Abstract
Background
Gastroparesis affects predominantly females; however, the biological basis for this gender bias is completely unknown. Several lines of evidence suggest that nitrergic dependent stomach motility function is reduced in diabetic gastroparesis and that nNOS is estrogen-regulated.
Aims
The purpose of this study was to investigate whether reduced levels of estradiol-17β (E2) down-regulates tetrahydrobiopterin (BH4, a cofactor for nNOS dimerization and enzyme activity) biosynthesis and therefore nNOS mediated gastric motility would be impaired in a mouse model of chronic estrogen deficiency, follicle stimulating hormone receptor knock-out female mice (FORKO).
Methods
In-bred 12-week-old female FORKO mice were obtained from our FORKO breeding colony. Gastric emptying was measured in overnight fasting mice. Nitrergic relaxation (AUC/mg tissue) was measured at 2 Hz through electric field stimulation using gastric antrum strips prepared from WT and FORKO mice. Protein expression for nNOSα, BH4 biosynthesis enzymes (GCH-1, DHFR) and estrogen receptors (α, β) were measured in gastric antrum by western blotting. Levels of BH4 and oxidized BH2, B biopterin levels were determined by HPLC.
Results
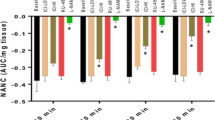
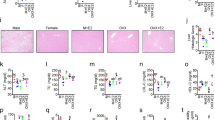
In FORKO, compared to wild type (WT) stomachs we indentified (1) reduced (%) gastric emptying (64 ± 2.5 vs. 77.6 ± 0.88), (2) greater reduction in nitregic relaxation (−0.13 ± 0.012 vs. −0.28 ± 0.012), (3) increased nNOS dimerization (0.48 ± 0.02 vs. 0.34 ± 0.05), (4) decreased NO release whether measured at 24 h (0.6 ± 0.04 vs. 1.7 ± 0.22, p < 0.05) or at 48 h (3.4 ± 0.26 vs. 5.0 ± 0.15, p < 0.05) of incubation, (5) decreased GCH-1 (1.9 ± 0.06 vs. 2.3 ± 0.04), DHFR (1.8 ± 0.14 vs. 2.4 ± 0.07) and ERα (2.7 ± 0.4 vs. 3.9 ± 0.4) and (6) increased oxidized biopterin levels and decreased ratio of BH4 versus BH2 + B.
Conclusion
We conclude that chronic estrogen deficiency negatively modifies the function of both BH4 and nNOS thereby contributing to the development of gastroparesis in a FORKO mouse model.




Similar content being viewed by others
References
Parkman HP, Yates K, Hasler WL, Nquyen L, et al. Clinical features of idiopathic gastroparesis vary with sex, body mass, symptom onset, delay in gastric emptying, and gastroparesis severity. Gastroenterology. 2011;140:101–115.
Takahashi T. Pathophysiological significance of neuronal nitric oxide synthase in the gastrointestinal tract. J Gastroenterol. 2003;38:421–430.
Gangula PR, Maner WL, Micci MA, Garfield RE, Pasricha PJ. Diabetes induces sex-dependent changes in neuronal nitric oxide synthase dimerization and function in the rat gastric antrum. Am J Physiol Gastrointest Liver Physiol. 2007;292:725–733.
Huang PL, Dawson TM, Bredt DS, Snyder SH, Fishman MC. Targeted disruption of the neuronal nitric oxide synthase gene. Cell. 1993;75:1273–1286.
Mashimo H, Kjellin A, Goyal RK. Gastric stasis in neuronal nitric oxide synthase-deficient knockout mice. Gastroenterology. 2000;119:766–773.
Klatt P, Pfeiffer S, List BM, Lehner D, et al. Characterization of heme-deficient neuronal nitric-oxide synthase reveals a role for heme in subunit dimerization and binding of the amino acid substrate and tetrahydrobiopterin. J Biol Chem. 1996;271:7336–7342.
Gangula PR, Mukhopadhyay S, Ravella K, Cai S, et al. Tetrahydrobiopterin (BH4), a cofactor for nNOS, restores gastric emptying and nNOS expression in female diabetic rats. Am J Physiol Gastrointest Liver Physiol. 2010;298:692–699.
Hutson WR, Roehrkasse RL, Wald A. Influence of gender and menopause on gastric emptying and motility. Gastroenterology. 1989;96:11–17.
Gangula PR, Sekhar KR, Mukhopadhyay S. Gender bias in gastroparesis: is nitric oxide the answer? Dig Dis Sci. 2011;56:2520–2527.
Danilovich N, Babu PS, Xing W, Gerdes M, Krishnamurthy H, Sairam MR. Estrogen deficiency, obesity, and skeletal abnormalities in follicle-stimulating hormone receptor knockout (FORKO) female mice. Endocrinology. 2000;141:4295–4308.
Gangula PR, Chinnathambi V, Hale AB, Mukhopadhyay S, Channon KM, Ravella K. Impairment of nitrergic system and delayed gastric emptying in low density lipoprotein receptor deficient female mice. Neurogastroenterol Motil. 2011;23:773-e335.
Shah S, Nathan L, Singh R, Fu YS, Chaudhuri G. E2 and not P4 increases NO release from NANC nerves of the gastrointestinal tract: implications in pregnancy. Am J Physiol Regul Integr Comp Physiol. 2001;280:1546–1554.
Kamada Y, Jenkins GJ, Lau M, Dunbar AY, Lowe ER, Osawa Y. Tetrahydrobiopterin depletion and ubiquitylation of neuronal nitric oxide synthase. Brain Res Mol Brain Res. 2005;142:19–27.
Omoto Y, Imamov O, Warner M, Gustafsson JA. Estrogen receptor α and imprinting of the neonatal mouse ventral prostate by estrogen. Proc Natl Acad Sci. 2005;102:1484–1489.
Crabtree MJ, Tatham AL, Al-Wakeel Y, Warrick N, et al. Quantitative regulation of intracellular endothelial nitric-oxide synthase (eNOS) coupling by both tetrahydrobiopterin-eNOS stoichiometry and biopterin redox status: insights from cells with tet-regulated GTP cyclohydrolase I expression. J Biol Chem. 2009;284:1136–1144.
Tayeh MA, Marletta MA. Macrophage oxidation of l-arginine to nitric oxide, nitrite, and nitrate: tetrahydrobiopterin is required as a cofactor. J Biol Chem. 1989;264:19654–19658.
Javeshghani D, Schiffrin EL, Sairam MR, Touyz RM. Potentiation of vascular oxidative stress and nitric oxide-mediated endothelial dysfunction by high-fat diet in a mouse model of estrogen deficiency and hyperandrogenemia. J Am Soc Hypertens. 2009;5:295–305.
Javeshghani D, Touyz RM, Sairam MR, Virdis A, Neves MF, Schiffrin EL. Attenuated responses to angiotensin II in follitropin receptor knockout mice, a model of menopause-associated hypertension. Hypertension. 2003;42:761–767.
Kim NN. Sex steroid hormones in diabetes-induced sexual dysfunction: focus on the female gender. J Sex Med. 2009;3:239–246.
Serova LI, Filipenko M, Schilt N, Veerasirikul M, Sabban EL. Estrogen-triggered activation of GTP cyclohydrolase 1 gene expression: role of estrogen receptor subtypes and interaction with cyclic AMP. Neuroscience. 2006;140:1253–1263.
Miyazaki-Akita A, Hayashi T, Ding QF, Shiraishi H, et al. 17Beta-estradiol antagonizes the down-regulation of endothelial nitric-oxide synthase and GTP cyclohydrolase I by high glucose: relevance to postmenopausal diabetic cardiovascular disease. J Pharmacol Exp Ther. 2007;320:591–598.
Wells CC, Riazi S, Mankhey RW, Bhatti F, Ecelbarger C, Maric C. Diabetic nephropathy is associated with decreased circulating estradiol levels and imbalance in the expression of renal estrogen receptors. Gend Med. 2005;2:227–237.
Cai S, Khoo J, Mussa S, Alp NG, Channon KM. Endothelial nitric oxide synthase dysfunction in diabetic mice: importance of tetrahydrobiopterin in eNOS dimerization. Diabetologia. 2005;48:1933–1940.
Arnal JF, Fontaine C, Billon-Galés A, Favre J, et al. Estrogen receptors and endothelium. Arterioscler Thromb Vasc Biol. 2010;30:1506–1512.
Bender AT, Demady DR, Osawa Y. Ubiquitination of neuronal nitric-oxide synthase in vitro and in vivo. J Biol Chem. 2000;275:17407–17411.
Mukhopadhyay S, Sekhar KR, Hale AB, et al. Loss of NRF2 impairs gastric nitrergic stimulation and function. Free Radic Biol Med. 2011;51:619–625.
Tiano JP, Mauvias-Jarvis F. Importance of estrogen receptors to preserve functional β-cell mass in diabetes. Nat Rev Endocrinol. 2012;8:342–351.
Verrengia M, Sachdeva P, Gaughan J, Fisher RS, Parkman HP. Variation of symptoms during the menstrual cycle in female patients with gastroparesis. Neurogastroenterol Motil. 2011;23:1365–1368.
Mankhey RW, Bhatti F, Maric C. 17beta-Estradiol replacement improves renal function and pathology associated with diabetic nephropathy. Am J Physiol Renal Physiol. 2005;288:F399–F405.
Maric C. Sex, diabetes and the kidney. Am J Physiol Renal Physiol. 2009;296:F680–F688.
Showkat Ali M, Tiscareno-Grejada I, Locovei S, Smiley R, Collins T, Sarosiek J, McCallum R. Gender and estradiol as major factors in the expression and dimerization of nNOS in rats with experimental diabetic gastroparesis. Dig Dis Sci. 2012; 57:2814–2825.
Acknowledgments
We thank Dr. Ayman Al-Hendy for the FORKO and SVE129 mice, and Dr. Diana Marver for review of the manuscript. Financial support for the original research was provided by NIH-NIDDK R21DKO76704 (PG), P60DK020593 pilot project funds (PG), and RCMI G12RR03032 (PG) start-up funds at Meharry Medical College, Nashville, TN, USA.
Conflict of interest
Dr. Gangula has filed a patent application for the use of BH4 in gastroparesis subjects through the University of Texas Medical Branch, Galveston, TX.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ravella, K., Al-Hendy, A., Sharan, C. et al. Chronic Estrogen Deficiency Causes Gastroparesis by Altering Neuronal Nitric Oxide Synthase Function. Dig Dis Sci 58, 1507–1515 (2013). https://doi.org/10.1007/s10620-013-2610-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-013-2610-4