Summary
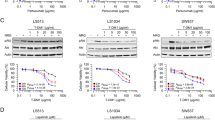
The aim of this study was to investigate the effect of lapatinib, a selective inhibitor of EGFR/HER2 tyrosine kinases, on pancreatic cancer cell lines both alone and in combination with chemotherapy. Two cell lines, BxPc-3 and HPAC, displayed the greatest sensitivity to lapatinib (IC50 < 2 μM). Lapatinib also demonstrated some activity in three K-Ras mutated pancreatic cancer cell lines which displayed resistance to erlotinib. Drug effect/combination index (CI) isobologram analysis was used to study the interactions of lapatinib with gemcitabine, cisplatin and 5’deoxy-5’fluorouridine. Concentration-dependent anti-proliferative effects of lapatinib in combination with chemotherapy were observed. To evaluate the potential effect of lapatinib in pancreatic cancer tumours, and to identify a subset of patient most likely to benefit from lapatinib, expression of EGFR and HER2 were investigated in 72 pancreatic cancer tumour specimens by immunohistochemistry. HER2 membrane expression was observed in only 1 % of cases, whereas 44 % of pancreatic tumours expressed EGFR. Based on our in vitro results, lapatinib may provide clinical benefit in EGFR positive pancreatic ductal adenocarcinoma.




Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ (2007) Cancer statistics, 2007. CA Cancer J Clin 57:43–66. doi:10.3322/canjclin.57.1.43
Procacci C, Biasiutti C, Carbognin G, Capelli P, El-Dalati G, Flaconi M et al (2001) Pancreatic neoplasms and tumor-like conditions. Eur Radiol 11(suppl. 2.):S167–92
Burris HA 3rd, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC et al (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15:2403–2413
Berlin JD, Catalano P, Thomas JP, Kugler JW, Haller DG, Benson AB 3rd (2002) Phase III study of gemcitabine in combination with fluorouracil versus gemcitabine alone in patients with advanced pancreatic carcinoma: Eastern Cooperative Oncology Group Trial E2297. J Clin Oncol 20:3270–3276. doi:10.1200/JCO.2002.11.149
Heinemann V, Quietzsch D, Gieseler F, Gonnermann M, Schonekas H, Rost A, Neuhaus H et al (2006) Randomized phase III trial of gemcitabine plus cisplatin compared with gemcitabine alone in advanced pancreatic cancer. J Clin Oncol 20:3948–3952. doi:10.1200/JCO.2005.05.1490
Louvet C, Labianca R, Hammel P, Lledo G, Zampino MG, Andre T, Zaniboni A, Ducreux M et al (2005) Gemcitabine in combination with oxaliplatin compared with gemcitabine alone in locally advanced or metastatic pancreatic cancer: results of a GERCOR and GISCAD phase III trial. J Clin Oncol 23:3509–3518. doi:10.1200/JCO.2005.06.023
Rocha Lima CM, Green MR, Rotche R, Miller WH Jr, Jeffrey GM, Cisar LA, Morganti A, Orlando N, Gruia G, Miller LL (2004) Irinotecan plus gemcitabine results in no survival advantage compared with gemcitabine monotherapy in patients with locally advanced or metastatic pancreatic cancer despite increased tumor response rate. J Clin Oncol 22:3778–3783. doi:10.1200/JCO.2004.12.082
Van Cutsem E, Findlay M, Osterwalder B, Kocha W, Dalley D, Pazdur R, Cassidy J, Dirix L, Twelves C, Allman D, Seitz JF, Scholmerich J, Burger HU, Verweij J (2000) Capecitabine, an oral fluoropyrimidine carbamate with substantial activity in advanced colorectal cancer: results of a randomized phase II study. J Clin Oncol 18:1337–1345
Cunningham D, Chau I, Stocken D, Davies C, Dunn J, Valle J, Smith D, Steward W, Harper P, Neoptolemos J (2009) Phase III randomised comparison of gemcitabine (GEM) versus gemcitabine plus capecitabine (GEM-CAP) in patients with advanced pancreatic cancer. J Clin Oncol 27:5513–5518. doi:10.1200/JCO.2009.24.2446
Herrmann R, Bodoky G, Ruhstaller T, Glimelius B, Bajetta E, Schuller J, Saletti P, Swiss Group for Clinical Cancer Research, Central European Cooperative Oncology Group et al (2007) Gemcitabine plus capecitabine compared with gemcitabine alone in advanced pancreatic cancer: a randomized, multicenter, phase III trial of the Swiss Group for Clinical Cancer Research and the Central European Cooperative Oncology Group. J Clin Oncol 25:2212–2217. doi:10.1200/JCO.2006.09.0886
Tobita K, Kijima H, Dowaki S, Kashiwagi H, Ohtani Y, Oida Y, Yamazaki H, Nakamura M, Ueyama Y, Tanaka M, Inokuchi S, Makuuchi H (2003) Epidermal growth factor receptor expression in human pancreatic cancer: significance for liver metastasis. Int J Mol Med 11:305–309
Bloomston M, Bhardwaj A, Ellison EC, Frankel WL (2006) Epidermal growth factor receptor expression in pancreatic carcinoma using tissue microarray technique. Dig Surg 23:74–79. doi:10.1159/000093497
Komoto M, Nakata B, Nishii T, Kawajiri H, Shinto O, Amano R, Yamada N, Yashiro M, Hirakawa K (2010) In vitro and in vivo evidence that a combination of lapatinib plus S-1 is a promising treatment for pancreatic cancer. Cancer Science 101:468–473. doi:10.1111/j.1349-7006.2009.01405.x
Garcea G, Neal CP, Pattenden CJ, Steward WP, Berry DP (2005) Molecular prognostic markers in pancreatic cancer: a systematic review. Eur J Cancer 41:2213–2236
Bruns CJ, Harbison MT, Davis DW, Portera CA, Tsan R, McConkey DJ, Evans DB, Abbruzzese JL, Hicklin DJ, Radinsky R (2000) Epidermal growth factor receptor blockade with C225 plus gemcitabine results in regression of human pancreatic carcinoma growing orthotopically in nude mice by antiangiogenic mechanisms. Clin Cancer Res 6:1936–1948
Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, Au HJ, Murawa P, Walde D, Wolff RA, Campos D, Lim R, Ding K, Clark G, Voskoglou-Nomikos T, Ptasynski M, Parulekar W, National Cancer Institute of Canada Clinical Trials Group (2007) Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 25:1960–1966. doi:10.1200/JCO.2006.07.9525
Safran H, Steinhoff M, Mangray S, Rathore R, King TC, Chai L, Berzein K, Moore T, Iannitti D, Reiss P, Pasquariello T, Akerman P, Quirk D, Mass R, Goldstein L, Tantravahi U (2001) Overexpression of the HER-2/neu oncogene in pancreatic adenocarcinoma. Am J Clin Oncol 24:496–499
Yamanaka Y, Friess H, Kobrin MS, Buchler M, Kunz J, Beger HG, Korc M (1993) Overexpression of HER2/neu oncogene in human pancreatic carcinoma. Hum Pathol 24:1127–1134
Day JD, Digiuseppe JA, Yeo C, Lai-Goldman M, Anderson SM, Goodman SN, Kern SE, Hruban RH (1996) Immunohistochemical evaluation of HER-2/neu expression in pancreatic adenocarcinoma and pancreatic intraepithelial neoplasms. Hum Pathol 27:119–124
Chou TC, Talalay P (1984) Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 22:27–55
Burris HA 3rd, Hurwitz HI, Dees EC, Dowlati A, Blackwell KL, O'Neil B, Marcom PK, Ellis MJ, Overmoyer B et al (2005) Phase I safety, pharmacokinetics, and clinical activity study of lapatinib (GW572016), a reversible dual inhibitor of epidermal growth factor receptor tyrosine kinases, in heavily pretreated patients with metastatic carcinomas. J Clin Oncol 23:5305–5313. doi:10.1200/JCO.2005.16.584
Konecny GE, Venkatesan N, Yang G, Dering J, Ginther C, Finn R, Rahmeh M, Fejzo MS, Toft D, Jiang SW, Slamon DJ, Podratz KC (2008) Activity of lapatinib a novel HER2 and EGFR dual kinase inhibitor in human endometrial cancer cells. Br J Cancer 98:1076–1084. doi:10.1038/sj.bjc.6604278
Konecny GE, Pegram MD, Venkatesan N, Finn R, Yang G, Rahmeh M, Untch M, Rusnak DW, Spehar G, Mullin RJ, Keith BR, Gilmer TM, Berger M, Podratz KC, Slamon DJ (2006) Activity of the dual kinase inhibitor lapatinib (GW572016) against HER-2-overexpressing and trastuzumab-treated breast cancer cells. Cancer Res 66:1630–1639. doi:10.1158/0008-5472.CAN-05-1182
Zhang D, Pal A, Bornmann WG, Yamasaki F, Esteva FJ, Hortobagyi GN, Bartholomeusz C, Ueno NT (2008) Activity of lapatinib is independent of EGFR expression level in HER2-overexpressing breast cancer cells. Mol Cancer Ther 7:1864–1850. doi:10.1158/1535-7163.MCT-08-0168
Yamasaki F, Zhang D, Bartholomeusz C, Sudo T, Hortobagyi GN, Kurisu K, Ueno NT (2007) Sensitivity of breast cancer cells to erlotinib depends on cyclin-dependent kinase 2 activity. Mol Cancer Ther 6:2168–2177. doi:10.1158/1535-7163.MCT-06-0514
Buck E, Eyzaguirre A, Haley JD, Gibson NW, Cagnoni P, Iwata KK (2006) Inactivation of Akt by the epidermal growth factor receptor inhibitor erlotinib is mediated by HER-3 in pancreatic and colorectal tumor cell lines and contributes to erlotinib sensitivity. Mol Cancer Ther 5:2051–2059. doi:10.1158/1535-7163.MCT-06-0007
Talar-Wojnarowska R, Gasiorowska A, Smolarz B, Romanowicz-Makowska H, Strzelczyk J, Janiak A, Kulig A, Malecka-Panas E (2004) Usefulness of p16 and K-ras mutation in pancreatic adenocarcinoma and chronic pancreatitis differential diagnosis. J Physiol Pharmacol 55:S129–38
Smit VT, Boot AJ, Smits AM, Fleuren GJ, Cornelisse CJ, Bos JL (1988) KRAS codon 12 mutations occur very frequently in pancreatic adenocarcinomas. Nucleic Acids Res 16:7773–7782. doi:10.1093/nar/16.16.7773
Lee J, Jang KT, Ki CS, Lim T, Park YS, Lim HY, Choi DW, Kang WK, Park K, Park JO (2007) Impact of epidermal growth factor receptor (EGFR) kinase mutations, EGFR gene amplifications, and KRAS mutations on survival of pancreatic adenocarcinoma. Cancer 109:1561–1569. doi:10.1002/cncr.22559
Pao W, Wang TY, Riely GJ, Miller VA, Pan Q, Ladanyi M, Zakowski MF, Heelan RT, Kris MG, Varmus HE (2005) KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib. PLoS Med 2:e17. doi:10.1371/journal.pmed.0020017
Lievre A, Bachet JB, Le Corre D, Boige V, Landi B, Emile JF, Cote JF, Tomasic G, Penna C, Ducreux M, Rougier P, Penault-Llorca F, Laurent-Puig P (2006) KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 66:3992–3995. doi:10.1158/0008-5472.CAN-06-0191
Moore MJ, da Cunha SG, Kamel-Reid S, Chin K, Tu D, Parulekar W, Ludkovski O, Squire J, Richardson F, Tsao M (2007) The relationship of K-ras mutations and EGFR gene copy number to outcome in patients treated with Erlotinib on National Cancer Institute of Canada Clinical Trials Group trial study PA.3. J Clin Oncol 25:S4521
Wang Z, Kong D, Banerjee S, Li Y, Adsay NV, Abbruzzese J, Sarkar FH (2007) Down-regulation of platelet-derived growth factor-D inhibits cell growth and angiogenesis through inactivation of Notch-1 and nuclear factor-kappaB signaling. Cancer Res 37:11377–11385. doi:10.1158/0008-5472.CAN-07-2803
Safran H, Miner T, Bahary N, Whiting S, Lopez CD, Sun W, Charpentier K, Shipley J et al (2011) Lapatinib and gemcitabine for metastatic pancreatic cancer: a phase II study. Am J Clin Oncol 34:50–52. doi:10.1097/COC.0b013e3181d26b01
Wu Z, Marshall J, Hwang JJ, Pishvaian MJ, Wellstein A, Weiner LM, He AR (2012) A phase II study of lapatinib and capecitabine in second-line treatment of metastatic pancreatic cancer. J Clin Oncol 30:e14569
McDermott R, Calvert P, Parker M (2011) A phase II study of lapatinib and capecitabine in first-line treatment of metastatic pancreatic cancer (ICORG 08–39). J Clin Oncol 29:e315
Selvarajan S, Bay BH, Chng MJ, Tan PH (2004) The HercepTest and routine C-erbB2 immunohistochemistry in breast cancer: any difference? Ann Acad Med Singapore 33:473–476
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL (1987) Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235:177–181
Ueda S, Ogata S, Tsuda H, Kawarabayashi N, Kimura M, Sugiura Y, Tamai S, Matsubara O, Hatsuse K, Mochizuki H (2004) The correlation between cytoplasmic overexpression of epidermal growth factor receptor and tumor aggressiveness: poor prognosis in patients with pancreatic ductal adenocarcinoma. Pancreas 29:1–8
Half E, Broaddus R, Danenberg KD, Danenberg PV, Ayers GD, Sinicrope FA (2004) HER-2 receptor expression, localization, and activation in colorectal cancer cell lines and human tumors. Int J Cancer 108:540–548. doi:10.1002/ijc.11599
Lin SY, Makino K, Xia W, Matin A, Wen Y, Kwong KY, Bourguignon L, Hung MC (2001) Nuclear localization of EGF receptor and its potential new role as a transcription factor. Nat Cell Biol 3:802–808. doi:10.1038/ncb0901-802
Lo HW, Xia W, Wei Y, Ali-Seyed M, Huang SF, Hung MC (2005) Novel prognostic value of nuclear epidermal growth factor receptor in breast cancer. Cancer Res 65:338–348
Psyrri A, Yu Z, Weinberger PM, Sasaki C, Haffty B, Camp R, Rimm D, Burtness BA (2005) Quantitative determination of nuclear and cytoplasmic epidermal growth factor receptor expression in oropharyngeal squamous cell cancer by using automated quantitative analysis. Clin Cancer Res 11:5856–5862. doi:10.1158/1078-0432.CCR-05-0420
Acknowledgments
This work was supported by funding from Ireland’s Higher Educational Authority Programme for Research in Third Level Institutions (PRTLI) Cycle 3 and 4, and the Cancer Clinical Research Trust, Ireland.
Disclosure/Conflict of Interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Fig S1
HER2 (A) and EGFR (B) expression levels as measured by ELISA (pg/μl of total protein). (TIFF 60 kb)
Fig. S2
Fixed combination proliferation assays of pancreatic cancer cell lines, BxPc-3 and HPAC treated with lapatinib in combination with chemotherapeutic drugs gemcitabine and 5dFUrd. Fixed concentration ratio of lapatinib to gemcitabine (1:0.01) and lapatinib to 5’dFUrd (1:8). (TIFF 87 kb)
Rights and permissions
About this article
Cite this article
Walsh, N., Kennedy, S., Larkin, A. et al. EGFR and HER2 inhibition in pancreatic cancer. Invest New Drugs 31, 558–566 (2013). https://doi.org/10.1007/s10637-012-9891-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-012-9891-x