Abstract
Background
Intraperitoneal (i.p.) administration of paclitaxel is useful for treating malignant tumors with peritoneal dissemination, but the therapeutic efficacy is limited. Chemoresistance due to paclitaxel-induced nuclear factor-kappa B (NF-κB) activation is an important cause of suboptimal therapeutic efficacy.
Aims
The purpose of this study was to prove that addition of nafamostat mesilate (FUT-175), a synthetic serine protease inhibitor and an NF-κB inhibitor, to i.p. paclitaxel enhances antitumor effects of paclitaxel against gastric cancer with peritoneal dissemination.
Methods
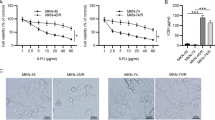
In vitro, we assessed NF-κB activity and apoptosis in response to treatment with FUT-175 alone, paclitaxel alone, or a combination of FUT-175 and paclitaxel in a human gastric cancer cell line (MKN-45). In vivo, we established peritoneal dissemination in nude mice by i.p. injection of MKN-45 cells. The animals received i.p. injections of FUT-175 alone three times a week (FUT-175 group), of paclitaxel alone once a week (paclitaxel group), or a combination of FUT-175 and paclitaxel (combination group) three times and once a week, respectively.
Results
In the combination group, paclitaxel-induced NF-κB activation was inhibited and apoptosis was enhanced in comparison with those in the other groups both in vitro and in vivo. In the combination group, number and weight of peritoneal nodules were significantly lower than those in the paclitaxel group (p = 0.0009 and p = 0.0417, respectively). In the survival analysis, the combination group had a significantly better survival than the paclitaxel group (p = 0.0048).
Conclusion
FUT-175 enhances the antitumor effect of i.p. paclitaxel against gastric cancer with peritoneal dissemination by inhibiting NF-κB activation in mice.






Similar content being viewed by others
References
Ries L, Eisner M, Kosary C. SEER cancer statistics review, 1975–2000. Bethesda, MD: National Cancer Institute; 2003.
Saito H, Tsujitani S, Kondo A, et al. Expression of vascular endothelial growth factor correlates with hematogenous recurrence in gastric carcinoma. Surgery. 1999;125:195–201.
Ajani JA, Fairweather J, Dumas P, et al. Phase II study of Taxol in patients with advanced gastric carcinoma. Cancer J Sci Am. 1998;4:269–274.
Rowinsky EK. Update on the antitumor activity of pacitaxel in clinical trials. Ann Pharmacother. 1994;28:S18–S22.
Yonemura Y, Bando E, Kawamura T, et al. Cytoreduction and intraperitoneal chemotherapy for carcinomatosis from gastric cancer. Cancer Treat Res. 2007;134:357–373.
Ishigami H, Kitayama J, Kaisaki S, et al. Phase II study of weekly intravenous and intraperitoneal paclitaxel combined with S-1 for advanced gastric cancer with peritoneal metastasis. Ann Oncol. 2010;21:67–70.
Armstrong DK, Bundy B, Wenzel L, et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med. 2006;354:34–43.
Patel NM, Nozaki S, Shortle NH, et al. Paclitaxel sensitivity of breast cancer cells with constitutively active NF-kappaB is enhanced by IkappaBalpha super-repressor and parthenolide. Oncogene. 2000;19:4159–4169.
Dong QG, Sclabas GM, Fujioka S, et al. The function of multiple IkappaB: NF-kappaB complexes in the resistance of cancer cells to Taxol-induced apoptosis. Oncogene. 2002;21:6510–6519.
Chen F, Castranova V, Shi X. New insights into the role of nuclear factor-kappaB in cell growth regulation. Am J Pathol. 2001;159:387–397.
Beg AA, Baltimore D. An essential role for NF-kappaB in preventing TNF-alpha-induced cell death. Science. 1996;274:782–784.
Van Antwerp DJ, Martin SJ, Kafri T, et al. Suppression of TNF-alpha-induced apoptosis by NF-kappaB. Science. 1996;274:787–789.
Karin M, Lin A. NF-kappaB at the crossroads of life and death. Nat Immunol. 2002;3:221–227.
Sasaki N, Morisaki T, Hashizume K, et al. Nuclear factor-kappaB p65 (RelA) transcription factor is constitutively activated in human gastric carcinoma tissue. Clin Cancer Res. 2001;7:4136–4142.
Yamanaka N, Sasaki N, Tasaki A, et al. Nuclear factor-kappaB p65 is a prognostic indicator in gastric carcinoma. Anticancer Res. 2004;24:1071–1075.
Long YM, Ye S, Rong J, et al. Nuclear factor kappa B: a marker of chemotherapy for human stage IV gastric carcinoma. World J Gastroenterol. 2008;14:4739–4744.
Wang W, Abbruzzese JL, Evans DB, et al. The nuclear factor-kappa B RelA transcription factor is constitutively activated in human pancreatic adenocarcinoma cells. Clin Cancer Res. 1999;5:119–127.
Fujioka S, Sclabas GM, Schmidt C, et al. Function of nuclear factor kappaB in pancreatic cancer metastasis. Clin Cancer Res. 2003;9:346–354.
Wang CY, Cusack JC Jr, Liu R, et al. Control of inducible chemoresistance: enhanced anti-tumor therapy through increased apoptosis by inhibition of NF-kappaB. Nat Med. 1999;5:412–417.
Yeh PY, Chuang SE, Yeh KH, et al. Increase of the resistance of human cervical carcinoma cells to cisplatin by inhibition of the MEK to ERK signaling pathway partly via enhancement of anticancer drug-induced NF kappa B activation. Biochem Pharmacol. 2002;63:1423–1430.
Wang W, McLeod HL, Cassidy J. Disulfiram-mediated inhibition of NF-kappaB activity enhances cytotoxicity of 5-fluorouracil in human colorectal cancer cell lines. Int J Cancer. 2003;104:504–511.
Arlt A, Gehrz A, Müerköster S, et al. Role of NF-kappaB and Akt/PI3 K in the resistance of pancreatic carcinoma cell lines against gemcitabine-induced cell death. Oncogene. 2003;22:3243–3251.
Oyaizu H, Adachi Y, Okumura T, et al. Proteasome inhibitor 1 enhances paclitaxel-induced apoptosis in human lung adenocarcinoma cell line. Oncol Rep. 2001;8:825–829.
Fujii S, Hitomi Y. New synthetic inhibitors of C1r, C1 esterase, thrombin, plasmin, kallikrein and trypsin. Biochim Biophys Acta. 1981;661:342–345.
Aoyama T, Ino Y, Ozeki M, et al. Pharmacological studies of FUT-175, nafamstat mesilate. I. Inhibition of protease activity in in vitro and in vivo experiments. Jpn J Pharmacol. 1984;35:203–227.
Iwaki M, Ino Y, Motoyoshi A, et al. Pharmacological studies of FUT-175, nafamostat mesilate. V. Effects on the pancreatic enzymes and experimental acute pancreatitis in rats. Jpn J Pharmacol. 1986;41:155–162.
Yoshikawa T, Murakami M, Furukawa Y, et al. Effects of FUT-175, a new synthetic protease inhibitor on endotoxin-induced disseminated intravascular coagulation in rats. Haemostasis. 1983;13:374–378.
Ohtake Y, Hirasawa H, Sugai T, et al. Nafamostat mesylate as anticoagulant in continuous hemofiltration and continuous hemodiafiltration. Contrib Nephrol. 1991;93:215–217.
Uwagawa T, Li Z, Chang Z, et al. Mechanisms of synthetic serine protease inhibitor (FUT-175)-mediated cell death. Cancer. 2007;109:2142–2153.
Furukawa K, Iida T, Shiba H, et al. Anti-tumor effect by inhibition of NF-kappaB activation using nafamostat mesilate for pancreatic cancer in a mouse model. Oncol Rep. 2010;24:843–850.
Uwagawa T, Chiao PJ, Gocho T, et al. Combination chemotherapy of nafamostat mesilate with gemcitabine for pancreatic cancer targeting NF-kappaB activation. Anticancer Res. 2009;29:3173–3178.
Uwagawa T, Misawa T, Sakamoto T, et al. A phase I study of full-dose gemcitabine and regional arterial infusion of nafamostat mesilate for advanced pancreatic cancer. Ann Oncol. 2009;20:239–243.
Uwagawa T, Misawa T, Tsutsui N, et al. Phase II study of gemcitabine in combination with regional arterial infusion of nafamostat mesilate for advanced pancreatic cancer. Am J Clin Oncol. 2011. doi:10.1097/COC.0b013e31823a53b2.
Fujiwara Y, Furukawa K, Shimada Y, et al. Combination paclitaxel and inhibitor of nuclear factor κB activation improves therapeutic outcome for model mice with peritoneal dissemination of pancreatic cancer. Pancreas. 2011;40:600–607.
Shimada Y, Fukuda T, Aoki K, et al. A protocol for immunoaffinity separation of the accumulated ubiquitin-protein conjugates solubilized with sodium dodecyl sulfate. Anal Biochem. 2008;377:77–82.
Kumar N. Taxol-induced polymerization of purified tubulin: mechanism of action. J Biol Chem. 1981;256:10435–10441.
Schiff PB, Horwitz SB. Taxol assembles tubulin in the absence of exogenous guanosine 5′-triphosphate or microtubule-associated proteins. Biochemistry. 1981;20:3247–3252.
Rowinsky EK, Donehower RC, Jones RJ, et al. Microtubule changes and cytotoxicity in leukemic cell lines treated with Taxol. Cancer Res. 1988;48:4093–4100.
Schiff PB, Faut J, Horwitz SB. Promotion of microtubule assembly in vitro by Taxol. Nature. 1979;277:665–667.
Horwitz SB. Mechanism of action taxol. Trends Pharmacol Sci. 1992;13:134–136.
Jacquet P, Sugarbaker PH. Peritoneal-plasma barrier. Cancer Treat Res. 1996;82:53–63.
Gelmon K. The taxoids: paclitaxel and docetaxel. Lancet. 1994;344:1267–1272.
Markman M. Intraperitoneal antineoplastic agents for tumors principally confined to the peritoneal cavity. Cancer Treat Rev. 1986;13:219–242.
Chang YF, Li LL, Wu CW, et al. Paclitaxel-induced apoptosis in human gastric carcinoma cell lines. Cancer. 1996;77:14–18.
Wang CY, Mayo MW, Korneluk RG, et al. NF-kappaB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science. 1998;281:1680–1683.
Mabuchi S, Ohmichi M, Nishio Y, et al. Inhibition of inhibitor of nuclear factor-kappaB phosphorylation increases the efficacy of paclitaxel in in vitro and in vivo ovarian cancer models. Clin Cancer Res. 2004;10:7645–7654.
Aggarwal BB, Shishodia S, Takada Y, et al. Curcumin suppresses the paclitaxel-induced nuclear factor-kappaB pathway in breast cancer cells and inhibits lung metastasis of human breast cancer in nude mice. Clin Cancer Res. 2005;11:7490–7498.
Inoue M, Matsumoto S, Saito H, et al. Intraperitoneal administration of a small interfering RNA targeting nuclear factor-kappa B with paclitaxel successfully prolongs the survival of xenograft model mice with peritoneal metastasis of gastric cancer. Int J Cancer. 2008;123:2696–2701.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haruki, K., Shiba, H., Fujiwara, Y. et al. Inhibition of Nuclear Factor-κB Enhances the Antitumor Effect of Paclitaxel Against Gastric Cancer with Peritoneal Dissemination in Mice. Dig Dis Sci 58, 123–131 (2013). https://doi.org/10.1007/s10620-012-2311-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2311-4



