Abstract
Background
Hepatocellular carcinoma (HCC) is the third leading cause of cancer-related deaths worldwide. It is indispensable to understanding molecular mechanisms of HCC progression and to developing clinically useful biomarkers for this disease.
Aim
In this article, we examined whether HOXC8 was associated with the poor prognosis of hepatocellular carcinoma and explored the possible underlying mechanism.
Methods
The HOXC8 and Ki67 expression levels in 86 patients with hepatocellular carcinoma were examined using immunohistochemistry. HOXC8 levels in HCC cells were downregulated by siRNA transfection. The cycle progression and cell proliferation status of HCC cells and the oxaliplatin effectiveness were evaluated by flow cytometry and CCK-8 assay. HOXC8, CyclinD1, PCNA, Nkd2, and cleaved caspase-3 levels were detected by western blot.
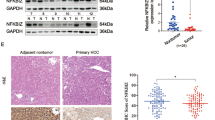
Results
HOXC8 was upregulated in HCC tissues, compared with adjacent non-tumor ones. HOXC8 expression levels in 86 patients with hepatocellular carcinoma were positively correlated with histological grade. Univariate and multivariate survival analysis revealed that HOXC8 was a significant predictor for overall survival of HCC patients. HOXC8 siRNA knockdown delayed the G1–S phase transition, inhibited cell proliferation, and attenuated resistance to oxaliplatin.
Conclusions
HOXC8 promoted HCC proliferation and predicted poor prognosis. Furthermore, upregulated HOXC8 expression was associated with oxaliplatin resistance in hepatocellular carcinoma.






Similar content being viewed by others
References
Morozov TK, Potekhina VA. Characteristics of performing a periodic medical examination in the transition to annual dispensary care for workers. Gigiena i sanitariia. 1986;2:28–30.
Schafer DF, Sorrell MF. Hepatocellular carcinoma. Lancet. 1999;353:1253–1257.
Cao H, Phan H, Yang LX. Improved chemotherapy for hepatocellular carcinoma. Anticancer Res. 2012;32:1379–1386.
Montero J, Morales A, Llacuna L, et al. Mitochondrial cholesterol contributes to chemotherapy resistance in hepatocellular carcinoma. Cancer Res. 2008;68:5246–5256.
Asghar U, Meyer T. Are there opportunities for chemotherapy in the treatment of hepatocellular cancer? J Hepatol. 2012;56:686–695.
Du H, Yang W, Chen L, et al. Role of autophagy in resistance to oxaliplatin in hepatocellular carcinoma cells. Oncol Rep. 2012;27:143–150.
Gehring WJ, Hiromi Y. Homeotic genes and the homeobox. Annu Rev Genet. 1986;20:147–173.
Pearson JC, Lemons D, McGinnis W. Modulating Hox gene functions during animal body patterning. Nat Rev Genet. 2005;6:893–904.
Mann RS, Morata G. The developmental and molecular biology of genes that subdivide the body of Drosophila. Annu Rev Cell Dev Biol. 2000;16:243–271.
McGinnis W, Krumlauf R. Homeobox genes and axial patterning. Cell. 1992;68:283–302.
Tiret L, Le Mouellic H, Maury M, Brulet P. Increased apoptosis of motoneurons and altered somatotopic maps in the brachial spinal cord of Hoxc-8-deficient mice. Development. 1998;125:279–291.
Kamel S, Kruger C, Salbaum JM, Kappen C. Morpholino-mediated knockdown in primary chondrocytes implicates Hoxc8 in regulation of cell cycle progression. Bone. 2009;44:708–716.
Yang X, Ji X, Shi X, Cao X. Smad1 domains interacting with Hoxc-8 induce osteoblast differentiation. J Biol Chem. 2000;275:1065–1072.
Min H, Lee JY, Bok J, Chung HJ, Kim MH. Proliferating cell nuclear antigen (Pcna) as a direct downstream target gene of Hoxc8. Biochem Biophys Res Commun. 2010;392:543–547.
Miller GJ, Miller HL, van Bokhoven A, et al. Aberrant HOXC expression accompanies the malignant phenotype in human prostate. Cancer Res. 2003;63:5879–5888.
Alami Y, Castronovo V, Belotti D, Flagiello D, Clausse N. HOXC5 and HOXC8 expression are selectively turned on in human cervical cancer cells compared to normal keratinocytes. Biochem Biophys Res Commun. 1999;257:738–745.
Waltregny D, Alami Y, Clausse N, de Leval J, Castronovo V. Overexpression of the homeobox gene HOXC8 in human prostate cancer correlates with loss of tumor differentiation. Prostate. 2002;50:162–169.
Axlund SD, Lambert JR, Nordeen SK. HOXC8 inhibits androgen receptor signaling in human prostate cancer cells by inhibiting SRC-3 recruitment to direct androgen target genes. Mol Cancer Res MCR. 2010;8:1643–1655.
Li Y, Guo Z, Chen H, et al. HOXC8-dependent cadherin 11 expression facilitates breast cancer cell migration through Trio and Rac. Genes Cancer. 2011;2:880–888.
Li Y, Zhang M, Chen H, et al. Ratio of miR-196s to HOXC8 messenger RNA correlates with breast cancer cell migration and metastasis. Cancer Res. 2010;70:7894–7904.
Wan C, Hou S, Ni R, et al. MIF4G domain containing protein regulates cell cycle and hepatic carcinogenesis by antagonizing CDK2-dependent p27 stability. Oncogene. 2015;34:237–245.
Lei H, Juan AH, Kim MS, Ruddle FH. Mouse naked cuticle 2 (mNkd2) as a direct transcriptional target of Hoxc8 in vivo. J Exp Zool Part A Ecol Genet Physiol. 2007;307:1–6.
Zhang S, Cagatay T, Amanai M, et al. Viable mice with compound mutations in the Wnt/Dvl pathway antagonists nkd1 and nkd2. Mol Cell Biol. 2007;27:4454–4464.
Rousset R, Mack JA, Wharton KA Jr, et al. Naked cuticle targets dishevelled to antagonize Wnt signal transduction. Genes Dev. 2001;15:658–671.
Yuzugullu H, Benhaj K, Ozturk N, et al. Canonical Wnt signaling is antagonized by noncanonical Wnt5a in hepatocellular carcinoma cells. Mol Cancer. 2009;8:90.
Katoh M. Molecular cloning, gene structure, and expression analyses of NKD1 and NKD2. Int J Oncol. 2001;19:963–969.
Prasad CP, Gupta SD, Rath G, Ralhan R. Wnt signaling pathway in invasive ductal carcinoma of the breast: relationship between beta-catenin, dishevelled and cyclin D1 expression. Oncology. 2007;73:112–117.
Ohhara Y, Suenaga M, Matsusaka S, Shinozaki E, Mizunuma N, Yamaguchi T. Comparison between three oxaliplatin-based regimens with bevacizumab in patients with metastatic colorectal cancer. OncoTargets Ther. 2015;8:529–537.
Wu P, Zhu X, Jin W, Hao S, Liu Q, Zhang L. Oxaliplatin triggers necrosis as well as apoptosis in gastric cancer SGC-7901 cells. Biochem Biophys Res Commun. 2015;460:183–190.
Wang XH, Meng XW, Sun X, Du YJ, Zhao J, Fan YJ. Wnt/b-catenin signaling pathway affects the protein expressions of caspase-3, XIAP and Grp-78 in hepatocellular carcinoma. Zhonghua gan zang bing za zhi= Zhonghua ganzangbing zazhi= Chinese Journal of Hepatology. 2011;19:599–602.
Pang RW, Poon RT. From molecular biology to targeted therapies for hepatocellular carcinoma: the future is now. Oncology. 2007;72:30–44.
Lee TK, Man K, Ho JW, et al. Significance of the Rac signaling pathway in HCC cell motility: implications for a new therapeutic target. Carcinogenesis. 2005;26:681–687.
Ma WL, Hsu CL, Wu MH et al. Androgen receptor is a new potential therapeutic target for the treatment of hepatocellular carcinoma. Gastroenterology. 2008;135:947–955, 955 e941–945.
Calvo R, West J, Franklin W, et al. Altered HOX and WNT7A expression in human lung cancer. Proc Natl Acad Sci USA. 2000;97:12776–12781.
Ferrell CM, Dorsam ST, Ohta H, et al. Activation of stem-cell specific genes by HOXA9 and HOXA10 homeodomain proteins in CD34+ human cord blood cells. Stem Cells. 2005;23:644–655.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Nos. 81472272, 81472708).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Pan Xu and Xiubing Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xu, P., Zhang, X., Ni, W. et al. Upregulated HOXC8 Expression Is Associated with Poor Prognosis and Oxaliplatin Resistance in Hepatocellular Carcinoma. Dig Dis Sci 60, 3351–3363 (2015). https://doi.org/10.1007/s10620-015-3774-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3774-x