and Recommendations 2021 Update. Liver Cancer 10:181–223
Reig M, Forner A, Rimola J et al (2022) BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J Hepatol 76:681–693
Funding
This work was supported by a Grant-in-Aid from the Japan Agency For Medical Research and Development (JP20fk0210067h0001).
Author information
Authors and Affiliations
Contributions
Data acquisition, Nobuharu Tamaki, Toshifumi Tada, Masayuki Kurosaki, Yutaka Yasui, Hironori Ochi, Toshie Mashiba, Azusa Sakamoto, Hiroyuki Marusawa, Ryoichi Narita, Yasushi Uchida, Takehiro Akahane, Masahiko Kondo, Nami Mori, Shintaro Takaki, Keiji Tsuji, Haruhiko Kobashi, Atsunori Kusakabe, Koichiro Furuta, Hirotaka Arai, Michiko Nonogi, Shinichiro Nakamura, Chitomi Hasebe, Kaoru Tsuchiya, Namiki Izumi; data analysis, Nobuharu Tamaki, Toshifumi Tada, Masayuki Kurosaki; data interpretation; Nobuharu Tamaki, Toshifumi Tada, Masayuki Kurosaki; drafting of the manuscript, Nobuharu Tamaki; critical revision of the manuscript, all authors; all authors reviewed the manuscript and approved the final submission.
Corresponding author
Ethics declarations
Conflicts of interest
Masayuki Kurosaki and Kaoru Tsuchiya received lecture fees from Chugai. The other authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tamaki, N., Tada, T., Kurosaki, M. et al. Optimal threshold of alpha-fetoprotein response in patients with unresectable hepatocellular carcinoma treated with atezolizumab and bevacizumab. Invest New Drugs 40, 1290–1297 (2022). https://doi.org/10.1007/s10637-022-01303-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-022-01303-w